Advanced Electrochemical Synthesis of Sultam Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient synthetic routes to access complex heterocyclic scaffolds essential for drug development. A groundbreaking approach detailed in patent CN114525527B introduces a novel electrochemical synthesis method for producing sultam derivatives, specifically transforming 2-isoallylbenzenesulfonamide derivatives into valuable cyclic structures. This technology represents a paradigm shift from traditional transition-metal catalysis to sustainable electro-organic synthesis, utilizing electricity as a clean reagent to drive molecular transformations. By leveraging inexpensive iodide salts and operating under ambient conditions, this method addresses critical pain points regarding cost, environmental impact, and operational simplicity. For R&D directors and procurement managers alike, this innovation offers a compelling alternative to legacy processes that rely on scarce resources and harsh reaction environments. The ability to generate high-value intermediates without external oxidants or reducing agents underscores the potential for substantial cost reduction in pharmaceutical intermediate manufacturing.
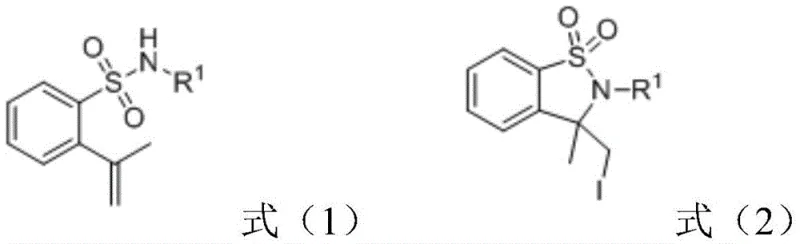
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of sultam frameworks has relied heavily on transition-metal catalysis, often employing precious metals such as palladium or iron complexes to facilitate cyclization. These conventional pathways frequently necessitate the use of stoichiometric amounts of chemical oxidants or reductants, which act as sacrificial reagents and generate significant quantities of hazardous waste. Furthermore, the reliance on expensive ligands and sensitive catalysts introduces complexity into the supply chain, creating vulnerabilities related to raw material availability and price volatility. High reaction temperatures and the need for inert atmospheres further escalate energy consumption and operational costs, making these processes less attractive for large-scale commercial production. The presence of residual heavy metals in the final product also poses a significant regulatory hurdle, requiring rigorous and costly purification steps to meet stringent pharmaceutical quality standards. Consequently, there is an urgent industry demand for methodologies that can bypass these limitations while maintaining high efficiency and selectivity.
The Novel Approach
The electrochemical strategy outlined in the patent data offers a transformative solution by replacing chemical oxidants with electrons, effectively turning the electrode into a traceless reagent. This method utilizes cheap and abundant iodide salts, such as sodium iodide, in a mixed solvent system of acetonitrile and water to drive the intramolecular cyclization efficiently. Operating at room temperature and under an air atmosphere eliminates the need for energy-intensive heating or specialized inert gas equipment, drastically simplifying the reactor setup. The absence of transition metal catalysts not only reduces raw material costs but also simplifies the downstream purification process by removing the risk of metal contamination. This green chemistry approach aligns perfectly with modern sustainability goals, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. The versatility of the reaction is demonstrated by its tolerance to various substituents on the aromatic ring, ensuring broad applicability across different drug candidates.
Mechanistic Insights into Electrochemical Iodocyclization
The core of this synthetic breakthrough lies in the anodic oxidation of iodide ions to generate reactive iodine species in situ, which then initiate the cyclization cascade. At the anode, iodide ions lose electrons to form molecular iodine or iodine radicals, which subsequently react with the alkene moiety of the 2-isoallylbenzenesulfonamide substrate. This electrophilic activation triggers an intramolecular nucleophilic attack by the sulfonamide nitrogen, leading to the formation of the five-membered sultam ring structure. The cathodic process typically involves the reduction of protons to hydrogen gas, balancing the electron flow without introducing foreign chemical species into the reaction mixture. This elegant mechanism ensures that the only byproducts are benign, avoiding the accumulation of toxic waste streams associated with traditional oxidants like peroxides or hypervalent iodine reagents. Understanding this redox cycle is crucial for optimizing current density and electrode materials to maximize Faradaic efficiency and product yield.
From an impurity control perspective, the metal-free nature of this electrochemical protocol provides a distinct advantage in managing the杂质 profile of the final active pharmaceutical ingredient. Traditional metal-catalyzed routes often suffer from difficult-to-remove metal traces that can catalyze degradation pathways or trigger toxicity concerns during clinical trials. By eliminating palladium, iron, or copper from the reaction equation, the burden on the purification team is significantly lightened, allowing for simpler crystallization or chromatography steps. The use of carbon electrodes further minimizes the risk of electrode corrosion contributing to particulate matter in the product. Moreover, the mild reaction conditions help preserve sensitive functional groups that might otherwise decompose under the harsh thermal or oxidative stress of conventional methods. This results in a cleaner crude reaction mixture, facilitating higher overall recovery rates and reducing the loss of valuable intermediates during workup.
How to Synthesize Sultam Derivatives Efficiently
To implement this electrochemical synthesis effectively, precise control over reaction parameters such as current intensity and solvent composition is essential for reproducibility. The patent specifies a molar ratio of substrate to iodide between 1:2 and 1:2.6, ensuring sufficient iodine generation to drive the reaction to completion without excessive reagent waste. A mixed solvent system of acetonitrile and water in a volume ratio of roughly 4:1 provides the optimal balance of solubility for organic substrates and conductivity for the electrolyte. The reaction is typically conducted using carbon rods as both anode and cathode, applying a constant current of 5mA to 15mA for a duration of 1.5 to 3 hours. Following the electrolysis, standard workup procedures involving extraction with ethyl acetate and drying over anhydrous sodium sulfate yield the crude product ready for purification. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining 2-isoallylbenzenesulfonamide derivatives and sodium iodide in a mixed solvent of acetonitrile and water.
- Conduct the electrolysis using carbon electrodes at a constant current of 5mA to 15mA under room temperature and air atmosphere for 1.5 to 3 hours.
- Perform workup by extracting with ethyl acetate, drying over anhydrous sodium sulfate, and purifying the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical methodology presents a strategic opportunity to optimize manufacturing costs and enhance supply security. The elimination of precious metal catalysts removes a major cost driver and mitigates the risk associated with the volatile pricing of commodities like palladium. Additionally, the use of commodity chemicals such as sodium iodide and common solvents ensures a stable and diversified supply base, reducing dependency on specialized reagent vendors. The simplified operational requirements, including the ability to run reactions at room temperature and in air, lower the barrier to entry for contract manufacturing organizations and reduce utility costs. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the complete removal of expensive transition metal catalysts and ligands, which traditionally account for a significant portion of raw material expenses in fine chemical synthesis. By substituting these with inexpensive electricity and bulk iodide salts, the variable cost per kilogram of product is drastically lowered. Furthermore, the absence of stoichiometric oxidants reduces the cost of reagents and the expense associated with disposing of hazardous chemical waste. The simplified purification process also translates to lower labor and consumable costs during the isolation phase. Overall, this creates a leaner manufacturing model that improves margin potential for commercial production.
- Enhanced Supply Chain Reliability: Relying on widely available reagents like sodium iodide and carbon electrodes minimizes the risk of supply disruptions that often plague specialized catalyst markets. The robustness of the reaction conditions allows for flexible manufacturing scheduling without the need for complex infrastructure upgrades or strict environmental controls. This flexibility enables faster response times to market demand fluctuations and shortens the lead time for high-purity pharmaceutical intermediates. Suppliers can maintain consistent inventory levels of key starting materials, ensuring uninterrupted production cycles even during global supply chain stresses. This reliability is critical for maintaining the continuity of drug development pipelines.
- Scalability and Environmental Compliance: Electrochemical processes are inherently scalable, as increasing production capacity often involves adding more electrode surface area or running parallel reactors rather than redesigning the entire process. The green nature of this synthesis, characterized by atom economy and reduced waste generation, aligns with increasingly stringent environmental regulations globally. Avoiding heavy metals and harsh oxidants simplifies wastewater treatment and reduces the environmental footprint of the manufacturing facility. This compliance advantage not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is becoming a key criterion for partnerships with major pharmaceutical companies. The process is well-suited for continuous flow electrochemistry, offering a clear path to ton-scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios. The answers reflect the practical realities of scaling electro-organic reactions in an industrial setting.
Q: What are the primary advantages of this electrochemical method over traditional palladium catalysis?
A: The electrochemical method eliminates the need for expensive precious metal catalysts like palladium and avoids the use of stoichiometric oxidants or reductants, significantly reducing raw material costs and metal residue concerns in the final API.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process operates under mild conditions (room temperature, air atmosphere) using simple carbon electrodes and common solvents, making it highly suitable for commercial scale-up without complex safety measures required for high-pressure or high-temperature reactions.
Q: What is the typical yield and purity profile for these sultam derivatives?
A: The patent reports high yields ranging from 80% to 96% across various substrates, with simple purification steps via column chromatography ensuring high purity suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sultam Derivatives Supplier
As the demand for efficient and sustainable synthetic routes grows, partnering with an experienced CDMO becomes essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves smoothly from gram-scale optimization to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle electrochemical processes safely and efficiently, adhering to stringent purity specifications required by global regulatory bodies. With rigorous QC labs and a dedicated team of process chemists, we guarantee the delivery of high-quality intermediates that meet your exacting standards. We are committed to being a reliable sultam derivatives supplier that supports your long-term growth.
We invite you to contact our technical procurement team to discuss how this electrochemical technology can be tailored to your specific project needs. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this metal-free route for your pipeline. Our experts are ready to provide specific COA data and route feasibility assessments to help you make informed decisions. Let us collaborate to accelerate your drug development timeline while optimizing your manufacturing costs.
