Advanced Catalytic Synthesis of Florfenicol Intermediates for Commercial Veterinary Drug Production
Advanced Catalytic Synthesis of Florfenicol Intermediates for Commercial Veterinary Drug Production
The pharmaceutical and veterinary industries are constantly seeking more efficient and sustainable pathways for the production of critical active ingredients, and the synthesis of florfenicol intermediates stands as a prime example of this technological evolution. Patent CN115160194A introduces a groundbreaking method for preparing florfenicol intermediates, specifically D-p-methylsulfonyl phenyl serine ester, utilizing a novel solid chiral catalyst system that fundamentally alters the economic and environmental landscape of this synthesis. This innovation addresses long-standing inefficiencies in chiral drug manufacturing by replacing traditional resolution methods with a direct asymmetric catalytic approach, thereby offering a robust solution for large-scale production. The significance of this patent lies not only in its chemical elegance but also in its practical applicability for industrial partners seeking to optimize their supply chains for veterinary antibiotics. By leveraging this advanced technology, manufacturers can achieve superior chiral purity while simultaneously reducing the ecological footprint associated with heavy metal usage and waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional industrial processes for producing florfenicol intermediates have historically relied on multi-step linear syntheses that involve the resolution of racemic mixtures, a method inherently plagued by low atom economy and significant material wastage. In the conventional route, p-toluenesulfonyl chloride undergoes a series of transformations including reduction, methylation, and bromination before reacting with glycine and copper sulfate to form a copper salt intermediate. This pathway necessitates a chiral resolution step using tartaric acid, which theoretically discards fifty percent of the produced isomer, leading to a maximum yield of only about forty percent for the desired configuration. Furthermore, the reliance on copper sulfate generates substantial amounts of heavy metal-containing wastewater, imposing severe environmental compliance costs and complicating waste management protocols for manufacturing facilities. The complexity of this route, involving numerous unit operations and purification steps, also extends production lead times and increases the risk of quality deviations during scale-up.
The Novel Approach
In stark contrast to the cumbersome traditional methods, the novel approach disclosed in the patent utilizes a highly efficient solid chiral catalyst to directly construct the chiral center in the initial step, bypassing the need for wasteful resolution processes. This streamlined strategy involves the reaction of p-methylthiobenzaldehyde with isocyanoacetic ester under the catalysis of a purified solid silver complex, which directly yields the chiral intermediate with significantly enhanced stereocontrol. The process eliminates the use of copper salts entirely, thereby removing the burden of heavy metal wastewater treatment and aligning the production process with modern green chemistry principles. By consolidating the synthesis into fewer steps with higher convergence, this method not only improves the overall throughput but also simplifies the operational requirements for plant personnel. The ability to recover and recycle the solid catalyst further amplifies the economic advantages, making this route exceptionally attractive for cost-sensitive commercial manufacturing environments.
Mechanistic Insights into Solid Chiral Catalyst Asymmetric Synthesis
The core of this technological breakthrough resides in the unique structure and function of the solid chiral catalyst, which is formed by the complexation of a specific quinoline-based ligand with silver oxide. This catalyst operates through a precise asymmetric induction mechanism, guiding the addition of the isocyanoacetate to the aldehyde substrate to favor the formation of the desired chiral configuration with exceptional selectivity. The patent details the preparation of the catalyst itself, involving the reaction of compound L with silver oxide in organic solvents followed by low-temperature crystallization to ensure high purity levels between ninety and ninety-nine percent. This high purity of the catalyst precursor is critical, as it directly correlates to the enantiomeric excess of the final product, ensuring that the resulting intermediate meets the stringent quality standards required for veterinary drug applications. The mechanistic advantage lies in the rigid chiral environment provided by the ligand, which effectively shields one face of the reacting molecule, thus minimizing the formation of unwanted stereoisomers.
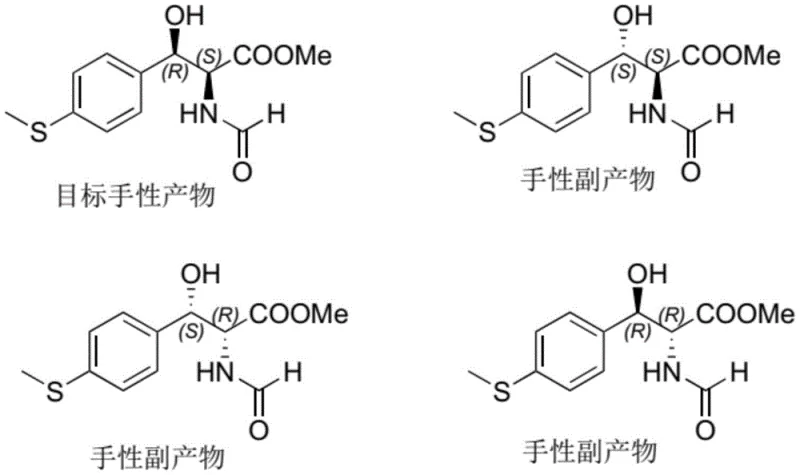
Understanding the stereochemical outcome is vital for R&D directors focused on impurity profiles, as the process inherently suppresses the formation of diastereomers that are difficult to separate in downstream processing. The patent illustrates the four possible stereoisomers that could theoretically form during the aldol-type reaction, highlighting how the specific catalyst geometry directs the reaction towards the target (R,S) configuration while suppressing the (S,S), (R,R), and (S,R) byproducts. This high level of stereocontrol means that the crude product requires less intensive purification, reducing solvent consumption and energy usage during isolation. Moreover, the stability of the solid catalyst allows for consistent performance across multiple batches, providing the reproducibility necessary for validated commercial production. The elimination of racemic waste not only improves yield but also simplifies the regulatory documentation regarding impurity characterization and control strategies.
How to Synthesize Florfenicol Intermediate Efficiently
The implementation of this synthesis route involves a carefully controlled three-step sequence that begins with the asymmetric catalytic reaction, followed by oxidation and finally deprotection to yield the target molecule. The first step requires dissolving p-methylthiobenzaldehyde and isocyanoacetic ester in a suitable organic solvent such as ethyl acetate or tetrahydrofuran, followed by the addition of the solid catalyst at controlled temperatures to initiate the chiral bond formation. Subsequent steps involve the oxidation of the methylthio group to a sulfone using oxidants like hydrogen peroxide or m-chloroperoxybenzoic acid, and finally, the removal of the formyl protecting group under acidic conditions to reveal the free amine. Detailed standardized synthetic steps see the guide below.
- React p-methylthiobenzaldehyde with isocyanoacetic ester using a purified solid chiral silver catalyst to form the chiral intermediate.
- Oxidize the methylthio group to a methylsulfonyl group using an oxidant like hydrogen peroxide or m-chloroperoxybenzoic acid.
- Remove the formyl protecting group under acidic conditions to obtain the final florfenicol intermediate TM.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology translates into tangible strategic benefits that extend beyond simple chemical yield improvements to encompass broader operational efficiencies. The elimination of the chiral resolution step effectively doubles the theoretical yield of the process relative to the starting materials, which drastically reduces the volume of raw materials required to produce a fixed quantity of the final intermediate. This reduction in material intensity directly correlates to lower procurement costs and decreased logistical burdens associated with storing and handling large volumes of chemical inputs. Additionally, the removal of copper sulfate from the process workflow mitigates the risks and costs associated with hazardous waste disposal, allowing facilities to operate with greater environmental compliance and reduced liability. The recyclability of the solid catalyst further contributes to cost stability, as the expensive chiral ligand system does not need to be purchased in stoichiometric quantities for every batch.
- Cost Reduction in Manufacturing: The transition to a direct asymmetric synthesis model eliminates the inherent fifty percent material loss associated with racemic resolution, leading to substantial cost savings in raw material consumption. By utilizing a catalyst that can be recovered and reused, the operational expenditure related to chiral auxiliaries is significantly minimized over the lifecycle of the product. The simplified process flow also reduces utility consumption, such as steam and cooling water, due to fewer unit operations and shorter reaction times. These factors combine to create a more economically resilient manufacturing model that is less susceptible to fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The streamlined nature of this synthesis route reduces the overall production lead time, enabling faster response to market demand fluctuations for veterinary antibiotics. With fewer intermediate isolation steps, the risk of production bottlenecks is minimized, ensuring a more consistent and reliable flow of goods to downstream customers. The use of common organic solvents and readily available oxidants further secures the supply chain against disruptions caused by specialty chemical shortages. This reliability is crucial for maintaining continuous production schedules in the highly regulated veterinary pharmaceutical sector.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to industrial-scale reactors without significant re-optimization. The absence of heavy metal contaminants simplifies the purification process and ensures that the final product meets stringent international residue limits without extensive additional cleaning steps. This environmental compatibility facilitates easier regulatory approvals in key markets, reducing the time to market for new formulations. Furthermore, the reduced waste profile supports corporate sustainability goals and enhances the brand reputation of manufacturers adopting this green technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method for florfenicol intermediates. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production frameworks.
Q: How does the solid catalyst improve yield compared to traditional resolution?
A: The solid chiral catalyst enables direct asymmetric synthesis, achieving yields of 75-90% compared to approximately 40% with conventional chiral resolution methods, significantly reducing raw material waste.
Q: What are the environmental benefits of this new process?
A: This method eliminates the use of anhydrous copper sulfate, thereby avoiding the generation of heavy metal wastewater and reducing the environmental burden associated with wastewater treatment.
Q: Can the catalyst be recovered and reused?
A: Yes, the solid chiral catalyst can be separated from the reaction mixture and recycled for subsequent batches, which further lowers production costs and enhances process sustainability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Florfenicol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global veterinary drug market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN115160194A can be successfully translated into robust industrial operations. We are committed to delivering high-purity florfenicol intermediates that meet stringent purity specifications, supported by our rigorous QC labs which employ state-of-the-art analytical techniques to verify chiral integrity and chemical identity. Our infrastructure is designed to handle complex chiral syntheses with the utmost precision, guaranteeing supply continuity for our partners.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your product portfolio. Let us partner together to drive efficiency and quality in the production of essential veterinary medicines.
