Revolutionizing Chalcone Production: A Green Ionic Liquid Catalysis Strategy for Commercial Scale-Up
Revolutionizing Chalcone Production: A Green Ionic Liquid Catalysis Strategy for Commercial Scale-Up
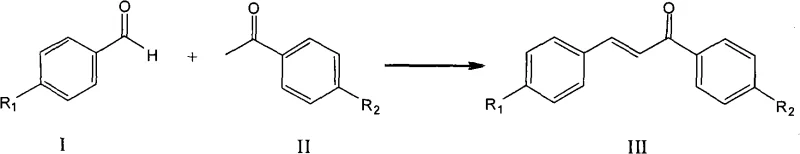
The global demand for high-purity chalcones and their derivatives continues to surge, driven by their critical applications as key intermediates in the synthesis of flavonoids, pharmaceuticals, and agrochemicals. However, traditional manufacturing routes have long been plagued by environmental inefficiencies and complex downstream processing. Patent CN101289378A introduces a transformative methodology that addresses these industrial pain points by utilizing sulfonic acid-functionalized ionic liquids as dual-purpose catalysts and solvents. This innovation represents a paradigm shift from conventional base-catalyzed aldol condensations, offering a robust pathway for the reliable chalcone supplier seeking to modernize their production capabilities. By eliminating the need for volatile organic solvents and enabling facile catalyst recovery, this technology not only aligns with stringent environmental regulations but also significantly streamlines the operational workflow for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of chalcones has relied heavily on the Claisen-Schmidt condensation reaction, typically employing strong bases such as sodium hydroxide or potassium hydroxide in alcoholic solvents like ethanol or methanol. While chemically effective, these legacy processes generate substantial volumes of alkaline wastewater and require energy-intensive distillation steps to recover solvents. Furthermore, the use of homogeneous base catalysts often leads to side reactions, including polymerization or over-condensation, which complicates the purification of the final API intermediate. The necessity for neutralization steps post-reaction introduces additional salt waste, creating a significant burden on waste treatment facilities and inflating the overall cost of goods sold. For procurement managers, these inefficiencies translate into higher raw material consumption and unpredictable supply chain disruptions due to environmental compliance issues.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes task-specific ionic liquids that function simultaneously as the reaction medium and the acidic catalyst. This approach fundamentally alters the reaction landscape by operating under solvent-free conditions, thereby eliminating the hazards associated with volatile organic compounds (VOCs). The unique physicochemical properties of these ionic liquids allow the reaction to proceed efficiently at temperatures ranging from 80°C to 200°C, with reaction times as short as 0.5 hours. Crucially, the system exhibits thermomorphic behavior where the product spontaneously separates from the catalyst phase upon completion, simplifying isolation to a mere decantation step. This technological leap provides a clear route for cost reduction in pharmaceutical intermediates manufacturing by drastically reducing solvent purchase costs and waste disposal fees.
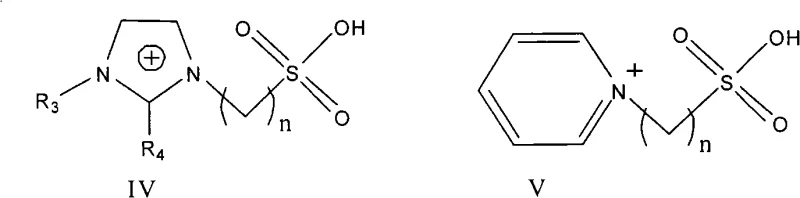
Mechanistic Insights into Sulfonic Acid-Functionalized Ionic Liquid Catalysis
The core of this innovation lies in the molecular design of the catalyst, specifically the incorporation of a sulfonic acid group (-SO3H) onto the cationic structure of imidazolium or pyridinium salts. As illustrated in the structural formulas, the cation serves as a stable scaffold while the tethered sulfonic acid moiety provides the necessary Brønsted acidity to activate the carbonyl group of the acetophenone. This intramolecular acid functionality ensures that the catalytic active sites are homogeneously distributed throughout the reaction medium, enhancing mass transfer rates compared to heterogeneous solid acid catalysts. The anion choice, ranging from bisulfate to tetrafluoroborate, further tunes the polarity and solubility parameters of the liquid, optimizing the interaction between the hydrophobic organic reactants and the ionic catalyst phase. This precise engineering allows for exceptional control over the reaction kinetics, minimizing the formation of by-products and ensuring high selectivity.
From an impurity control perspective, the mechanism offers distinct advantages over traditional base catalysis. In basic media, chalcones are susceptible to Michael addition reactions or degradation, leading to complex impurity profiles that are difficult to remove. The acidic environment provided by the ionic liquid suppresses these base-catalyzed side reactions, resulting in a cleaner crude product with selectivity often exceeding 99%. Furthermore, the non-volatile nature of the ionic liquid prevents the entrainment of catalyst residues in the vapor phase during heating, a common issue with low-boiling acid catalysts. This inherent stability ensures that the final high-purity chalcone meets rigorous quality specifications required for downstream pharmaceutical applications, reducing the need for extensive chromatographic purification and lowering the overall production cost.
How to Synthesize Chalcone Efficiently
The implementation of this ionic liquid-mediated synthesis is straightforward and adaptable to existing reactor infrastructure, requiring minimal capital expenditure for retrofitting. The process begins with the precise charging of benzaldehyde and acetophenone substrates along with the pre-synthesized ionic liquid catalyst into a standard stirred tank reactor. To prevent oxidative degradation of the aldehyde substrate, the reaction vessel is purged with inert gas such as argon before heating. The mixture is then agitated and heated to the optimal temperature window, typically around 140°C, where the reaction proceeds to completion within a few hours. Detailed standardized synthetic steps see the guide below.
- Charge a reactor with benzaldehyde, acetophenone, and the sulfonic acid-functionalized ionic liquid catalyst (mass ratio 1: 20 to 10:1 relative to reactants).
- Heat the mixture to a reaction temperature between 80°C and 200°C (optimally 140°C) under argon protection and stir for 0.5 to 12 hours.
- Allow the reaction mixture to stand for phase separation; decant the upper organic layer containing the crude chalcone product for recrystallization.
- Recover the lower ionic liquid layer by vacuum drying at 110°C for 1-6 hours for immediate reuse in subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement strategists, the adoption of this ionic liquid technology offers compelling economic and operational benefits that extend beyond simple yield improvements. The elimination of bulk organic solvents removes a major variable cost component and mitigates the risks associated with solvent price volatility and supply shortages. Additionally, the simplified workup procedure reduces the cycle time per batch, effectively increasing the throughput capacity of existing manufacturing assets without the need for new construction. The ability to recycle the catalyst multiple times after a simple vacuum drying step creates a closed-loop system that dramatically lowers the consumption of auxiliary chemicals. These factors combined contribute to a more resilient and cost-effective supply chain capable of meeting fluctuating market demands.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dual role of the ionic liquid as both catalyst and solvent, which eliminates the need for purchasing and recovering large volumes of ethanol or methanol. By removing the solvent recovery distillation step and the associated energy consumption, the utility costs per kilogram of product are significantly lowered. Furthermore, the high selectivity of the reaction reduces the loss of valuable starting materials to by-products, maximizing the atom economy of the process. The absence of aqueous waste streams also translates to substantial savings in wastewater treatment costs, which are increasingly becoming a major financial burden for chemical manufacturers.
- Enhanced Supply Chain Reliability: Relying on traditional solvent-heavy processes exposes manufacturers to supply chain vulnerabilities related to petrochemical feedstock availability. This novel method reduces dependency on such commodities by utilizing stable, reusable ionic materials that have a long operational lifespan. The robustness of the catalyst under thermal stress ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. Moreover, the simplified logistics of handling non-volatile, non-flammable ionic liquids improve workplace safety and reduce insurance premiums, further stabilizing the operational budget.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is facilitated by the benign nature of the reaction medium, which does not require specialized pressure vessels or explosion-proof equipment typically needed for volatile solvents. The negligible vapor pressure of the ionic liquid ensures that emissions are virtually non-existent, helping facilities maintain compliance with strict environmental protection agency regulations regarding VOC emissions. This 'green' credential is increasingly valuable for securing contracts with multinational corporations that have aggressive sustainability goals, positioning the manufacturer as a preferred partner for eco-friendly sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this ionic liquid catalysis system. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent literature, aimed at clarifying the operational parameters for potential adopters. Understanding these nuances is essential for R&D teams planning to integrate this technology into their existing process development pipelines.
Q: How does the ionic liquid catalyst facilitate product separation compared to traditional solvents?
A: Unlike traditional alcohol-water systems that require complex extraction, the ionic liquid used in this process forms a distinct biphasic system with the chalcone product upon cooling. The product naturally separates as an upper organic layer, allowing for simple decantation without the need for volatile organic extraction solvents.
Q: What is the thermal stability and reusability profile of the catalyst?
A: The sulfonic acid-functionalized imidazolium or pyridinium ionic liquids exhibit high thermal stability up to 200°C. Experimental data indicates that after vacuum drying at 110°C, the catalyst retains its activity and can be recycled multiple times without significant loss in yield or selectivity.
Q: Can this method be adapted for diverse chalcone derivatives?
A: Yes, the method is highly versatile. By varying the substituents R1 and R2 on the benzaldehyde and acetophenone starting materials (such as alkyl or alkoxy groups), a wide range of chalcone derivatives can be synthesized with consistently high selectivity (>98%) and yields exceeding 94%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chalcone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry technologies like the ionic liquid-catalyzed synthesis of chalcones. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of chalcone intermediate meets the highest international standards. We are committed to leveraging advanced catalytic strategies to deliver superior value to our global clientele.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your specific supply chain requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic benefits of switching to this solvent-free methodology. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and competitive advantage in the global market.
