Advanced Metal-Free Synthesis of Quinoxalinone Derivatives for Commercial Pharmaceutical Applications
Advanced Metal-Free Synthesis of Quinoxalinone Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable pathways for constructing nitrogen-containing heterocyclic scaffolds. Patent CN113527218A introduces a groundbreaking methodology for the synthesis of quinoxalinone derivatives, a core structure prevalent in bioactive molecules ranging from cystic fibrosis transmembrane regulator activators to antitumor agents. This technology represents a significant paradigm shift from traditional transition-metal catalyzed cross-coupling reactions, offering a metal-free alternative that utilizes readily available 2(1H)-quinoxalinone and aromatic amines. By leveraging a green solvent system composed of dimethyl sulfoxide and water, this process not only aligns with stringent global environmental regulations but also simplifies the downstream purification workflow. For R&D directors and procurement specialists, this patent data signals a viable route to high-purity intermediates with reduced supply chain complexity and enhanced cost efficiency, positioning it as a critical asset for modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the arylation modification at the 3-position of the quinoxalinone skeleton has relied heavily on the use of noble metal catalysts such as palladium, copper, or rhodium complexes. These conventional methodologies often necessitate harsh reaction conditions, including high temperatures and the presence of specialized ligands, which drive up the operational expenditure significantly. Furthermore, the reliance on toxic organic solvents and the generation of heavy metal waste streams create substantial environmental liabilities and complicate the regulatory approval process for pharmaceutical intermediates. The removal of trace metal residues to meet ppm-level specifications requires additional purification steps, such as scavenger treatments or extensive chromatography, which inevitably erodes overall yield and extends production lead times. Consequently, traditional routes often struggle to meet the dual demands of atom economy and commercial viability required by today's competitive fine chemical market.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent data utilizes a transition-metal-free strategy that activates the C-H bond directly under thermal conditions. By employing a binary solvent system of dimethyl sulfoxide and water, the reaction achieves excellent solubility for both organic substrates and inorganic byproducts, facilitating a homogeneous reaction environment that enhances selectivity. This method eliminates the need for expensive catalysts and additives, thereby drastically reducing the raw material cost profile and simplifying the reaction setup. The mild reaction conditions, typically ranging from 60°C to 120°C, ensure energy efficiency while maintaining high conversion rates. This technological advancement allows manufacturers to bypass the complex waste management protocols associated with heavy metals, offering a cleaner, safer, and more economically attractive pathway for the commercial scale-up of complex pharmaceutical intermediates.
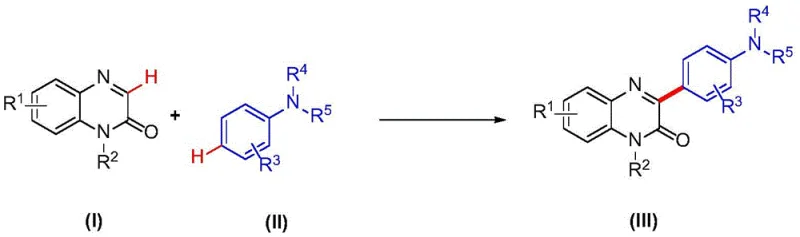
Mechanistic Insights into Metal-Free C-H Arylation
The core of this synthetic innovation lies in the unique reactivity of the 2(1H)-quinoxalinone scaffold under oxidative thermal conditions. The mechanism likely involves the generation of a radical species or a nucleophilic attack facilitated by the polar aprotic nature of dimethyl sulfoxide, which stabilizes the transition state without the need for external metal coordination. The presence of water in the solvent mixture plays a crucial role in proton transfer processes and may assist in the activation of the aromatic amine nucleophile. This synergistic effect between the solvent components allows for the direct functionalization of the C3 position with high regioselectivity, avoiding the formation of unwanted isomers that often plague traditional electrophilic substitution reactions. Understanding this mechanistic nuance is vital for R&D teams aiming to optimize the reaction for specific substrates, as it highlights the importance of solvent polarity and temperature control in driving the reaction to completion.
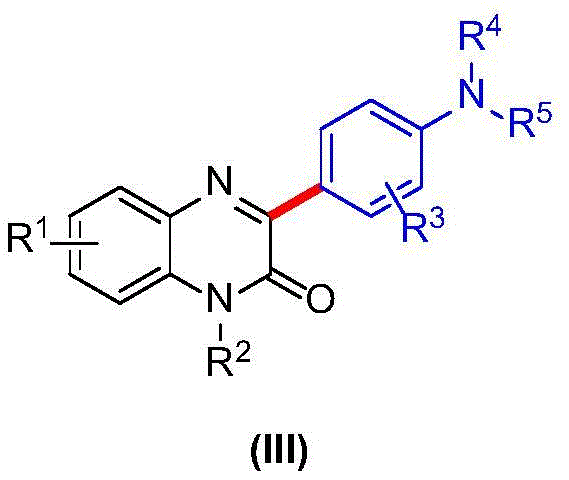
Furthermore, the tolerance of this system towards various functional groups on the aromatic amine ring demonstrates its versatility for library synthesis. The patent data indicates that electron-donating and electron-withdrawing groups are well-tolerated, allowing for the introduction of diverse pharmacophores essential for structure-activity relationship (SAR) studies. The impurity profile is notably clean, primarily consisting of unreacted starting materials which are easily separated during the aqueous workup phase. This high level of chemoselectivity ensures that the final product meets the stringent purity specifications required for clinical trial materials. For process chemists, this means fewer purification cycles and a more predictable manufacturing process, reducing the risk of batch failure and ensuring consistent quality across large-scale production runs.
How to Synthesize Quinoxalinone Derivative Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and thermal profiles as outlined in the patent examples. The process begins with the charging of 2(1H)-quinoxalinone and the selected aromatic amine into a reactor, followed by the addition of the DMSO-water solvent mixture. Maintaining the reaction temperature between 80°C and 120°C for a duration of 8 to 16 hours is critical to achieving optimal yields, as demonstrated by the experimental data where yields exceeded 80% under optimized conditions. The detailed standardized synthesis steps, including specific workup procedures and purification parameters, are provided in the technical guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the reaction mixture by combining 2(1H)-quinoxalinone and aromatic amine in a molar ratio of 1: 2 within a reaction vessel.
- Add a mixed solvent system of dimethyl sulfoxide and water (ratio 1: 2) and heat the mixture to 100°C for 8 to 10 hours with stirring.
- Upon completion, cool the reaction, extract with ethyl acetate, dry the organic layer, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this metal-free synthesis route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The elimination of noble metal catalysts removes a significant variable cost component and mitigates the risk of supply disruptions associated with precious metal sourcing. Additionally, the use of water as a co-solvent reduces the volume of hazardous organic waste, leading to substantial cost savings in waste disposal and environmental compliance. The simplicity of the workup procedure, involving standard extraction and drying, allows for faster batch turnover times, thereby enhancing overall production capacity and responsiveness to market demand fluctuations without compromising product quality.
- Cost Reduction in Manufacturing: The absence of expensive palladium or copper catalysts significantly lowers the bill of materials for each production batch. Furthermore, the simplified purification process reduces the consumption of silica gel and elution solvents, which are major cost drivers in intermediate manufacturing. By avoiding the need for specialized metal scavengers, the process also reduces the operational complexity and labor costs associated with quality control testing for residual metals. These cumulative efficiencies translate into a more competitive pricing structure for the final quinoxalinone derivatives, enabling better margin management for downstream drug manufacturers.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically 2(1H)-quinoxalinone and various aromatic amines, are commodity chemicals with robust global supply chains. This reduces the dependency on single-source suppliers for specialized catalysts or ligands, thereby minimizing the risk of production delays due to raw material shortages. The stability of the reaction conditions also allows for flexible scheduling and inventory management, as the process is less sensitive to minor variations in environmental conditions. This reliability ensures a continuous flow of high-quality intermediates, supporting just-in-time manufacturing models and long-term supply agreements.
- Scalability and Environmental Compliance: The green nature of this solvent system aligns perfectly with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions and heavy metal discharge. Scaling this process from laboratory to pilot and commercial plant levels involves minimal engineering changes, as the thermal and mixing requirements are standard for existing chemical infrastructure. The reduced environmental footprint not only lowers regulatory compliance costs but also enhances the corporate sustainability profile of the manufacturing entity. This makes the technology particularly attractive for companies aiming to meet ESG (Environmental, Social, and Governance) targets while maintaining high production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinoxalinone synthesis technology. These answers are derived directly from the patent specifications and experimental data to provide accurate guidance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production lines or new drug development projects.
Q: Does this synthesis method require expensive noble metal catalysts?
A: No, the patented method described in CN113527218A operates under metal-free conditions, utilizing thermal activation in a DMSO-water solvent system, which significantly reduces raw material costs and eliminates heavy metal contamination risks.
Q: What is the typical yield range for this quinoxalinone derivative synthesis?
A: Experimental data from the patent indicates yields ranging from 56% to 84%, depending on the specific aromatic amine substrate and reaction temperature optimization, demonstrating robust efficiency for industrial scaling.
Q: Is the purification process scalable for commercial production?
A: Yes, the workup involves standard aqueous dilution and ethyl acetate extraction followed by column chromatography, which can be adapted to continuous chromatography or recrystallization processes for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxalinone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of this metal-free synthesis technology in accelerating the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of quinoxalinone derivative delivered meets the highest industry standards for safety and efficacy. We are committed to providing a seamless supply chain experience that supports your critical timelines.
We invite you to collaborate with our technical procurement team to explore how this innovative route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of adopting this green synthesis method for your pipeline. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a data-driven approach to your supply chain strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
