Advanced Montelukast Intermediate Manufacturing: Overcoming Impurity Challenges with Novel Thiolactone Chemistry
Advanced Montelukast Intermediate Manufacturing: Overcoming Impurity Challenges with Novel Thiolactone Chemistry
The pharmaceutical industry continuously seeks robust synthetic pathways for high-volume active pharmaceutical ingredients (APIs) like Montelukast, a cornerstone treatment for asthma and allergic rhinitis. Patent CN101356158A introduces a transformative approach to Montelukast synthesis, addressing long-standing challenges regarding intermediate stability, impurity profiles, and scalability. This patent discloses the use of novel solid-state thiolactone intermediates, specifically compound (11), and optimized reaction conditions involving activated cerium salts to achieve exceptional chemical and optical purity. For R&D directors and procurement specialists, understanding these mechanistic improvements is crucial for securing a reliable supply of high-purity pharmaceutical intermediates. The technology shifts the paradigm from handling unstable oils to isolating crystalline solids, fundamentally enhancing process control and reducing the risk of batch failures in commercial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Montelukast, such as those described in earlier patents like US5565473, often rely on liquid intermediates that are difficult to purify and prone to degradation. A significant bottleneck in these conventional processes is the formation of persistent ketone impurities, specifically compound (12), during the reductive methylation steps involving methylmagnesium halides. These ketone by-products arise from the enolization of intermediate species and are notoriously difficult to remove in downstream processing, often carrying through to the final API and compromising quality specifications. Furthermore, the reliance on unstable reagents like the bromo-ester (5a) introduces risks of rearrangement reactions, leading to cyclobutane derivatives that contaminate the product stream. The inability to isolate key intermediates in a solid, stable form forces manufacturers to carry impurities through multiple steps, increasing waste and lowering overall yield.
The Novel Approach
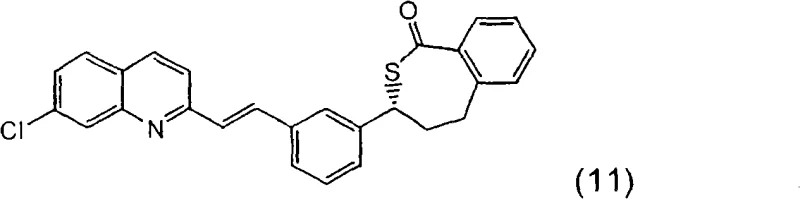
The methodology outlined in CN101356158A overcomes these hurdles by introducing a stable thiolactone intermediate, compound (11), which can be isolated and purified as a solid or as a stable acid addition salt like the hydrochloride (11a). This solid-state isolation acts as a powerful purification checkpoint, allowing manufacturers to remove by-products before proceeding to the final coupling stages. Additionally, the patent proposes the use of a novel sulfonate reagent, compound (5b), which features a p-methoxybenzenesulfonyloxy leaving group. Unlike the traditional bromo-ester, this new reagent exhibits enhanced chemical stability against rearrangement while maintaining sufficient reactivity for nucleophilic substitution. By combining stable solid intermediates with optimized reagents, this approach significantly streamlines the production of high-purity pharmaceutical intermediates, offering a more predictable and controllable manufacturing process.
Mechanistic Insights into Cerium-Activated Grignard Reactions
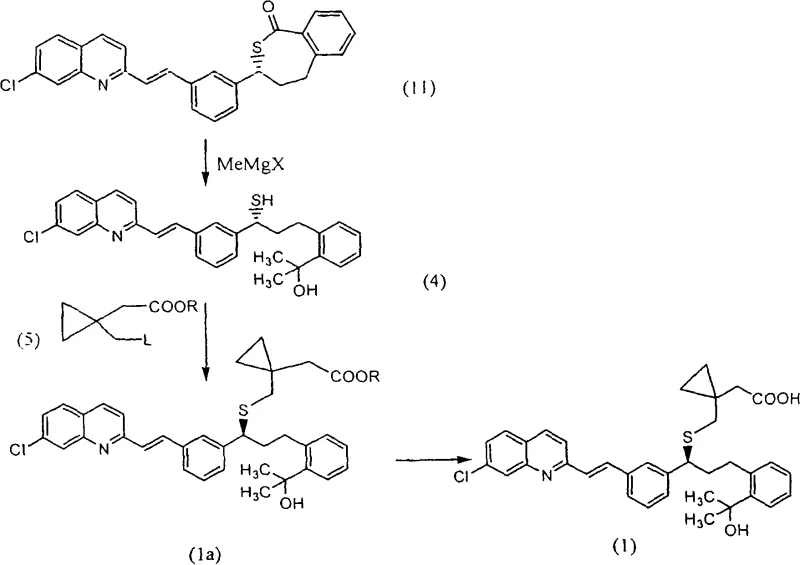
A critical innovation in this patent is the strategic use of cerium(III) compounds, particularly activated cerium(III) chloride, to modulate the reactivity of Grignard reagents. During the conversion of acetylthioester (20) to thiolactone (11), or directly to thiol intermediate (4), the reaction with methylmagnesium halides typically generates a ketone impurity (12) due to incomplete reduction or enolization. The patent reveals that adding activated cerium(III) salts effectively suppresses this enolization pathway. The cerium species likely coordinates with the carbonyl oxygen, altering the electronic environment and preventing the formation of the stable enolate that leads to impurity (12). This activation allows for the use of less expensive methylmagnesium chloride or bromide instead of the more costly iodide, without sacrificing conversion efficiency. The result is a reaction mixture where the desired tertiary alcohol is formed with minimal ketone contamination, drastically reducing the burden on downstream purification units.
Furthermore, the stability of the new coupling reagent (5b) is rooted in the electronic properties of the p-methoxybenzenesulfonyloxy group. The electron-donating effect of the methoxy group provides a balance of stability and reactivity that prevents the spontaneous rearrangement observed in halogenated analogs. This mechanistic stability ensures that the reagent remains intact during storage and reaction setup, minimizing the generation of structurally related impurities that are hard to separate from the final Montelukast product. For process chemists, this means a wider operating window and reduced sensitivity to minor variations in temperature or addition rates, which is essential for scaling up complex organic syntheses to multi-ton production levels.
How to Synthesize Montelukast Efficiently
The synthesis of Montelukast via this novel route involves a sequence of well-defined steps starting from the methyl ester compound (18). The process begins with the conversion of the hydroxyl group into a reactive mesylate, followed by substitution with thioacetic acid to yield the acetylthioester (20). This intermediate is then subjected to a cerium-activated Grignard reaction to form the thiolactone (11), which is isolated as a high-purity solid. Subsequent ring-opening with excess Grignard reagent generates the thiol intermediate (4), which is immediately coupled with the stable sulfonate reagent (5b) to form the final carbon-sulfur bond.
- Convert methyl ester compound (18) to acetylthioester (20) via mesylation and thioacetate substitution, isolating as a stable solid acid addition salt.
- React acetylthioester (20) with methylmagnesium halide in the presence of activated cerium(III) chloride to form thiolactone (11) while suppressing ketone impurities.
- Open the thiolactone ring with excess Grignard reagent to generate thiol intermediate (4), then couple with novel sulfonate reagent (5b) to finalize Montelukast.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift towards the chemistry described in CN101356158A offers tangible benefits in terms of cost reduction in pharmaceutical intermediates manufacturing and supply security. The ability to isolate intermediates like compound (11) and compound (20) as stable solids eliminates the need for complex chromatographic purifications often required for oily residues. This simplification of the workflow translates directly into reduced processing time and lower solvent consumption, driving down the overall cost of goods sold. Moreover, the enhanced stability of the intermediates allows for longer storage periods without degradation, providing greater flexibility in inventory management and reducing the risk of material loss due to shelf-life expiration.
- Cost Reduction in Manufacturing: The elimination of toxic hydrazine reagents and the ability to use cheaper methylmagnesium chloride instead of iodide significantly lowers raw material costs. Additionally, the suppression of ketone impurities reduces the load on purification steps, leading to substantial cost savings in solvent usage and waste disposal. The process avoids the expensive and hazardous handling of unstable oils, replacing them with robust solid forms that are easier to handle and transport.
- Enhanced Supply Chain Reliability: The robustness of the solid intermediates ensures consistent quality across batches, reducing the likelihood of out-of-specification results that can disrupt supply. The use of stable reagents like (5b) minimizes the risk of reaction failures due to reagent degradation, ensuring a more predictable production schedule. This reliability is critical for maintaining continuous supply lines for high-demand asthma medications, preventing stockouts and ensuring market availability.
- Scalability and Environmental Compliance: The process is designed for commercial scale-up of complex pharmaceutical intermediates, with reaction conditions that are easily controlled in large reactors. The reduction in hazardous reagents and the improvement in atom economy contribute to a greener manufacturing profile, aligning with increasingly stringent environmental regulations. The ability to recycle solvents and minimize waste streams further enhances the sustainability of the production process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this novel Montelukast synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the practical application of the technology.
Q: How does the novel thiolactone route improve Montelukast purity compared to traditional methods?
A: The thiolactone route allows for the isolation of stable solid intermediates like compound (11) and its salts, which can be purified to >99% purity before the final coupling step. This contrasts with traditional oil-based intermediates that trap impurities.
Q: What is the role of Cerium(III) chloride in this synthesis?
A: Activated Cerium(III) chloride is critical for suppressing the formation of ketone impurity (12) during the Grignard reaction. It inhibits enolization of the intermediate ketone, ensuring complete conversion to the desired tertiary alcohol.
Q: Why is reagent (5b) preferred over the traditional bromo-ester (5a)?
A: Reagent (5b), a p-methoxybenzenesulfonyloxy compound, offers superior chemical stability compared to the bromo-ester (5a), which is prone to rearrangement into cyclobutane derivatives. This stability reduces side reactions and simplifies purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Montelukast Intermediate Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of purity and consistency in the production of life-saving medications. Our technical team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN101356158A can be successfully translated from the lab to the plant. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Montelukast intermediate meets the highest global standards. Our commitment to quality ensures that our partners receive materials that facilitate smooth downstream processing and final API certification.
We invite you to discuss how our advanced manufacturing capabilities can support your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your operational efficiency and reduce total landed costs.
