Revolutionizing Antibiotic Intermediate Production via One-Bacterium Multi-Enzyme Whole-Cell Catalysis
Introduction to Advanced Biocatalytic Synthesis of Chiral Antibiotic Intermediates
The pharmaceutical industry continuously seeks robust and sustainable pathways for producing critical chiral building blocks, particularly for broad-spectrum antibiotics like florfenicol and thiamphenicol. Patent CN110951799A introduces a groundbreaking methodology for the asymmetric synthesis of (2S,3R)-p-methylsulfonylphenylserine, utilizing a sophisticated "one-bacterium multi-enzyme" whole-cell catalytic system. This innovation represents a paradigm shift from traditional chemical synthesis and earlier enzymatic attempts, leveraging engineered Escherichia coli BL21(DE3) to co-express L-threonine transaldolase, alcohol dehydrogenase, and formate dehydrogenase simultaneously. By integrating these biological catalysts into a single cellular factory, the process achieves high-efficiency conversion under mild physiological conditions, effectively addressing long-standing challenges regarding by-product inhibition and cofactor regeneration that have historically plagued the industrial production of beta-amino alcohol antibiotics.
This technological advancement is not merely an incremental improvement but a structural optimization of the supply chain for key veterinary and human pharmaceutical intermediates. The ability to conduct the synthesis in a one-step reaction at normal temperature and pressure drastically reduces energy consumption and operational complexity. For R&D directors and process chemists, this patent offers a compelling alternative to metal-catalyzed routes, providing a pathway that inherently minimizes heavy metal contamination risks while maximizing stereochemical purity. The strategic implementation of this whole-cell system ensures that the production of (2S,3R)-p-methylsulfonylphenylserine becomes more predictable, scalable, and environmentally compliant, aligning perfectly with the global trend towards green chemistry and sustainable manufacturing practices in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of (2S,3R)-p-methylsulfonylphenylserine has relied heavily on chemical synthesis routes that are fraught with inefficiencies and environmental hazards. The traditional method typically involves an aldol condensation reaction between p-methylsulfonylbenzaldehyde and glycine in the presence of copper sulfate and ammonia water, followed by esterification and a subsequent chiral resolution step using D-tartaric acid. This multi-step chemical process is inherently limited by the theoretical maximum yield of the resolution step, which cannot exceed 50%, leading to significant wastage of raw materials and increased disposal costs for the unwanted enantiomer. Furthermore, the reliance on heavy metal catalysts like copper sulfate necessitates rigorous and costly purification steps to ensure the final API meets stringent residual metal specifications, adding layers of complexity to the downstream processing.
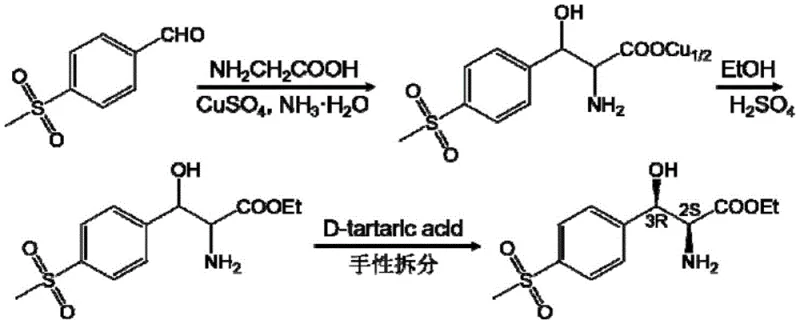
In addition to yield limitations, the conventional chemical route depicted in the prior art suffers from harsh reaction conditions and poor atom economy. The requirement for strong acids during esterification and the use of toxic solvents create significant safety and environmental burdens for manufacturing facilities. Earlier enzymatic approaches attempted to mitigate some of these issues but often fell short due to the need for separate enzyme fermentation and purification processes, which are capital-intensive and time-consuming. Moreover, single-enzyme systems frequently encountered product inhibition issues, where the accumulation of by-products like acetaldehyde would stall the reaction, resulting in incomplete conversion and lower overall productivity. These cumulative drawbacks highlight the urgent need for a more integrated and efficient biosynthetic strategy that can overcome thermodynamic barriers without compromising on purity or cost.
The Novel Approach
The novel approach detailed in patent CN110951799A fundamentally reengineers the biosynthetic pathway by employing a dual-plasmid system within a single host organism. By co-expressing L-threonine transaldolase (PsLTTA) alongside a cofactor regeneration system comprising alcohol dehydrogenase (ApADH) and formate dehydrogenase (CbFDH), the process creates a self-sustaining catalytic cycle within the whole cell. This "one-bacterium multi-enzyme" architecture allows for the in situ removal of acetaldehyde, a potent inhibitor of the primary transaldolase enzyme, by converting it into ethanol while simultaneously regenerating the essential reduced coenzyme I (NADH). This clever coupling of reactions drives the equilibrium strongly towards the desired product, achieving substrate conversion rates exceeding 99% and effectively bypassing the 50% yield ceiling associated with traditional resolution methods.
From a manufacturing perspective, this whole-cell catalytic method offers unparalleled simplicity and robustness. The elimination of enzyme purification steps means that the wet biomass harvested from fermentation can be directly used as the biocatalyst, significantly reducing the timeline from gene to product. The reaction proceeds efficiently at ambient temperatures (25-35°C) and neutral pH, removing the need for expensive heating or cooling infrastructure and specialized corrosion-resistant equipment. This streamlined process not only enhances the economic viability of producing (2S,3R)-p-methylsulfonylphenylserine but also ensures a consistent supply of high-purity material with an enantiomeric excess greater than 99.9%, making it an ideal solution for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
Mechanistic Insights into Multi-Enzyme Cascade Catalysis
The core of this technological breakthrough lies in the precise orchestration of a three-enzyme cascade within the cytoplasm of the engineered E. coli cells. The primary catalyst, L-threonine transaldolase (PsLTTA), facilitates the stereoselective aldol addition of L-threonine to p-methylsulfonylbenzaldehyde, generating the target chiral amino acid along with acetaldehyde as a by-product. In isolated systems, the accumulation of acetaldehyde would rapidly inhibit PsLTTA activity, stalling the reaction. However, the co-expressed alcohol dehydrogenase (ApADH) immediately scavenges this acetaldehyde, reducing it to ethanol. This secondary reaction consumes NADH, which is continuously replenished by the third enzyme, formate dehydrogenase (CbFDH), through the oxidation of sodium formate to carbon dioxide. This intricate network ensures that the concentration of the inhibitory by-product remains negligible while maintaining a high steady-state concentration of the necessary cofactor.
Furthermore, the spatial confinement of these enzymes within the whole cell creates a microenvironment that enhances catalytic efficiency and stability. The cellular membrane acts as a natural barrier, protecting the enzymes from external denaturants while allowing substrates and products to diffuse freely. This compartmentalization also simplifies the separation process, as the biocatalyst can be easily removed via centrifugation or filtration after the reaction is complete. The result is a highly selective transformation that favors the formation of the (2S,3R) diastereomer with a diastereomeric excess of 94.1%. Such high stereocontrol is critical for the downstream synthesis of florfenicol, where the presence of incorrect isomers can complicate purification and reduce the efficacy of the final antibiotic, underscoring the value of this mechanistic design for high-purity API manufacturing.
How to Synthesize (2S,3R)-p-Methylsulfonylphenylserine Efficiently
The implementation of this whole-cell biocatalytic process involves a straightforward sequence of genetic engineering and fermentation steps designed for industrial scalability. Initially, the requisite genes encoding PsLTTA, ApADH, and CbFDH are cloned into compatible expression vectors and transformed into the host strain. Following optimized fermentation to achieve high cell density and enzyme expression, the wet cells are harvested and resuspended in a buffered solution containing the substrates. The reaction is then initiated under mild agitation, allowing the intracellular enzymes to convert the starting materials into the desired chiral intermediate with minimal operational oversight. This simplified workflow eliminates the need for complex enzyme isolation protocols, making the technology accessible for rapid deployment in commercial settings.
- Construct double-plasmid engineering bacteria by transforming E. coli BL21(DE3) with pET28a-PsLTTA and pETDuet-ApADH/CbFDH plasmids.
- Culture the engineered bacteria in LB medium with antibiotics, induce protein expression with IPTG at OD600 0.5, and harvest cells via centrifugation.
- Perform the asymmetric synthesis by reacting p-methylsulfonylbenzaldehyde and L-threonine with the wet whole cells in Tris-HCl buffer at 30°C for 4 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this whole-cell biocatalytic technology translates into tangible strategic benefits that extend beyond simple unit cost savings. The consolidation of multiple enzymatic functions into a single fermentation and reaction step drastically simplifies the manufacturing workflow, reducing the number of unit operations and the associated labor and utility costs. By eliminating the need for expensive chromatographic purification of individual enzymes and avoiding the use of costly heavy metal catalysts, the overall cost of goods sold (COGS) is significantly reduced. This economic efficiency allows suppliers to offer more competitive pricing structures while maintaining healthy margins, providing a distinct advantage in the highly price-sensitive market for generic antibiotic intermediates.
- Cost Reduction in Manufacturing: The transition from chemical synthesis or single-enzyme methods to this multi-enzyme whole-cell system removes several cost-intensive bottlenecks. The avoidance of chiral resolution steps, which traditionally discard half of the produced material, effectively doubles the theoretical yield from the same amount of raw materials, leading to substantial raw material savings. Additionally, the omission of enzyme purification processes reduces both the capital expenditure on separation equipment and the operational expenditure on buffers and resins. The mild reaction conditions further contribute to cost efficiency by lowering energy consumption for heating and cooling, creating a leaner and more profitable production model for high-volume pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Reliability in the supply of critical intermediates is paramount for pharmaceutical manufacturers, and this biocatalytic route offers superior consistency compared to chemical methods prone to batch-to-batch variability. The use of genetically defined strains ensures that the catalytic activity remains stable over time, reducing the risk of failed batches due to catalyst degradation or impurity buildup. Furthermore, the reliance on renewable biological feedstocks and the reduction of hazardous chemical usage mitigate regulatory risks associated with environmental compliance and worker safety. This robustness ensures a continuous and dependable flow of materials, safeguarding downstream production schedules against disruptions caused by raw material shortages or regulatory inspections.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is often more straightforward than scaling complex chemical syntheses involving hazardous reagents. The aqueous nature of the reaction medium and the absence of volatile organic solvents simplify waste treatment and reduce the environmental footprint of the manufacturing facility. This alignment with green chemistry principles not only lowers waste disposal costs but also enhances the corporate sustainability profile of the supplier. As global regulations on industrial emissions tighten, adopting such environmentally benign technologies future-proofs the supply chain, ensuring long-term operational continuity and compliance with increasingly stringent international environmental standards without the need for costly retrofits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel biocatalytic synthesis method. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering clarity on how this technology compares to existing industry standards. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing supply chains or R&D pipelines.
Q: How does the 'one-bacterium multi-enzyme' system improve yield compared to single-enzyme catalysis?
A: The system co-expresses L-threonine transaldolase (PsLTTA) with alcohol dehydrogenase (ApADH) and formate dehydrogenase (CbFDH). This coupled system removes the by-product acetaldehyde, which otherwise inhibits PsLTTA, thereby driving the reaction equilibrium forward and significantly increasing yield without needing external enzyme purification.
Q: What are the stereochemical purity specifications achieved by this biocatalytic method?
A: The process achieves exceptional stereocontrol, delivering the target (2S,3R) isomer with an enantiomeric excess (ee) greater than 99.9% and a diastereomeric excess (de) of 94.1%, eliminating the need for complex chiral resolution steps typical in chemical synthesis.
Q: Why is whole-cell catalysis more cost-effective than using purified enzymes for this synthesis?
A: Whole-cell catalysis bypasses the expensive and time-consuming steps of enzyme fermentation, separation, and purification. The cells act as natural immobilized catalysts, reducing downstream processing costs and simplifying the overall manufacturing workflow for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S,3R)-p-Methylsulfonylphenylserine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in reshaping the landscape of pharmaceutical intermediate production. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN110951799A can be seamlessly translated into robust industrial operations. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of (2S,3R)-p-methylsulfonylphenylserine meets the exacting standards required for the synthesis of life-saving antibiotics. We are committed to delivering high-quality intermediates that support the global demand for effective veterinary and human therapeutics.
We invite forward-thinking pharmaceutical companies and procurement teams to collaborate with us to leverage this cutting-edge technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-purity supply of this critical chiral building block, ensuring your production lines remain efficient and competitive in the global marketplace.
