Scalable Synthesis of Enantiopure Tert-Butyl Sulfinamide for Industrial Pharmaceutical Applications
The pharmaceutical industry's relentless pursuit of chiral purity has placed enantiopure tert-butyl sulfinamide at the forefront of asymmetric synthesis strategies. As a critical chiral auxiliary, this compound facilitates the construction of chiral amines found in numerous blockbuster drugs, including antiallergics and anticancer agents. Patent CN108409615B, published in September 2020, introduces a transformative methodology for synthesizing this vital intermediate. Unlike traditional pathways that rely on hazardous cryogenic conditions and prolonged reaction times, this invention offers a streamlined, environmentally benign route. By leveraging selective oxidation and efficient chiral resolution, the disclosed process addresses the longstanding bottlenecks of scalability and cost. For R&D directors and supply chain leaders, this patent represents a significant leap forward in securing a reliable supply of high-purity chiral building blocks essential for modern drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of enantiopure tert-butyl sulfinamide has been plagued by severe operational and environmental challenges that hinder industrial adoption. One prevalent conventional route involves the asymmetric oxidation of tert-butyl disulfide using vanadium catalysts followed by reduction under cryogenic conditions with liquid ammonia and metal lithium. As illustrated in the reaction scheme below, this pathway is not only technically demanding but also economically inefficient due to the excessive consumption of liquid ammonia and the generation of hazardous waste.
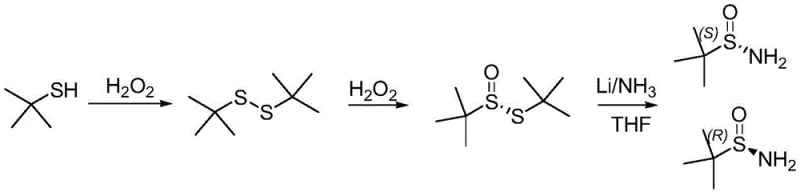
Furthermore, the reaction time for the asymmetric chiral oxidation step in these traditional methods can extend up to four days, creating a significant bottleneck for production throughput. The reliance on liquid ammonia poses serious safety risks and complicates waste management, as the ammonia discharge process is difficult to recycle effectively. Additionally, the use of tert-butyl mercaptan as a starting material in some variations leads to the release of foul odors, causing severe environmental pollution and requiring expensive abatement systems. These factors collectively result in high production costs and limited scalability, making conventional methods unsuitable for the growing demand of the global pharmaceutical market.
The Novel Approach
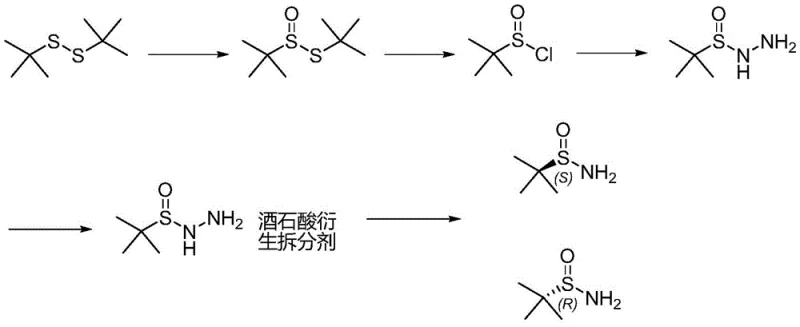
In stark contrast, the novel approach detailed in patent CN108409615B circumvents these issues through a clever redesign of the synthetic sequence. The new method initiates with the selective oxidation of tert-butyl disulfide using hydrogen peroxide, a greener oxidant, to form tert-butyl thiosulfinate. This intermediate is then converted into a sulfinyl halide using standard acylating reagents, avoiding the need for complex chiral catalysts in the initial oxidation step. The subsequent formation of tert-butyl sulfinyl hydrazine allows for a highly effective resolution using tartaric acid derivatives. This strategic shift moves the chirality introduction to a later stage where separation is more efficient and less energy-intensive. The final reduction step utilizes zinc and acetic acid under mild thermal conditions, completely eliminating the need for cryogenic liquid ammonia.
Mechanistic Insights into Selective Oxidation and Chiral Resolution
The core of this technological breakthrough lies in the precise control of oxidation states and the efficiency of the resolution mechanism. The process begins with the catalytic oxidation of tert-butyl disulfide. By employing a vanadium catalyst such as VO(acac)2 in ethanol or acetic acid, the reaction selectively targets the sulfur-sulfur bond to generate tert-butyl thiosulfinate with high atom economy. The reaction temperature is carefully maintained between 25°C and 35°C, ensuring rapid conversion within 3 to 7 hours, a drastic improvement over the multi-day timelines of prior art. This mild condition preserves the integrity of the sensitive sulfur species while minimizing side reactions that could lead to sulfone impurities.
Following the formation of the thiosulfinate, the introduction of chirality is achieved through a robust resolution strategy rather than asymmetric catalysis. The thiosulfinate is first converted to a sulfinyl chloride or bromide using reagents like NBS, NCS, or halogens at low temperatures (0°C to 10°C). This reactive intermediate is then treated with hydrazine hydrate to yield tert-butylsulfinyl hydrazide. The true elegance of the mechanism is revealed in the resolution step, where tartaric acid derivatives such as DTTA or DBTA form diastereomeric salts with the racemic hydrazide. These salts exhibit distinct solubility profiles, allowing for the isolation of single enantiomers with an enantiomeric excess (EE) exceeding 90% initially, and up to 99% after recrystallization. The final cleavage of the N-S bond using zinc powder in acetic acid proceeds smoothly at 40-45°C, delivering the target enantiopure tert-butyl sulfinamide with exceptional purity.
How to Synthesize Tert-Butyl Sulfinamide Efficiently
The synthesis of enantiopure tert-butyl sulfinamide via this patented route offers a practical blueprint for laboratory and pilot-scale operations. The procedure is designed to maximize yield while minimizing the handling of hazardous materials. By following the specific sequence of selective oxidation, halogenation, hydrazide formation, and chiral resolution, manufacturers can achieve consistent results. The detailed standardized synthesis steps provided below outline the precise conditions required to replicate the high purity and enantiomeric excess reported in the patent documentation.
- Selectively oxidize tert-butyl disulfide with hydrogen peroxide and a vanadium catalyst to form tert-butyl thiosulfinate.
- React the thiosulfinate with an acylating agent to generate tert-butyl sulfinyl chloride or bromide.
- Convert the sulfinyl halide to hydrazide using hydrazine hydrate, followed by chiral resolution with tartaric acid derivatives and zinc reduction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers compelling economic and logistical benefits. The elimination of cryogenic conditions and the reduction in reaction time directly translate to lower energy consumption and increased facility throughput. By removing the dependency on liquid ammonia and complex vanadium-based asymmetric catalysts, the process simplifies the supply chain and reduces exposure to volatile raw material markets. The ability to recycle solvents and resolving agents further drives down the cost of goods sold, making the production of high-purity chiral intermediates more financially sustainable.
- Cost Reduction in Manufacturing: The new process significantly lowers production costs by replacing expensive and hazardous reagents with cheap, commercially available alternatives like hydrogen peroxide and zinc powder. The avoidance of cryogenic liquid ammonia eliminates the need for specialized low-temperature equipment and the associated high energy costs for cooling. Furthermore, the recovery and reuse of tartaric acid resolving agents and organic solvents create a closed-loop system that minimizes waste disposal expenses. These cumulative efficiencies result in a substantially reduced cost structure for manufacturing enantiopure tert-butyl sulfinamide compared to legacy methods.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is straightforward, as tert-butyl disulfide and hydrogen peroxide are commodity chemicals with stable global supply chains. Unlike methods relying on specialized chiral ligands or difficult-to-handle gases, the reagents used here are robust and easy to transport. The simplified workflow reduces the risk of production delays caused by equipment failure or reagent shortages. This reliability ensures a consistent supply of critical chiral auxiliaries, safeguarding downstream drug manufacturing schedules against interruptions.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, operating at near-ambient temperatures and atmospheric pressure in key steps. The absence of foul-smelling tert-butyl mercaptan emissions and the reduction of ammonia waste align with stringent environmental regulations, facilitating easier permitting and operation in regulated jurisdictions. The high atom utilization rate and the potential for solvent recycling demonstrate a commitment to green chemistry principles, enhancing the corporate sustainability profile of manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of enantiopure tert-butyl sulfinamide. These answers are derived directly from the technical specifications and experimental data provided in the patent literature. They serve to clarify the operational parameters and quality standards achievable through this advanced manufacturing route.
Q: What are the primary advantages of this new synthesis method over conventional routes?
A: The new method eliminates the need for cryogenic liquid ammonia conditions and significantly reduces reaction time from days to hours. It utilizes cheap, easily obtained raw materials and allows for the recycling of solvents and resolving agents, making it highly suitable for industrial mass production.
Q: How is high enantiomeric excess (EE) achieved in this process?
A: High EE is achieved through a robust resolution step using tartaric acid derivatives such as DTTA or DBTA. The process forms a chiral salt which is then dissociated, yielding products with an EE greater than 99% after purification.
Q: Is this process environmentally friendly compared to traditional methods?
A: Yes, the process avoids the generation of foul-smelling tert-butyl mercaptan emissions associated with some older routes and eliminates the massive discharge of liquid ammonia. The ability to recycle solvents and resolving agents further enhances its environmental profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tert-Butyl Sulfinamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of tert-butyl sulfinamide meets the highest industry standards for enantiomeric excess and chemical purity. Our commitment to excellence makes us a trusted partner for pharmaceutical companies seeking to optimize their supply chains.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your drug development timeline with reliable, cost-effective chiral solutions.
