Advanced Transaminase Mutants for High-Purity Sitagliptin Manufacturing and Commercial Scale-Up
Advanced Transaminase Mutants for High-Purity Sitagliptin Manufacturing and Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of critical active pharmaceutical ingredients (APIs), and the synthesis of sitagliptin, a premier DPP-4 inhibitor for type II diabetes, stands as a prime example of this evolution. Patent CN112094856B introduces a groundbreaking advancement in this field by disclosing a series of highly engineered transaminase mutants derived from Neosartorya fischeri. These novel biocatalysts, particularly the culminating mutant AFATA21X, demonstrate remarkably improved enzyme activity and exceptional stereoselectivity, capable of efficiently catalyzing the conversion of sitagliptin precursor ketone into sitagliptin with an optical purity exceeding 99.95% e.e. This technological leap represents a significant departure from traditional chemical methods, offering a robust, green, and economically viable solution for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of the chiral (R)-amino group in sitagliptin synthesis has been a formidable challenge, often relying on chemically intensive routes that burden supply chains with high costs and environmental hazards. Traditional methods, such as those utilizing chiral ruthenium phosphorus catalysts for asymmetric hydrogenation, while effective, require expensive reagents and sophisticated handling of precious metals. Furthermore, alternative routes involving chiral rhodium catalysts, although recognized for their efficiency, still suffer from the inherent drawbacks of transition metal catalysis, including the risk of heavy metal contamination in the final product. These chemical processes necessitate rigorous and costly purification steps to meet stringent regulatory limits on residual metals, thereby inflating the overall cost of goods sold (COGS) and complicating the waste management profile for manufacturers.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages the power of directed evolution to create transaminase mutants that operate with unparalleled specificity and efficiency. By systematically mutating key amino acid residues in the wild-type enzyme, researchers have developed variants like AFATA21X that not only tolerate high substrate concentrations but also drive the reaction equilibrium towards the desired chiral amine product. This biocatalytic strategy eliminates the need for expensive chiral metal catalysts and harsh reaction conditions, replacing them with a mild, aqueous-based system that utilizes isopropylamine as a cheap and abundant amine donor. The result is a streamlined process that inherently produces high-purity sitagliptin, drastically reducing the burden on downstream processing and aligning perfectly with the principles of green chemistry.
Mechanistic Insights into Transaminase-Catalyzed Asymmetric Synthesis
The core of this technological breakthrough lies in the precise engineering of the enzyme's active site to accommodate the bulky sitagliptin precursor ketone. The wild-type transaminase, originally derived from Neosartorya fischeri, undergoes a multi-round mutation process where specific residues such as L56, E115, W147, and I237 are altered to reduce steric hindrance and enhance substrate binding affinity. The cumulative effect of mutations like H53L, K84H, and G127F creates a more spacious and hydrophobic pocket that favors the binding of the trifluorophenyl and triazolopyrazine moieties of the substrate. This structural optimization allows the enzyme to maintain high catalytic turnover even at elevated substrate loadings, a critical factor for industrial feasibility.
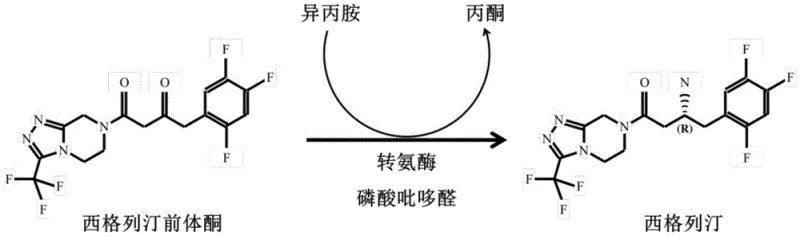
Furthermore, the mechanism ensures exceptional stereocontrol through the rigid geometry of the pyridoxal phosphate (PLP) cofactor within the mutated active site. The enzyme facilitates the transfer of the amino group from isopropylamine to the prochiral ketone substrate via a ping-pong bi-bi mechanism, strictly enforcing the formation of the (R)-enantiomer. The patent data indicates that the final mutant AFATA21X achieves a sitagliptin generation rate of up to 96.7% in crude enzyme systems, with an e.e. value consistently above 99.95%. This level of impurity control is intrinsic to the biocatalyst, meaning that the formation of the unwanted (S)-isomer is virtually suppressed at the source, thereby simplifying the impurity profile and ensuring consistent product quality batch after batch.
How to Synthesize Sitagliptin Efficiently
Implementing this advanced biocatalytic route requires a systematic approach to strain construction and process optimization, as outlined in the patent examples. The process begins with the cloning of the optimized gene sequence into a suitable expression vector, followed by fermentation in E. coli to produce the biocatalyst. The subsequent transformation step utilizes whole cells or crude enzyme lysates in a buffered system containing the necessary cofactors and amine donors. For a comprehensive understanding of the specific operational parameters, including induction times, cell densities, and workup procedures, please refer to the detailed standardized synthesis steps provided below.
- Construct the expression vector containing the gene for the AFATA21X mutant (SEQ ID NO: 15) and transform into E. coli BL21(DE3).
- Culture the transformed bacteria in TB medium, induce expression with IPTG at OD600 0.6-0.8, and harvest cells via centrifugation.
- Perform the biotransformation in a buffer system (pH 8.5) with isopropylamine as the amine donor and PLP as a cofactor at 45°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic technology offers profound strategic advantages that extend far beyond simple yield improvements. By shifting from a metal-catalyzed chemical process to a biocatalytic one, companies can achieve substantial cost savings through the elimination of expensive chiral ligands and precious metal catalysts. The simplified downstream processing, driven by the high selectivity of the enzyme, reduces solvent consumption and waste disposal costs, directly impacting the bottom line. Moreover, the reliance on renewable biological resources rather than finite metal ores enhances the long-term sustainability and resilience of the supply chain against raw material volatility.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the removal of costly transition metal catalysts and the associated purification steps required to meet heavy metal specifications. Since the enzymatic reaction proceeds with high specificity in an aqueous environment, the need for extensive chromatographic separation is minimized, leading to significantly reduced solvent usage and energy consumption. This streamlined workflow translates to a lower cost of goods sold (COGS) and a more competitive pricing structure for the final API, allowing manufacturers to capture greater margin in a crowded market.
- Enhanced Supply Chain Reliability: Utilizing a genetically engineered E. coli strain for production ensures a highly reliable and scalable source of the catalyst, independent of the geopolitical fluctuations often associated with the mining and refining of rare earth metals. The fermentation process is robust and can be easily scaled from laboratory to industrial volumes, ensuring consistent supply continuity. Additionally, the stability of the mutant enzyme under process conditions reduces the risk of batch failures, providing procurement teams with greater confidence in meeting delivery schedules for critical diabetes medications.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, with patent data supporting substrate concentrations up to 50g/L, which is indicative of a mature technology ready for commercial deployment. From an environmental perspective, the absence of heavy metals and the use of benign amine donors like isopropylamine significantly reduce the toxicological footprint of the manufacturing process. This alignment with green chemistry principles facilitates easier regulatory approval and compliance with increasingly strict environmental regulations, future-proofing the manufacturing asset against evolving global standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this transaminase technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D and technical procurement teams assessing the feasibility of technology transfer.
Q: What is the optical purity of sitagliptin produced by this mutant?
A: The engineered transaminase mutant AFATA21X achieves an optical purity (e.e. value) of greater than 99.95%, meeting stringent pharmaceutical standards.
Q: How does this enzymatic route compare to traditional chemical synthesis?
A: Unlike chemical routes requiring expensive chiral ruthenium or rhodium catalysts, this biocatalytic method operates under mild conditions without heavy metals, significantly simplifying downstream purification.
Q: What is the substrate loading capacity of this process?
A: The optimized reaction system supports substrate concentrations up to 50g/L, demonstrating high efficiency suitable for industrial-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis route described in patent CN112094856B and are fully equipped to leverage this technology for our global partners. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our state-of-the-art facilities are designed to handle complex biocatalytic processes with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to unlock the full commercial potential of this high-efficiency sitagliptin synthesis route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this biocatalytic approach can optimize your supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of high-purity sitagliptin for the global market.
