Revolutionizing Sultam Production: Green Electrochemical Synthesis for Commercial Scale-up
Revolutionizing Sultam Production: Green Electrochemical Synthesis for Commercial Scale-up
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards sustainable and economically viable processes, particularly in the synthesis of complex heterocyclic scaffolds. Patent CN114525527A introduces a groundbreaking electrochemical synthesis method for producing sultam derivatives, a class of compounds pivotal in the development of pharmaceuticals for metabolic disorders, hypertension, and non-steroidal anti-inflammatory treatments. This technology leverages constant current electrolysis to drive the cyclization of 2-isoallylbenzenesulfonamide derivatives in the presence of simple iodide salts. By replacing traditional stoichiometric oxidants and precious metal catalysts with electricity, this method offers a compelling value proposition for R&D directors seeking cleaner impurity profiles and procurement managers aiming for significant cost reduction in pharmaceutical intermediate manufacturing. The ability to operate under ambient conditions without inert gas protection further simplifies the operational complexity, making it an ideal candidate for reliable sultam derivative supplier networks looking to scale production efficiently.
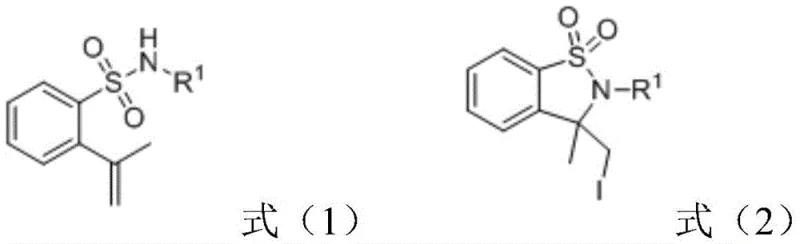
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the sultam core has relied heavily on transition metal catalysis, often involving palladium or iron complexes that introduce significant supply chain vulnerabilities and cost burdens. Traditional protocols frequently necessitate the use of stoichiometric amounts of chemical oxidants or reductants, which generate substantial waste streams and complicate downstream purification processes. For instance, previous methodologies reported by research groups such as Manabe and Liu required specific ligands and elevated temperatures, creating energy-intensive workflows that are difficult to justify in a modern green chemistry context. The reliance on heavy metals also poses a critical challenge for pharmaceutical applications, where strict regulatory limits on residual metal impurities demand expensive and time-consuming scavenging steps. Furthermore, the use of harsh reaction conditions can lead to poor functional group tolerance, limiting the substrate scope and reducing the overall yield of high-purity OLED material or API precursors. These factors collectively hinder the commercial scale-up of complex polymer additives and pharmaceutical intermediates, driving the industry to seek alternative synthetic routes.
The Novel Approach
The electrochemical strategy outlined in the patent data represents a decisive break from these conventional constraints by utilizing electrons as the primary reagent to drive the oxidative cyclization. This novel approach employs inexpensive iodide salts, such as sodium iodide or tetrabutylammonium iodide, which serve as redox mediators to facilitate the formation of the sulfur-nitrogen bond without the need for external chemical oxidants. The reaction proceeds smoothly in a mixed solvent system of acetonitrile and water at room temperature, eliminating the energy costs associated with heating and the safety risks of high-pressure reactors. By operating under air atmosphere, the process removes the logistical burden of maintaining inert gas lines, thereby enhancing supply chain reliability and reducing lead time for high-purity intermediates. The simplicity of the setup, utilizing basic carbon or platinum electrodes, allows for easy adaptation to flow chemistry systems, offering a scalable pathway for the commercial production of diverse sultam derivatives with varying R1 substituents including phenyl, benzyl, and alkyl groups.
Mechanistic Insights into Electrochemical Oxidative Cyclization
At the heart of this transformation lies an elegant anodic oxidation mechanism where iodide ions are oxidized at the anode surface to generate reactive iodine species, likely molecular iodine or iodine radicals, in situ. These electro-generated species then interact with the electron-rich alkene moiety of the 2-isoallylbenzenesulfonamide substrate to form an iodonium ion intermediate. This activation step is crucial as it renders the alkene susceptible to nucleophilic attack by the adjacent sulfonamide nitrogen atom, triggering an intramolecular cyclization event. The subsequent deprotonation and rearomatization steps lead to the formation of the stable sultam ring system with the incorporation of an iodomethyl group at the 3-position. This mechanism avoids the formation of high-energy radical intermediates often seen in thermal radical reactions, resulting in a cleaner reaction profile with fewer side products. The use of a mixed solvent system enhances the conductivity of the solution, ensuring efficient electron transfer while maintaining the solubility of both the organic substrate and the inorganic iodide salt.
From an impurity control perspective, the absence of transition metals fundamentally alters the impurity spectrum, removing the risk of metal-catalyzed homocoupling or decomposition pathways that often plague traditional methods. The mild reaction conditions prevent thermal degradation of sensitive functional groups on the aromatic ring, such as halogens or alkoxy groups, ensuring high fidelity in the final product structure. The electrochemical generation of the oxidant occurs at a controlled rate determined by the applied current, preventing the local accumulation of highly reactive species that could lead to over-oxidation or polymerization. This precise control contributes to the high yields observed across a broad range of substrates, as evidenced by the successful synthesis of derivatives with electron-donating and electron-withdrawing groups. For R&D teams, understanding this mechanism provides confidence in the robustness of the process, facilitating the design of experiments for new analogues without the fear of catalyst poisoning or ligand incompatibility.

How to Synthesize 2-Phenyl-3-iodomethyl-2,3-benzisothiazole 1,1-dioxide Efficiently
To implement this cutting-edge technology in a laboratory or pilot plant setting, operators must adhere to specific parameters regarding current density, solvent composition, and electrode material to maximize efficiency. The process begins with the preparation of a homogeneous solution containing the sulfonamide precursor and the iodide source in the optimized acetonitrile-water mixture. Maintaining a constant current within the range of 5mA to 15mA is critical to balance the rate of iodine generation with the consumption by the substrate, preventing side reactions. The reaction is typically monitored by TLC or HPLC, and upon completion, the workup involves standard extraction and purification techniques that are familiar to any synthetic chemist. Detailed standardized synthesis steps see the guide below for precise operational instructions.
- Prepare the reaction mixture by dissolving 2-isoallylbenzenesulfonamide derivatives and sodium iodide in a mixed solvent of acetonitrile and water.
- Set up an undivided cell with carbon electrodes and apply a constant current of 5mA to 15mA at room temperature under air atmosphere.
- After reaction completion, extract the mixture with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical methodology translates directly into tangible operational efficiencies and risk mitigation strategies. The elimination of precious metal catalysts removes a major cost driver and supply bottleneck, as the price volatility of palladium and other rare earth metals no longer impacts the production budget. Additionally, the simplified workup procedure, which does not require specialized metal scavenging resins or complex filtration steps, drastically reduces the consumption of auxiliary materials and labor hours. The ability to run reactions at room temperature significantly lowers utility costs associated with heating and cooling, contributing to a lower overall cost of goods sold. Furthermore, the use of benign solvents and the absence of hazardous oxidants improve the safety profile of the manufacturing site, potentially lowering insurance premiums and regulatory compliance burdens.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts and stoichiometric oxidants with electricity and cheap iodide salts results in substantial raw material savings. By removing the need for metal scavengers and extensive purification steps to meet residual metal specifications, the downstream processing costs are significantly reduced. The energy efficiency of running reactions at ambient temperature further contributes to a leaner manufacturing cost structure, allowing for more competitive pricing in the global market. This economic advantage is compounded by the high atom economy of the electrochemical process, which minimizes waste disposal fees and maximizes the yield of the valuable sultam scaffold.
- Enhanced Supply Chain Reliability: Sourcing simple inorganic salts like sodium iodide is far more stable and predictable than relying on specialized organometallic catalysts that may have long lead times or single-source suppliers. The robustness of the reaction under air atmosphere means that production schedules are less likely to be disrupted by equipment failures related to inert gas systems or gloveboxes. This reliability ensures a consistent supply of high-purity intermediates, enabling pharmaceutical partners to maintain their own production timelines without interruption. The scalability of the electrochemical setup allows for flexible production volumes, adapting quickly to fluctuations in market demand without the need for massive capital investment in new reactor types.
- Scalability and Environmental Compliance: The electrochemical nature of this synthesis aligns perfectly with modern environmental regulations, as it generates minimal hazardous waste compared to traditional oxidation methods. The absence of heavy metal contamination simplifies the treatment of effluent streams, reducing the environmental footprint of the manufacturing facility. Scaling this process from gram to kilogram quantities is straightforward, as electrochemical reactors can be easily paralleled or scaled up using established flow chemistry technologies. This ease of scale-up ensures that the transition from R&D to commercial production is seamless, supporting the rapid deployment of new drug candidates to the market while maintaining strict adherence to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of sultam derivatives, based on the detailed experimental data provided in the patent literature. These insights are designed to clarify the operational feasibility and strategic benefits of adopting this technology for large-scale manufacturing. Understanding these nuances helps stakeholders make informed decisions about integrating this method into their existing production portfolios.
Q: Does this electrochemical method require expensive transition metal catalysts?
A: No, the method described in patent CN114525527A eliminates the need for precious metal catalysts like palladium or iron, utilizing inexpensive iodide salts instead.
Q: What are the optimal solvent conditions for this sultam synthesis?
A: The process utilizes a mixed solvent system of acetonitrile and water, typically in a volume ratio ranging from 2:1 to 4:1, to ensure high conductivity and solubility.
Q: Is the reaction sensitive to air or moisture?
A: The reaction is robust and can be conducted under air atmosphere at room temperature, removing the need for rigorous inert gas protection or anhydrous conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sultam Derivative Supplier
As the pharmaceutical industry continues to demand greener and more cost-effective synthetic routes, NINGBO INNO PHARMCHEM stands at the forefront of implementing these advanced electrochemical technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is smooth and efficient. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of sultam derivatives meets the highest standards required for API synthesis. Our expertise in handling complex heterocyclic chemistry allows us to optimize reaction parameters for maximum yield and minimal impurity formation, providing our partners with a distinct competitive advantage.
We invite potential partners to engage with our technical procurement team to discuss how this innovative electrochemical method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this metal-free protocol for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your next project benefits from the latest advancements in sustainable organic synthesis. Let us be your trusted partner in navigating the complexities of modern chemical manufacturing.
