Advanced Ketoreductase Mutant Enables Efficient Synthesis of High-Purity Darunavir Intermediates
Advanced Ketoreductase Mutant Enables Efficient Synthesis of High-Purity Darunavir Intermediates
The pharmaceutical industry continuously seeks robust biocatalytic solutions to replace traditional chemical synthesis, particularly for complex chiral intermediates required in antiretroviral therapy. Patent CN111662889B introduces a groundbreaking ketoreductase mutant derived from Starmerella magnoliae, specifically engineered to produce the critical Darunavir intermediate (2S,3S)-1-chloro-3-tert-butoxycarbonylamino-4-phenyl-2-butanol. This innovation addresses long-standing challenges in enzymatic stability and substrate tolerance, offering a pathway to significantly lower manufacturing costs and higher purity profiles. By leveraging site-directed mutagenesis to introduce key amino acid changes, the disclosed technology achieves a dramatic improvement in catalytic efficiency compared to wild-type enzymes. For global procurement leaders and R&D directors, this patent represents a pivotal shift towards more sustainable and economically viable production methods for high-value HIV protease inhibitor precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the Darunavir intermediate has relied heavily on chemical routes involving aluminum isopropoxide reductions or early-generation biocatalysts with significant operational drawbacks. Traditional chemical methods, while established, often necessitate the use of hazardous reagents and generate substantial metal-containing waste streams that require costly remediation. Furthermore, earlier biological approaches, such as those utilizing RasADH from Ralstonia sp., were severely limited by low substrate tolerance, typically capping at concentrations around 10g/L. This low loading capacity necessitates large reactor volumes and extensive downstream processing to recover the product, leading to inflated operational expenditures. Additionally, the optical purity achieved by these legacy enzymes often hovered around 90%, which is insufficient for stringent pharmaceutical specifications without additional purification steps. The reliance on expensive cofactors like NADPH without efficient recycling mechanisms further exacerbated the cost structure, rendering these processes less attractive for large-scale commercial adoption.
The Novel Approach
The novel approach detailed in the patent utilizes a specifically engineered ketoreductase mutant, designated as Sma-1, which overcomes the kinetic and thermodynamic barriers of previous generations. This mutant exhibits a remarkable ability to tolerate substrate concentrations as high as 110g/L, representing an order-of-magnitude improvement over prior art. The process employs a highly efficient coenzyme recycling system driven by isopropanol, allowing for a substrate-to-NADP ratio of up to 1100:1, which drastically reduces the dependency on expensive external cofactors. The reaction proceeds under mild conditions, primarily in an aqueous environment with minimal organic co-solvents, aligning perfectly with green chemistry principles. By eliminating the need for heavy metal catalysts and reducing the volume of solvent required per unit of product, this method offers a streamlined, cost-effective alternative that is inherently safer and more environmentally compliant. The enhanced stereoselectivity ensures that the desired (2S,3S) enantiomer is produced with high fidelity, minimizing the formation of genotoxic impurities.
Mechanistic Insights into Sma-1 Catalyzed Asymmetric Reduction
The superior performance of the Sma-1 mutant is rooted in three specific amino acid substitutions: I54E, S85A, and K182V. These mutations are strategically located within the enzyme's active site or substrate binding pocket, altering the electrostatic environment and steric constraints to favor the binding of the bulky chloroketone substrate. The substitution of Isoleucine with Glutamic acid at position 54 likely introduces a negative charge that stabilizes the transition state or facilitates proton transfer during the hydride addition from NADPH. Similarly, the Serine to Alanine mutation at position 85 reduces steric hindrance, allowing for tighter accommodation of the tert-butoxycarbonyl group of the substrate. The Lysine to Valine change at position 182 may further optimize the hydrophobic interactions necessary for high-affinity binding. Collectively, these modifications result in an enzyme variant with alcohol dehydrogenase activity that is 2 to 15 times higher than the wild-type sequence, enabling rapid conversion rates even at high substrate loads.
Impurity control is intrinsically managed through the high stereospecificity of the Sma-1 mutant, which selectively reduces the pro-chiral ketone to the desired (2S,3S) alcohol configuration. The enzymatic mechanism avoids the non-specific reduction pathways often seen in chemical catalysis, thereby suppressing the formation of diastereomeric impurities that are difficult to separate. The use of isopropanol not only serves as a co-solvent to solubilize the hydrophobic substrate but also acts as the hydrogen donor for regenerating NADPH from NADP+, creating a closed-loop catalytic cycle. This self-sustaining cycle minimizes the accumulation of oxidized cofactor byproducts and ensures that the reaction equilibrium is driven strongly towards product formation. The result is a clean reaction profile where the primary byproduct is acetone, which is easily removed during workup, simplifying the purification process and ensuring the final API intermediate meets rigorous quality standards.
How to Synthesize Darunavir Intermediate Efficiently
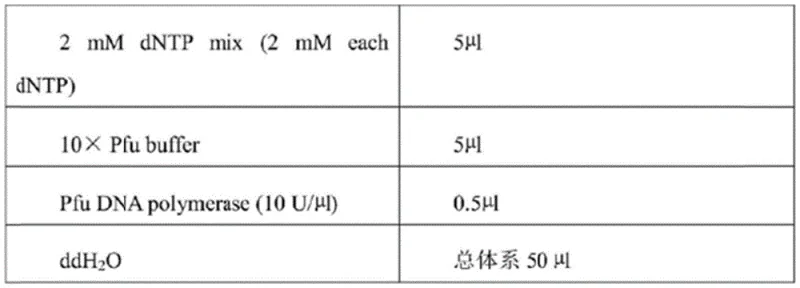
The implementation of this biocatalytic route begins with the construction of a robust expression system, followed by optimized fermentation and bioconversion steps. The process starts with the synthesis of the polynucleotide sequence encoding the mutant enzyme, which is then cloned into a suitable expression vector for production in E. coli. Following cell cultivation and induction, the crude enzyme is harvested and directly applied to the bioconversion reaction containing the chloroketone substrate. The entire workflow is designed for scalability, moving seamlessly from shake-flask optimization to fed-batch fermentation in large bioreactors. The following image illustrates the genetic construct used to express the high-activity Sma-1 enzyme, which is the cornerstone of this efficient manufacturing process.
- Construct the expression plasmid by synthesizing the polynucleotide sequence (SEQ ID NO. 1) and cloning it into a vector such as pET30a for expression in E. coli.
- Perform fed-batch fermentation of the recombinant host cells in a controlled bioreactor, inducing expression with IPTG to produce the crude enzyme Sma-1.
- Conduct the bioconversion reaction by mixing the substrate (3S-1-chloro-3-tert-butoxycarbonylamino-4-phenyl-2-butanone) with the crude enzyme and isopropanol for coenzyme recycling at 30°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the Sma-1 mutant technology translates into tangible strategic advantages regarding cost stability and supply security. The shift from metal-catalyzed chemistry to biocatalysis eliminates the volatility associated with precious metal pricing and the regulatory burdens of heavy metal residue testing. The high substrate loading capacity means that existing manufacturing infrastructure can produce significantly more product per batch without requiring capital-intensive expansion of reactor farms. This intensification of the process leads to a substantial reduction in utility consumption, including water and energy, as less solvent needs to be heated, cooled, or distilled. Furthermore, the aqueous nature of the reaction medium simplifies waste treatment protocols, reducing the environmental compliance costs that often burden chemical synthesis facilities. These factors combine to create a resilient supply chain capable of meeting high-volume demand with consistent quality and lower overall production costs.
- Cost Reduction in Manufacturing: The elimination of expensive aluminum-based reagents and the drastic reduction in cofactor usage through efficient recycling directly lower the bill of materials. The ability to run reactions at 110g/L substrate concentration means that fixed costs such as labor, equipment depreciation, and facility overhead are amortized over a much larger output volume. Additionally, the simplified downstream processing, driven by high conversion rates and fewer byproducts, reduces the consumption of chromatography resins and extraction solvents. This holistic reduction in variable and fixed costs ensures a highly competitive price point for the final intermediate, providing a buffer against market fluctuations.
- Enhanced Supply Chain Reliability: The reliance on recombinant E. coli fermentation leverages a well-established, globally available biological platform that is less susceptible to geopolitical supply disruptions than specialized chemical reagents. The robustness of the Sma-1 mutant ensures consistent batch-to-batch performance, minimizing the risk of production failures or off-spec material that could delay downstream API synthesis. The use of common, commodity-grade chemicals like isopropanol and glucose for the process further secures the supply line against raw material shortages. This reliability is critical for maintaining continuous production schedules for life-saving antiretroviral medications, ensuring that patients have uninterrupted access to therapy.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in fed-batch fermentation systems that can be readily expanded to industrial scales ranging from hundreds of kilograms to multi-ton annual production capacities. The green chemistry profile, characterized by water as the primary solvent and the absence of toxic heavy metals, aligns with increasingly stringent global environmental regulations. This compliance future-proofs the manufacturing process against tightening emission standards and reduces the liability associated with hazardous waste disposal. Companies adopting this technology can market their supply chain as sustainable and eco-friendly, adding brand value and meeting the ESG (Environmental, Social, and Governance) goals of major pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ketoreductase technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these specifics is essential for making informed decisions about process adoption and supplier qualification.
Q: What specific mutations define the novel ketoreductase mutant Sma-1?
A: The mutant Sma-1 is characterized by three specific amino acid substitutions compared to the wild-type sequence: I54E, S85A, and K182V. These mutations result in significantly enhanced alcohol dehydrogenase activity.
Q: How does the substrate loading capacity of this mutant compare to prior art methods?
A: The Sma-1 mutant supports a substrate concentration as high as 110g/L, which is approximately 10 times higher than the 10g/L reported in previous biological methods using RasADH, drastically improving volumetric productivity.
Q: What is the mechanism for coenzyme recycling in this process?
A: The process utilizes isopropanol as a co-substrate to regenerate the expensive cofactor NADP in situ. This allows for a substrate-to-NADP molar ratio as high as 1100:1, minimizing cofactor costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Darunavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of key antiviral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from pilot studies to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities designed to meet stringent purity specifications required for pharmaceutical applications. We are committed to delivering the Sma-1 mediated process with the highest levels of operational excellence, guaranteeing consistency and reliability for your global supply chain.
We invite you to engage with our technical procurement team to discuss how this innovative biocatalytic route can optimize your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this enzymatic method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a partnership that drives both efficiency and growth in the competitive pharmaceutical landscape.
