Scalable Synthesis of Phenethylamine Derivatives Using Robust Nickel Catalysis for Commercial API Production
The pharmaceutical industry continuously seeks robust, scalable, and economically viable synthetic routes for active pharmaceutical ingredients (APIs) and their key intermediates. Patent CN1481354A presents a significant technological advancement in the preparation of phenethylamine derivatives, a class of compounds critically important as synthetic intermediates for central nervous system (CNS) antidepressants, most notably Venlafaxine. This patent discloses a novel hydrogenation process that replaces expensive noble metal catalysts with more economical Nickel or Cobalt-based systems. By shifting away from traditional Rhodium-catalyzed methods, this technology addresses critical pain points regarding raw material costs and catalyst recycling complexities. The invention specifically targets the reduction of phenylacetonitrile derivatives to their corresponding amines with high selectivity and yield. For global procurement teams and R&D directors, this represents a strategic opportunity to optimize the supply chain for high-volume antidepressant production. The process utilizes readily available base metals and standard hydrogenation equipment, making it highly adaptable for commercial scale-up from kilogram to multi-ton scales.
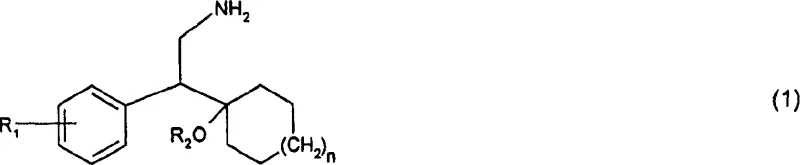
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in US-A-4,535,186, have historically relied on the use of Rhodium catalysts for the hydrogenation of nitrile precursors to phenethylamine derivatives. While Rhodium is known for its high catalytic activity, its application in large-scale industrial manufacturing is fraught with significant economic and logistical disadvantages. The primary limitation is the exorbitant cost associated with Platinum Group Metals (PGMs), which directly inflates the cost of goods sold (COGS) for the final API. Furthermore, to mitigate these costs, manufacturers are forced to implement complex catalyst recovery and recycling loops. These recycling processes add additional unit operations to the manufacturing workflow, increasing processing time and energy consumption. There is also a inherent risk that the catalytic activity or selectivity may degrade upon recycling, leading to batch-to-batch variability and potential quality issues. The reliance on a single source or a limited supply chain for Rhodium also introduces substantial supply chain vulnerability, where geopolitical instability or mining disruptions can halt production entirely.
The Novel Approach
The present invention offers a transformative solution by substituting noble metals with Nickel or Cobalt catalysts, specifically Raney Nickel or Raney Cobalt. This shift fundamentally alters the economic model of the synthesis, replacing a precious metal with abundant base metals. The novelty lies not just in the metal substitution, but in the specific activation and pretreatment protocols that ensure the base metal catalysts perform with comparable or superior efficiency to their noble counterparts. The process involves hydrogenating the nitrile precursor in the presence of these catalysts, often promoted with specific additives to enhance performance. This approach eliminates the need for expensive metal recovery infrastructure, simplifying the plant design and operational overhead. By utilizing standard hydrogenation conditions compatible with existing stainless steel autoclaves, the technology facilitates immediate adoption without requiring capital-intensive equipment upgrades. The result is a streamlined, cost-effective pathway that maintains high chemical fidelity while drastically reducing the financial burden on the manufacturer.

Mechanistic Insights into Nickel-Catalyzed Nitrile Hydrogenation
The core of this technology is the heterogeneous hydrogenation of the cyano group (-CN) to a primary amine (-CH2NH2). In the presence of a Nickel or Cobalt catalyst, molecular hydrogen is adsorbed onto the metal surface and activated. The nitrile substrate then coordinates to the active sites, undergoing stepwise reduction. A critical challenge in nitrile hydrogenation is the prevention of secondary and tertiary amine formation, which occurs when the intermediate imine reacts with the primary amine product. The patent addresses this by conducting the reaction in the presence of a base, such as ammonia or ammonium hydroxide. The excess ammonia competes for the active sites and reacts with any imine intermediates, effectively suppressing the formation of unwanted by-products and driving the selectivity towards the desired primary amine. Furthermore, the patent highlights the importance of catalyst pretreatment. Modifying the Raney Nickel surface with carboxylic acids (like acetic acid) or transition metal compounds (such as Vanadium acetylacetonate) alters the electronic properties of the catalyst surface. This promotion enhances the catalyst's ability to discriminate between the nitrile group and other sensitive functional groups, ensuring high chemoselectivity.

Impurity control is rigorously managed through both the catalytic step and the downstream purification strategy. The use of promoted catalysts minimizes the generation of over-reduced species or coupling by-products during the hydrogenation phase. Following the reaction, the crude amine is often converted into a salt form, such as the formate salt, which serves as a powerful purification handle. The solubility differences between the desired amine formate and organic impurities allow for effective crystallization. As demonstrated in the patent examples, recrystallization of the formate salt can elevate the purity of the intermediate to greater than 98% by HPLC. This solid-state purification step is crucial for removing trace metal residues and organic side products before the final conversion to the hydrochloride salt or further derivatization. This dual-layer approach of catalytic selectivity followed by crystallization-based purification ensures that the final material meets the stringent quality standards required for pharmaceutical intermediates.
How to Synthesize Venlafaxine Intermediate Efficiently
The synthesis of the target phenethylamine derivative, specifically the Venlafaxine intermediate, follows a logical progression from the nitrile precursor. The process begins with the careful preparation of the catalyst, where Raney Nickel is washed and treated with a promoter solution to activate its surface. This pretreated catalyst is then charged into a pressure vessel along with the nitrile starting material, an alcoholic solvent like ethanol or methanol, and a source of ammonia. The system is pressurized with hydrogen and heated to moderate temperatures, typically between 20°C and 80°C, to drive the reduction to completion. Once the hydrogen uptake ceases, indicating full conversion, the catalyst is filtered off, and the filtrate is processed to isolate the amine. The detailed standardized synthesis steps, including specific stoichiometric ratios, temperature ramps, and workup procedures, are outlined in the guide below.
- Pretreat the Raney Nickel or Cobalt catalyst with a modifying agent such as acetic acid, vanadium compounds, or tungsten compounds to enhance selectivity and activity.
- Charge the autoclave with the phenylacetonitrile precursor, alcohol solvent, aqueous ammonia, and the pretreated catalyst under an inert atmosphere.
- Conduct hydrogenation at 20-120°C and 1-200 bar pressure, followed by filtration and salt formation to isolate the high-purity amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Nickel-catalyzed process offers profound strategic benefits that extend beyond simple chemistry. The most immediate impact is on the raw material cost structure. By eliminating the dependency on Rhodium, a metal known for its extreme price volatility and scarcity, manufacturers can stabilize their long-term cost projections. The switch to Nickel, a commodity metal with a deep and liquid global market, insulates the production budget from the speculative fluctuations common in the precious metals sector. Additionally, the simplification of the process flow—removing the need for complex noble metal recovery units—reduces both capital expenditure (CAPEX) for new facilities and operating expenditure (OPEX) for existing ones. This leaner operational footprint translates directly into improved margins and a more competitive pricing position in the global API market.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts results in substantial cost savings. Unlike Rhodium, which requires rigorous and costly recycling protocols to be economically feasible, Nickel and Cobalt catalysts are inexpensive enough that even if they were single-use, the cost benefit would be significant. However, the patent further enhances this advantage by demonstrating that the catalyst can be reused multiple times without significant loss of performance. This reusability means the effective catalyst cost per kilogram of product becomes negligible. Furthermore, the use of common solvents like ethanol and methanol, rather than specialized organic media, reduces solvent procurement and disposal costs, contributing to a lower overall manufacturing cost profile.
- Enhanced Supply Chain Reliability: Relying on base metals significantly de-risks the supply chain. The global supply of Nickel and Cobalt is robust, with multiple sourcing options across different geographies, unlike Rhodium which is sourced from a very limited number of mines primarily in South Africa and Russia. This diversification ensures continuity of supply even during regional disruptions. Moreover, the catalyst's ability to be reused reduces the frequency of catalyst replenishment orders, simplifying inventory management and reducing the administrative burden on the procurement team. The stability of the catalyst over multiple cycles, as evidenced by the patent data, guarantees consistent production throughput without unexpected stops for catalyst replacement or regeneration.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard high-pressure hydrogenation equipment found in most fine chemical plants. The reaction conditions (moderate temperature and pressure) are safe and manageable on a multi-ton scale. From an environmental perspective, the removal of heavy noble metals from the waste stream simplifies effluent treatment. While Nickel and Cobalt still require proper handling, the absence of Rhodium reduces the toxicity profile of the waste. The high selectivity of the reaction also minimizes the formation of by-products, leading to higher atom economy and less chemical waste generation. This aligns with modern green chemistry principles and helps facilities meet increasingly stringent environmental regulations without costly end-of-pipe treatments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogenation technology. These answers are derived directly from the experimental data and claims within patent CN1481354A, providing a factual basis for evaluating the process feasibility. Understanding these details is essential for technical teams assessing the transferability of this method to their own manufacturing sites.
Q: What is the primary advantage of using Nickel catalysts over Rhodium for Venlafaxine intermediates?
A: The primary advantage is economic efficiency and supply chain stability. Rhodium is a precious metal with high volatility in pricing and availability, whereas Nickel and Cobalt are base metals that are significantly more affordable and readily available globally, reducing the overall cost of goods sold (COGS) without compromising yield.
Q: Can the Nickel catalyst be reused in this hydrogenation process?
A: Yes, the patent explicitly demonstrates that the Nickel or Cobalt catalyst can be reused multiple times (up to 100 times theoretically, with examples showing stable performance over 4 cycles) without significant loss of activity or selectivity, provided appropriate pretreatment and washing protocols are followed.
Q: How is high purity achieved in the final phenethylamine derivative?
A: High purity (>98%) is achieved through a combination of selective hydrogenation using promoted catalysts (e.g., Vanadium-promoted Raney Nickel) and a subsequent purification step involving the formation and recrystallization of the formate salt, which effectively removes impurities before conversion to the final hydrochloride salt.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Venlafaxine Intermediate Supplier
The technological pathway outlined in CN1481354A demonstrates immense potential for optimizing the production of CNS antidepressant intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this patented chemistry into commercial reality. As a seasoned CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our engineering teams are adept at handling high-pressure hydrogenation reactions safely and efficiently, ensuring that the benefits of the Nickel-catalyzed route are fully realized in a GMP-compliant environment. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch meets the required identity and purity profiles before release.
We invite you to collaborate with us to leverage this cost-effective synthesis route for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data from our pilot runs and comprehensive route feasibility assessments. Let us help you secure a stable, high-quality supply of phenethylamine derivatives while significantly reducing your manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
