Advanced Manufacturing of Palbociclib Intermediates via Low-Cost Palladium Catalysis
Introduction to Next-Generation Palbociclib Intermediate Manufacturing
The global demand for CDK4/6 inhibitors, particularly Palbociclib, has necessitated the development of more efficient and sustainable synthetic routes for its key intermediates. Patent CN112898299A discloses a groundbreaking preparation method for 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one, a critical building block in oncology drug synthesis. This technical insight report analyzes the proprietary methodology which overcomes significant bottlenecks in traditional manufacturing, specifically addressing the high cost of noble metal catalysts and the complexity of waste stream management. By shifting from homogeneous amine bases to inorganic systems and drastically reducing palladium loading, this route offers a compelling value proposition for pharmaceutical supply chains seeking reliability and cost-efficiency.
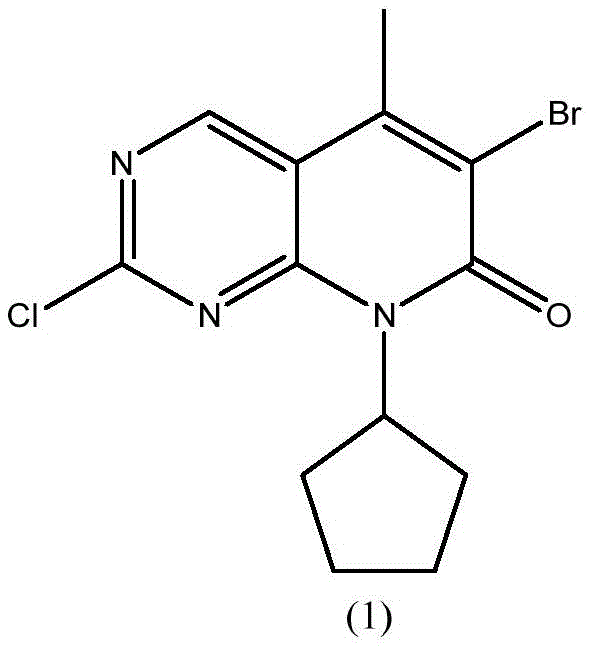
For procurement leaders and R&D directors, the transition to this optimized pathway represents a strategic opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials at a reduced cost basis. The structural integrity of the final product, as depicted in the associated chemical diagrams, is maintained through rigorous control of reaction parameters, ensuring that the intermediate meets the stringent specifications required for downstream API synthesis. This report delves into the mechanistic advantages and commercial implications of adopting this novel technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this pyrido-pyrimidinone scaffold has relied on processes described in earlier literature, such as CN105008357A. These conventional routes typically employ ethanol as a solvent and triethylamine as an acid-binding agent during the initial amination step. While chemically feasible, this approach generates wastewater laden with triethylamine hydrochloride and ethanol, creating a complex effluent profile that is difficult and costly to treat. Furthermore, the subsequent Heck cyclization step in traditional methods requires a relatively high loading of palladium acetate, often around 0.03 equivalents. Given the volatility and high market price of palladium, this stoichiometric requirement constitutes a significant portion of the total manufacturing cost, reportedly accounting for 60-70% of the expense in some legacy processes.
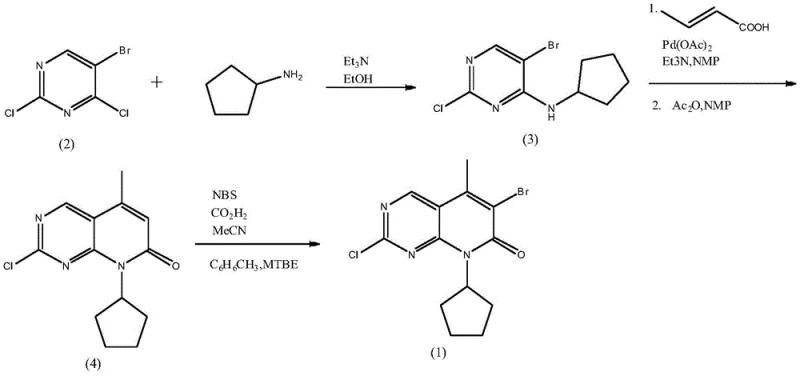
The Novel Approach
The innovative methodology presented in CN112898299A fundamentally re-engineers the synthesis to address these economic and environmental pain points. By substituting the organic solvent/amine system with a biphasic dichloromethane and water mixture utilizing inorganic bases like sodium carbonate or potassium hydroxide, the process generates simple inorganic salt wastewater which is far easier to manage. More critically, the novel approach introduces tetrabutylammonium bromide (TBAB) as a phase transfer catalyst alongside trace amounts of water in the Heck reaction. This modification enables the dramatic reduction of the palladium catalyst loading to between 0.002 and 0.005 equivalents. This represents a roughly tenfold decrease in precious metal consumption compared to the prior art, directly translating to substantial cost reduction in pharmaceutical intermediates manufacturing without compromising yield or purity.
Mechanistic Insights into Trace-Palladium Catalyzed Cyclization
The core innovation of this synthesis lies in the optimization of the Heck coupling and subsequent cyclization steps. In the traditional mechanism, high catalyst loading is often required to drive the reaction to completion due to catalyst deactivation or insufficient activation of the aryl halide. However, the inclusion of TBAB facilitates the transport of ionic species between the organic and aqueous phases, enhancing the availability of the base and stabilizing the catalytic cycle. The presence of trace water is also identified as a promoter, likely assisting in the solubilization of inorganic components and maintaining the active state of the palladium species. This synergistic effect allows the reaction to proceed efficiently at 80-85°C with minimal catalyst input.
Following the coupling with crotonic acid, the intermediate undergoes dehydration and cyclization upon the addition of acetic anhydride. The mechanistic pathway ensures the formation of the rigid pyrido[2,3-d]pyrimidin-7(8H)-one core with high regioselectivity. The subsequent bromination step utilizes N-bromosuccinimide (NBS) in acetonitrile with oxalic acid, a condition that effectively installs the bromine atom at the 6-position while minimizing poly-bromination side products. This precise control over the impurity profile is crucial for R&D teams focused on commercial scale-up of complex pharmaceutical intermediates, as it simplifies downstream purification and ensures consistent quality.
How to Synthesize 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one Efficiently
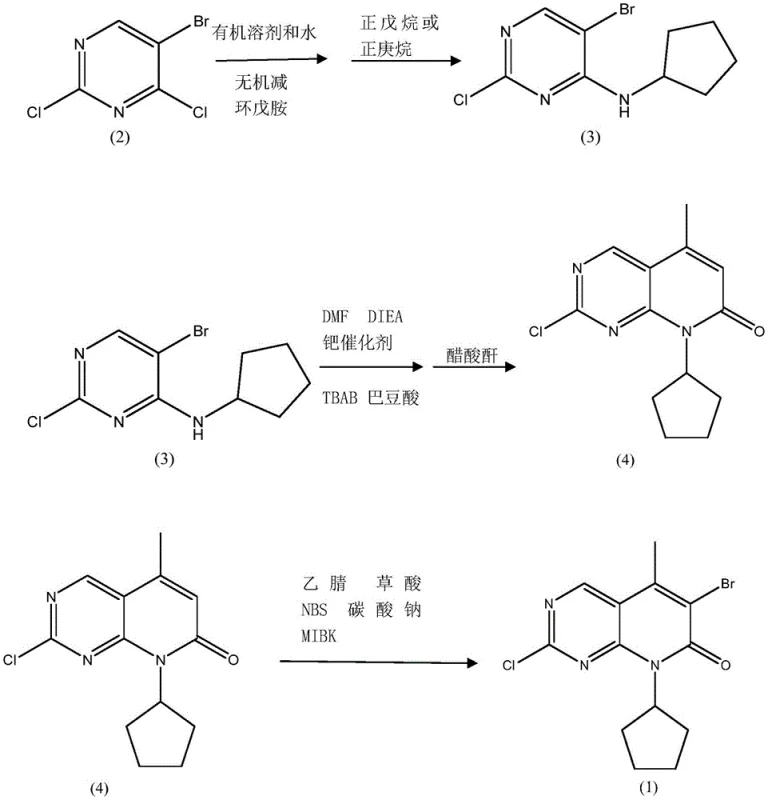
The execution of this synthesis requires careful attention to the biphasic conditions and the sequential addition of reagents to maximize the efficiency of the trace palladium system. The process is divided into three distinct operational stages: the initial selective amination, the catalytic cyclization, and the final functionalization. Each step has been optimized to allow for solvent recovery and recycling, further enhancing the green chemistry profile of the route. For detailed operational parameters, temperature controls, and workup procedures, please refer to the standardized synthesis guide below.
- React 5-bromo-2,4-dichloropyrimidine with cyclopentylamine in a dichloromethane/water biphasic system using an inorganic base to form the amino-pyrimidine intermediate.
- Perform a Heck coupling with crotonic acid using trace palladium (0.002-0.005 eq), TBAB, and DIEA in DMF, followed by acetic anhydride cyclization.
- Brominate the cyclized ketone using NBS and oxalic acid in acetonitrile, followed by purification via MIBK recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented methodology offers transformative benefits for supply chain stability and cost management. The primary driver of value is the drastic reduction in raw material costs associated with the palladium catalyst. By lowering the catalyst loading by an order of magnitude, manufacturers can insulate their production costs from the volatility of the precious metals market. Additionally, the ability to recover and reuse solvents like dichloromethane and bases like DIEA (with reported recovery rates exceeding 80%) creates a closed-loop system that minimizes waste disposal fees and raw material procurement needs.
- Cost Reduction in Manufacturing: The elimination of expensive triethylamine salts and the reduction of palladium usage from 0.03 equivalents to trace levels (0.002-0.005 eq) fundamentally alters the cost structure. The patent explicitly notes a reduction in manufacturing costs from approximately 13,000 CNY/kg to the 2,000-2,500 CNY/kg range, driven largely by the catalyst savings. This efficiency allows for competitive pricing strategies in the global market for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The use of readily available inorganic bases and common solvents like DCM and acetonitrile reduces dependency on specialized reagents. The robustness of the reaction conditions, which tolerate trace water and utilize stable phase transfer catalysts, ensures consistent batch-to-batch performance. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates and maintaining continuous supply to API manufacturers.
- Scalability and Environmental Compliance: The generation of inorganic salt wastewater instead of complex organic amine waste significantly lowers the burden on effluent treatment plants. This environmental advantage facilitates easier regulatory approval for large-scale production facilities. The process is designed for industrial scalability, with demonstrated success in multi-kilogram batches, ensuring that the reliable pharmaceutical intermediates supplier can meet surging market demands without ecological compromise.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of the technology.
Q: How does the new process reduce palladium consumption compared to prior art?
A: The patented method reduces palladium catalyst loading from 0.03 equivalents in conventional routes to a trace range of 0.002-0.005 equivalents by utilizing TBAB as a phase transfer catalyst and optimizing water content.
Q: What are the environmental advantages of the solvent system used?
A: The process replaces ethanol and triethylamine with a dichloromethane/water system and inorganic bases, generating inorganic salt wastewater instead of complex amine salts, significantly simplifying effluent treatment.
Q: Is the diisopropylethylamine (DIEA) recoverable in this synthesis?
A: Yes, the mother liquor from the cyclization step allows for the recovery of DIEA with high purity (up to 99.3%) and a recovery rate exceeding 80%, enabling solvent reuse and cost reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role this intermediate plays in the synthesis of life-saving oncology therapies. Our technical team has extensively analyzed the pathway described in CN112898299A and possesses the expertise to implement this low-cost, high-efficiency route at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest standards required for GMP API synthesis.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our adoption of this optimized synthesis can enhance your project's economic viability and timeline.
