Scalable Asymmetric Synthesis of Optical Pure Nicotine via Protection-Free Catalysis
Introduction to Advanced Nicotine Manufacturing Technologies
The global demand for high-purity optical isomers of nicotine has surged, driven by its expanding applications in pharmaceutical therapies for neurodegenerative diseases and as a specialized agrochemical intermediate. Traditional extraction methods from tobacco plants are increasingly insufficient due to supply chain volatility and purity limitations, necessitating robust chemical synthesis alternatives. Patent CN114874134A introduces a groundbreaking protection-free asymmetric synthesis process that addresses these critical industry pain points. This innovative methodology leverages a concise four-step reaction sequence starting from nicotinic acid esters, bypassing the cumbersome protection and deprotection stages that typically plague alkaloid synthesis. By integrating highly efficient asymmetric catalytic reduction, this technology delivers optically pure nicotine with exceptional enantiomeric excess, setting a new benchmark for cost-effective and environmentally sustainable manufacturing in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing optical pure nicotine have been fraught with significant economic and operational inefficiencies that hinder large-scale adoption. Early strategies often relied on the resolution of racemic mixtures, a process inherently wasteful as it discards up to half of the synthesized material, thereby doubling raw material costs and complicating waste management. Furthermore, direct asymmetric synthesis routes reported in prior art frequently depended on prohibitively expensive noble metal catalysts like Iridium and Ruthenium in stoichiometric amounts, or required harsh cryogenic conditions using reagents such as n-butyllithium at temperatures as low as -78°C. Other methods necessitated the use of protecting groups, adding multiple synthetic steps for installation and removal, which not only increased the overall production timeline but also introduced additional impurities that were difficult to separate, ultimately compromising the final product quality and yield.
The Novel Approach
In stark contrast, the disclosed technology offers a streamlined, protection-free pathway that drastically simplifies the molecular construction of nicotine. The core innovation lies in the direct condensation of nicotinic acid esters with gamma-butyrolactone, followed by a highly stereoselective hydrogenation step that establishes the crucial chiral center with precision. This route eliminates the need for auxiliary chiral resolving agents and avoids the use of hazardous cryogenic reagents, operating instead under moderate temperatures and pressures. The process achieves high atom economy by ensuring that the majority of atoms from the starting materials are incorporated into the final product, minimizing byproduct formation. This strategic simplification not only enhances the overall yield but also aligns perfectly with green chemistry principles, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations seeking to optimize their manufacturing footprint.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
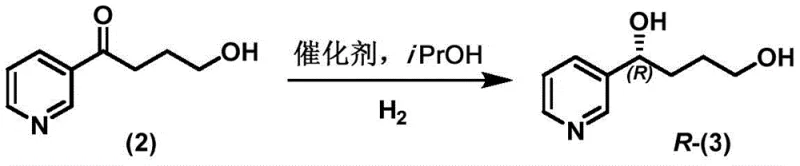
The heart of this synthetic breakthrough is the asymmetric catalytic reduction of the ketone intermediate, a step that dictates the optical purity of the final nicotine molecule. The patent details the use of a sophisticated Iridium-based catalytic system, specifically generated in situ from [Ir(COD)Cl]2 and a chiral polydentate ligand known as f-phamidol (L9). This catalyst architecture creates a highly defined chiral environment around the metal center, allowing for exceptional stereocontrol during the hydrogen transfer to the prochiral ketone substrate. Mechanistic studies within the patent indicate that this system operates with remarkable turnover numbers (TON) reaching up to 200,000, demonstrating that the catalyst remains active and selective even at extremely low loadings. The interaction between the ligand's specific steric bulk and the substrate ensures that hydrogen addition occurs exclusively from one face of the carbonyl group, effectively suppressing the formation of the unwanted enantiomer.
Furthermore, the robustness of this catalytic cycle contributes significantly to impurity control, a critical parameter for R&D directors overseeing quality assurance. Unlike earlier methods where optical purity degraded during subsequent cyclization steps, this process maintains high enantiomeric integrity throughout the synthesis. The use of isopropanol as a solvent not only facilitates the reaction kinetics but also acts as a green medium that simplifies downstream processing. The ability to tune the chirality of the final product simply by switching the enantiomer of the ligand provides a flexible platform for producing both (S)- and (R)-nicotine derivatives, catering to diverse research and development needs without altering the core process infrastructure. This level of mechanistic precision ensures that the resulting chiral alcohol intermediate possesses the necessary fidelity for the subsequent cyclization into high-purity nicotine.
How to Synthesize Optical Pure Nicotine Efficiently
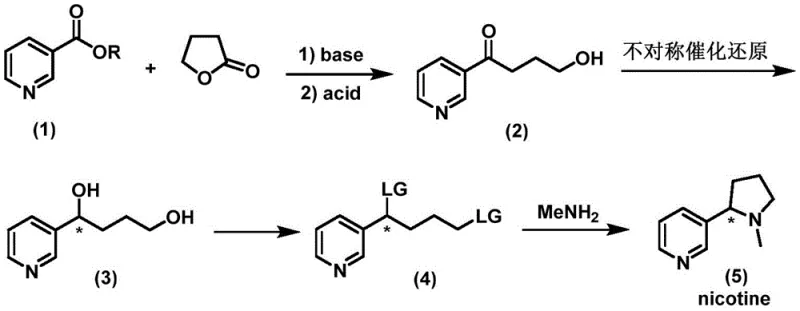
Implementing this advanced synthesis route requires careful attention to reaction parameters, particularly during the catalytic hydrogenation and cyclization phases. The process begins with the base-mediated condensation of ethyl nicotinate and gamma-butyrolactone, followed by acid hydrolysis to generate the key ketone precursor. The subsequent asymmetric reduction is performed under hydrogen pressure using the optimized Iridium/f-phamidol catalyst system in isopropanol, yielding the chiral diol with superior enantioselectivity. Following activation of the hydroxyl group to a leaving group such as a mesylate, the final ring closure is achieved through nucleophilic substitution with methylamine. For laboratory and pilot-scale execution, strict adherence to the specified molar ratios and temperature controls is essential to replicate the high yields and purity reported in the patent documentation. The detailed standardized synthesis steps see the guide below.
- Condense nicotinic acid ester with gamma-butyrolactone using a base, followed by acid hydrolysis to form the ketone intermediate.
- Perform asymmetric catalytic reduction on the ketone intermediate using an Iridium/f-phamidol catalyst system under hydrogen pressure to obtain the chiral diol.
- Activate the chiral diol by converting the hydroxyl group into a leaving group such as mesylate or tosylate.
- React the activated intermediate with methylamine to induce cyclization and form the final optically pure nicotine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this protection-free synthesis methodology represents a strategic opportunity to enhance supply security and reduce total cost of ownership. By eliminating the need for expensive protecting group reagents and the associated unit operations for their installation and removal, the process inherently reduces the consumption of raw materials and solvents. This streamlining of the synthetic sequence translates directly into lower manufacturing overheads and a reduced environmental footprint, as fewer chemical wastes are generated per kilogram of product. The reliance on commercially available starting materials like nicotinic esters and gamma-butyrolactone ensures a stable supply chain that is not subject to the geopolitical or agricultural fluctuations that affect plant-extracted nicotine. Additionally, the high efficiency of the catalyst system means that precious metal usage is minimized, further insulating the production cost from volatility in the noble metal markets.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection steps significantly shortens the production cycle, leading to substantial savings in labor, energy, and equipment utilization time. By avoiding the use of cryogenic reagents and harsh conditions, the process reduces the need for specialized low-temperature infrastructure, allowing for production in standard reactor setups. The high atom economy ensures that raw material costs are optimized, as a greater proportion of the input mass is converted into valuable product rather than waste byproducts. Furthermore, the ability to recover and recycle the green solvent isopropanol adds another layer of economic efficiency to the overall operation.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals as starting materials mitigates the risk of supply disruptions often associated with specialized chiral pools or agricultural extracts. The robust nature of the catalytic system allows for consistent batch-to-batch reproducibility, which is critical for maintaining long-term supply contracts with pharmaceutical partners. The process scalability has been demonstrated from gram to hectogram scales with maintained performance, indicating a clear path to multi-ton commercial production without significant re-engineering. This reliability ensures that downstream customers can depend on a continuous flow of high-quality intermediates for their own manufacturing schedules.
- Scalability and Environmental Compliance: The use of isopropanol and the absence of halogenated solvents in key steps align with increasingly stringent environmental regulations regarding volatile organic compound emissions. The reduction in three-waste discharge simplifies the permitting process for new manufacturing facilities and lowers the costs associated with waste treatment and disposal. The mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint for the manufactured goods. This environmental compatibility makes the process attractive for companies aiming to meet sustainability goals and corporate social responsibility targets in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this route for their specific production requirements. The answers reflect the balance between high-performance chemistry and practical industrial application.
Q: What are the advantages of this protection-free nicotine synthesis method?
A: This method eliminates the need for expensive protecting groups and harsh reagents like n-butyllithium, significantly reducing production costs and waste generation while simplifying the purification process.
Q: Which catalyst system provides the highest enantioselectivity for this process?
A: The Iridium complex formed in situ with the chiral ligand f-phamidol (L9) demonstrates superior performance, achieving over 99% ee and high conversion rates even at low catalyst loadings.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process utilizes readily available starting materials, green solvents like isopropanol, and operates under mild conditions with high atom economy, making it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nicotine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this protection-free asymmetric synthesis technology for the global pharmaceutical and agrochemical markets. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chiral molecules like nicotine can be manufactured with the highest standards of quality and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise measurement of enantiomeric excess required for sensitive biological applications. We are committed to leveraging our technical expertise to bring this efficient and sustainable manufacturing route to reality for our clients.
We invite you to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this route offers compared to your current supply sources. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with a solid foundation of technical and commercial viability.
