Scalable Synthesis of Enantiopure Tert-Butyl Sulfinamide for Chiral Drug Manufacturing
Introduction to Advanced Chiral Auxiliary Synthesis
The pharmaceutical industry's relentless pursuit of enantiomerically pure drugs has placed chiral auxiliaries like tert-butyl sulfinamide at the forefront of synthetic strategy. Patent CN108409615B introduces a groundbreaking methodology for synthesizing enantiopure tert-butyl sulfinamide, addressing critical bottlenecks in traditional manufacturing. This innovation shifts the paradigm from hazardous, cryogenic-dependent processes to a streamlined, ambient-temperature workflow that leverages selective oxidation and chiral resolution. By replacing volatile liquid ammonia and expensive metal reductants with stable hydrazine derivatives and zinc powder, this technology offers a robust pathway for producing high-purity chiral building blocks essential for complex API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically pure tert-butyl sulfinamide has been plagued by severe operational hazards and inefficiencies. Traditional routes often rely on the asymmetric oxidation of tert-butyl disulfide using vanadium catalysts followed by reduction under cryogenic conditions involving liquid ammonia and metallic lithium. These processes are not only energy-intensive due to the requirement for extremely low temperatures but also pose significant safety risks associated with handling large volumes of liquid ammonia. Furthermore, the generation of foul-smelling tert-butyl mercaptan as a byproduct creates substantial environmental compliance challenges, while the extended reaction times, often spanning up to four days for asymmetric oxidation, severely limit throughput and increase production costs.
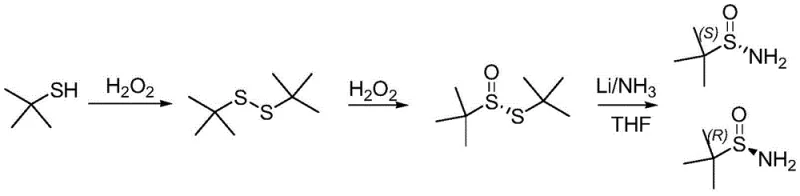
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a温和 (mild) and highly efficient sequence starting from the selective oxidation of tert-butyl disulfide. Instead of dangerous cryogenic reductions, the process employs hydrazine hydrate to form a stable sulfinyl hydrazide intermediate, which is then subjected to chiral resolution using tartaric acid derivatives. This strategic shift eliminates the need for liquid ammonia entirely, drastically simplifying the equipment requirements and safety protocols. The final cleavage step uses zinc powder in acetic acid at moderate temperatures (40-45°C), ensuring a safe, scalable, and environmentally friendly conclusion to the synthesis that is perfectly suited for industrial amplification.
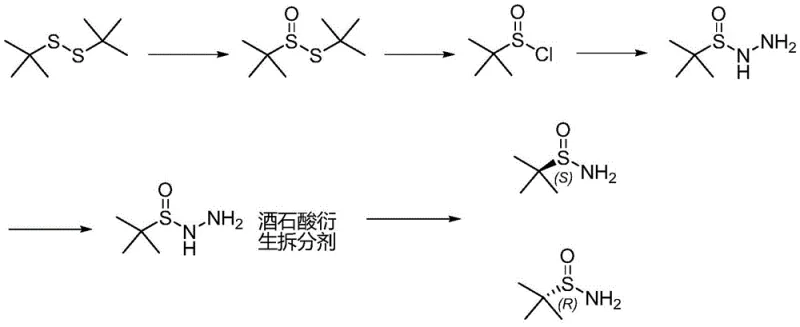
Mechanistic Insights into Selective Oxidation and Chiral Resolution
The core of this technological breakthrough lies in the precise control of oxidation states and stereochemistry. The process initiates with the catalytic oxidation of tert-butyl disulfide using hydrogen peroxide and a vanadium catalyst (VO(acac)2) at controlled temperatures between 25°C and 35°C. This step selectively generates tert-butyl thiosulfinate with high atom economy, avoiding over-oxidation to sulfones. Subsequent reaction with acylating agents such as N-bromosuccinimide (NBS) or chlorine gas at 0-10°C converts the thiosulfinate into the reactive tert-butyl sulfinyl halide. This intermediate is then captured by hydrazine hydrate to form the key tert-butylsulfinyl hydrazide, a stable precursor that facilitates the subsequent separation of enantiomers without the racemization risks associated with harsher conditions.
Chiral purity is achieved through a sophisticated resolution strategy utilizing derivatives of tartaric acid, such as Di-p-toluoyl-tartaric acid (DTTA) or Dibenzoyl-L-tartaric acid (DBTA). The racemic hydrazide forms diastereomeric salts with these resolving agents, which exhibit different solubilities allowing for effective separation via crystallization. Following dissociation, the optically pure hydrazide undergoes reductive cleavage using zinc powder in acetic acid. This final transformation cleanly removes the hydrazine moiety to yield the target enantiopure tert-butyl sulfinamide. The entire mechanism is designed to maximize yield, with reported external standard yields reaching up to 93% in the oxidation step and maintaining high enantiomeric excess (EE > 99%) throughout the purification stages.
How to Synthesize Enantiopure Tert-Butyl Sulfinamide Efficiently
Implementing this synthesis requires careful attention to temperature control during the exothermic oxidation and halogenation phases to prevent side reactions. The protocol begins with the preparation of tert-butyl thiosulfinate, followed by conversion to the sulfinyl halide and subsequent hydrazide formation. The critical resolution step involves refluxing the hydrazide with the chiral acid in a solvent like 2-methyltetrahydrofuran or ethyl acetate, followed by slow cooling to induce crystallization of the desired diastereomer. For a comprehensive guide on reagent ratios, specific workup procedures, and purification techniques including low-temperature pulping with n-heptane and toluene, please refer to the standardized protocol below.
- Oxidize tert-butyl disulfide with hydrogen peroxide and a vanadium catalyst to form tert-butyl thiosulfinate.
- React the thiosulfinate with an acylating agent like NBS or chlorine to generate tert-butyl sulfinyl halide.
- Convert the halide to tert-butylsulfinyl hydrazide using hydrazine hydrate, followed by resolution with tartaric acid derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, this patented process represents a significant opportunity to optimize the sourcing of critical chiral intermediates. By eliminating the dependency on cryogenic infrastructure and hazardous reagents like liquid ammonia, the manufacturing footprint is drastically reduced, leading to lower capital expenditure and operational overheads. The use of commercially available, low-cost raw materials such as tert-butyl disulfide and hydrogen peroxide ensures a stable supply chain that is less susceptible to the volatility often seen with specialized organometallic reagents. Furthermore, the ability to recycle solvents and resolving agents contributes to a more sustainable and cost-effective production model.
- Cost Reduction in Manufacturing: The elimination of expensive metal catalysts and cryogenic cooling systems results in substantial cost savings across the production lifecycle. The process operates at near-ambient temperatures for key steps, significantly reducing energy consumption compared to traditional methods that require maintaining temperatures below -70°C. Additionally, the high atom utilization rate and the recovery of resolving agents mean that raw material costs are minimized, allowing for a more competitive pricing structure for the final high-purity product without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit from the use of bulk commodity chemicals rather than niche, hazard-classified reagents. The removal of liquid ammonia from the process flow mitigates regulatory hurdles and transportation risks associated with hazardous materials, ensuring smoother logistics and fewer delays. The robustness of the synthesis, characterized by high yields and simple operation, guarantees consistent batch-to-batch quality, which is crucial for maintaining uninterrupted supply lines for downstream pharmaceutical manufacturing partners who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, avoiding the engineering complexities of handling large volumes of toxic gases or pyrophoric metals. Waste streams are easier to manage and treat, aligning with increasingly stringent global environmental regulations regarding VOC emissions and hazardous waste disposal. The simplified workup procedures, involving standard extraction and crystallization techniques, facilitate rapid scale-up from pilot plant to multi-ton commercial production, enabling suppliers to respond quickly to surging market demand for chiral drug intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is vital for R&D teams evaluating process transfer and procurement officers assessing vendor capabilities. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance for decision-making purposes regarding quality assurance and regulatory compliance.
Q: How does this new method improve upon traditional liquid ammonia routes?
A: The patented process eliminates the need for cryogenic liquid ammonia and metal lithium, replacing them with mild hydrazine hydrate reactions and zinc/acetic acid cleavage, significantly improving safety and operational simplicity.
Q: What level of enantiomeric excess (EE) can be achieved?
A: Through optimized resolution using agents like DTTA or DBTA followed by low-temperature pulping, the process consistently achieves an EE greater than 99% and chemical purity exceeding 99%.
Q: Is the process suitable for large-scale industrial production?
A: Yes, the method utilizes cheap, easily obtained raw materials and avoids foul-smelling byproducts and hazardous cryogenic conditions, making it highly scalable and environmentally compliant for mass manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tert-Butyl Sulfinamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral auxiliaries play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of tert-butyl sulfinamide meets the exacting standards required for GMP pharmaceutical manufacturing, providing our partners with the confidence needed to advance their drug candidates.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient route. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique project requirements, ensuring a collaborative approach to achieving your production goals.
