Advanced Transaminase Mutants for Scalable Chiral Amine Manufacturing and Process Optimization
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards greener, more efficient synthetic methodologies, particularly in the production of optically pure chiral amines which serve as critical building blocks for numerous active pharmaceutical ingredients. Patent CN116064449A introduces a groundbreaking advancement in this sector by disclosing a novel transaminase mutant derived from Xenophilus sp. AP218F, engineered through rational design to overcome the longstanding limitations of biocatalytic amination. This technology addresses the persistent challenge of low catalytic efficiency and poor stability associated with wild-type enzymes when processing sterically hindered substrates, offering a robust solution for the scalable manufacturing of complex molecular structures. By leveraging specific site-directed mutations, this innovation enables the direct asymmetric synthesis of bulky chiral amines with unprecedented conversion rates and thermal resilience, positioning it as a vital tool for any reliable pharmaceutical intermediate supplier seeking to optimize their production capabilities.
Furthermore, the implications of this patent extend beyond mere laboratory curiosity, representing a tangible opportunity for significant cost reduction in chiral amine manufacturing through the simplification of process workflows. The ability to utilize a biocatalyst that maintains high activity under industrial conditions reduces the dependency on harsh chemical reagents and energy-intensive purification steps, thereby aligning with modern sustainability goals while enhancing economic viability. As global demand for high-purity API intermediate continues to rise, the adoption of such advanced enzymatic technologies becomes a strategic imperative for maintaining competitive advantage in the market. This report analyzes the technical merits and commercial potential of this transaminase mutant, providing actionable insights for R&D directors and procurement managers aiming to integrate superior biocatalytic routes into their supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis of chiral amines often relies on asymmetric hydrogenation or chiral resolution techniques, both of which present substantial drawbacks in terms of environmental impact and operational complexity. Asymmetric hydrogenation typically requires the use of expensive transition metal catalysts, such as rhodium or ruthenium complexes, which necessitate rigorous removal processes to meet stringent regulatory limits on residual metals in pharmaceutical products. Additionally, these chemical methods frequently operate under high pressure and temperature conditions, increasing safety risks and energy consumption while potentially compromising the integrity of sensitive functional groups within the molecule. On the biological front, wild-type transaminases have historically struggled with substrate specificity, particularly when encountering ketones with bulky substituents adjacent to the carbonyl group, leading to negligible conversion rates and inefficient process economics.
The narrow substrate scope of natural enzymes often forces manufacturers to resort to multi-step synthetic routes or inefficient kinetic resolutions that inherently cap the maximum theoretical yield at fifty percent, generating significant amounts of unwanted isomeric waste that must be separated and disposed of. This inefficiency not only drives up the cost of goods sold but also complicates the waste management profile of the manufacturing facility, creating bottlenecks in the commercial scale-up of complex pharmaceutical intermediates. Furthermore, the thermal instability of many wild-type biocatalysts restricts their utility to mild operating conditions, which can result in slow reaction kinetics and prolonged batch cycles that hinder overall production throughput. These cumulative factors underscore the critical need for engineered solutions that can bridge the gap between laboratory feasibility and industrial robustness.
The Novel Approach
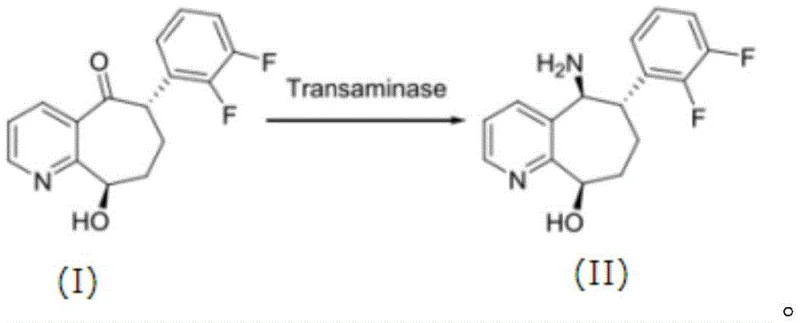
The technology disclosed in CN116064449A offers a transformative alternative by utilizing a specifically engineered transaminase mutant that has been optimized through site-directed mutagenesis to accommodate sterically demanding substrates with high efficiency. Unlike the wild-type enzyme which exhibits less than one percent activity on bulky ketones, the mutant variant achieves conversion rates exceeding sixty percent within twelve hours, demonstrating a dramatic enhancement in catalytic power that directly translates to reduced reactor volume requirements and shorter processing times. This novel approach eliminates the need for heavy metal catalysts entirely, replacing them with a renewable biocatalytic system that operates under mild aqueous conditions, thereby simplifying the downstream purification process and ensuring a cleaner final product profile. The strategic modification of key amino acid residues within the enzyme's active site allows for precise control over stereoselectivity, ensuring the production of the desired optical isomer with high purity.
Moreover, the enhanced thermal stability of the mutant enzyme permits operation at elevated temperatures, such as fifty degrees Celsius, without significant loss of activity, which further accelerates reaction rates and improves solubility of hydrophobic substrates. This robustness is a key differentiator that facilitates the commercial scale-up of complex pharmaceutical intermediates, as it allows for more flexible process parameters and reduced sensitivity to minor fluctuations in fermentation or reaction conditions. By adopting this novel biocatalytic route, manufacturers can achieve a more sustainable and cost-effective production model that aligns with the increasing regulatory and consumer pressure for green chemistry solutions. The ability to synthesize high-purity chiral amines directly from prochiral ketones without the need for protecting groups or harsh reagents represents a significant leap forward in process chemistry.
Mechanistic Insights into Site-Directed Mutagenesis and Catalytic Enhancement
The core of this technological breakthrough lies in the precise rational design of the transaminase protein structure, where specific amino acid residues have been substituted to alter the geometry and electrostatic environment of the active site pocket. The patent details a series of mutations, including W60A, F89K, S121D, Y153A, E226R, V261A, Q380F, and A417F, which collectively work to expand the binding cavity and reduce steric clashes with large substrate molecules. For instance, the substitution of Tryptophan at position 60 with Alanine removes a bulky aromatic side chain that would otherwise obstruct the entry of sterically hindered ketones, while the introduction of charged residues like Lysine or Arginine at other positions may enhance substrate binding through favorable electrostatic interactions. This level of molecular engineering ensures that the enzyme can effectively recognize and transform substrates that were previously considered inaccessible to biocatalysis, opening new avenues for the synthesis of diverse chemical scaffolds.
In addition to improving substrate acceptance, these mutations significantly enhance the structural rigidity and thermal stability of the enzyme, as evidenced by the mutant's half-life of greater than forty-eight hours at fifty degrees Celsius compared to merely twelve hours for the wild-type. This increased stability is crucial for industrial applications where enzymes are subjected to prolonged stress during fermentation and catalytic cycles, as it reduces the frequency of enzyme replenishment and lowers the overall biocatalyst cost per kilogram of product. The mechanistic understanding of these mutations allows for the prediction of enzyme behavior under various process conditions, enabling engineers to design reactors that maximize catalytic efficiency while minimizing degradation. Such deep mechanistic insights are invaluable for R&D teams aiming to implement reducing lead time for high-purity chiral amines in their development pipelines, as it provides a clear pathway from gene sequence to commercial production.
How to Synthesize Bulky Chiral Amines Efficiently
The implementation of this transaminase technology involves a streamlined workflow that begins with the fermentation of recombinant Escherichia coli strains harboring the mutant gene, followed by the preparation of whole-cell catalysts for the biotransformation step. Detailed standard operating procedures for the expression, harvesting, and application of the biocatalyst are essential to ensure consistent performance across different production batches and scales. The process leverages the high cell density fermentation capabilities of modern bioprocessing to generate sufficient enzyme quantities economically, while the whole-cell format simplifies the handling and reuse of the biocatalyst in repeated batch operations. Understanding the nuances of this synthesis route is critical for technical teams looking to integrate this capability into their existing infrastructure.
- Prepare the recombinant engineered bacteria expressing the specific transaminase mutant (e.g., SEQ ID NO.3) via fermentation in optimized media.
- Conduct the whole-cell catalytic reaction using the wet cells, ketone substrate, and amine donor at controlled pH 8.5 and temperature 50°C.
- Perform downstream processing including pH adjustment, filtration, and solvent extraction to isolate the high-purity chiral amine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this engineered transaminase mutant offers compelling advantages that directly impact the bottom line and operational reliability of chemical manufacturing operations. The elimination of expensive heavy metal catalysts and the associated scavenging resins results in substantial cost savings on raw materials and waste disposal, while the higher conversion rates reduce the volume of starting materials required to produce a given amount of final product. This efficiency gain is particularly significant for high-value pharmaceutical intermediates where material costs constitute a major portion of the total production expense, allowing companies to improve their margins or offer more competitive pricing to their customers. Furthermore, the robust nature of the enzyme reduces the risk of batch failures due to catalyst instability, ensuring a more predictable and reliable supply of critical intermediates.
- Cost Reduction in Manufacturing: The biocatalytic process fundamentally alters the cost structure by removing the need for precious metal catalysts and high-pressure hydrogenation equipment, leading to drastically simplified capital expenditure requirements and lower operating costs. The high specificity of the enzyme minimizes the formation of by-products, which reduces the burden on downstream purification units and lowers solvent consumption, contributing to a more lean and efficient manufacturing process. Additionally, the ability to operate at higher substrate concentrations due to improved enzyme tolerance further enhances volumetric productivity, maximizing the output of existing fermentation and reaction assets without the need for immediate capacity expansion.
- Enhanced Supply Chain Reliability: The thermal stability and robustness of the mutant enzyme mitigate the risks associated with catalyst degradation during storage and transport, ensuring that the biocatalyst remains effective even under less-than-ideal logistical conditions. This reliability translates to fewer disruptions in the production schedule and a more consistent ability to meet delivery commitments, which is paramount for maintaining strong relationships with downstream pharmaceutical clients. The use of a renewable biological catalyst also diversifies the supply base away from geographically concentrated sources of rare earth metals, reducing exposure to geopolitical supply risks and price volatility in the raw materials market.
- Scalability and Environmental Compliance: The aqueous nature of the biocatalytic reaction and the absence of toxic heavy metals simplify the regulatory compliance landscape, making it easier to obtain necessary environmental permits and adhere to strict emission standards. The process generates less hazardous waste and consumes less energy compared to traditional chemical methods, aligning with corporate sustainability goals and enhancing the brand reputation of the manufacturer as a responsible producer. Scalability is further supported by the compatibility of the enzyme with standard fermentation and bioprocessing equipment, allowing for a seamless transition from pilot scale to multi-ton commercial production without significant process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this transaminase mutant technology, based on the data provided in the patent documentation. These answers are designed to clarify the operational advantages and feasibility of adopting this biocatalytic route for the production of chiral amines. Understanding these details helps stakeholders make informed decisions about integrating this technology into their manufacturing portfolios.
Q: How does this transaminase mutant improve upon wild-type enzymes for bulky substrates?
A: The mutant features specific amino acid substitutions (e.g., W60A, F89K) that enlarge the active site pocket, allowing sterically hindered ketones to bind effectively, resulting in conversion rates exceeding 60% compared to less than 1% for wild-type enzymes.
Q: What are the thermal stability advantages of this biocatalyst?
A: The engineered enzyme demonstrates a half-life of greater than 48 hours at 50°C, significantly outperforming the wild-type which degrades within 12 hours, enabling robust operation at elevated industrial temperatures.
Q: Does this process eliminate the need for heavy metal catalysts?
A: Yes, the biocatalytic transamination replaces traditional chemical hydrogenation or resolution methods, removing the requirement for expensive transition metal catalysts and subsequent heavy metal scavenging steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Transaminase Mutant Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in modernizing the production of high-value chemical intermediates, and we are committed to supporting our partners in leveraging these innovations for commercial success. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory technologies like the transaminase mutant described in CN116064449A can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of material meets the exacting standards required by the global pharmaceutical industry, providing you with a secure and high-quality supply source.
We invite you to engage with our technical procurement team to discuss how this specific biocatalytic route can be tailored to your specific synthesis needs, offering a Customized Cost-Saving Analysis that quantifies the potential economic benefits for your organization. By requesting specific COA data and route feasibility assessments from us, you can gain a clearer understanding of how this technology fits into your broader supply chain strategy and identify opportunities for optimization. Let us collaborate to engineer a more efficient, sustainable, and cost-effective future for your chemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
