Advanced Manufacturing of Palbociclib Intermediates: A Low-Cost, High-Purity Route for Global Supply Chains
Advanced Manufacturing of Palbociclib Intermediates: A Low-Cost, High-Purity Route for Global Supply Chains
The global demand for CDK4/6 inhibitors, particularly Palbociclib, has necessitated the development of robust, scalable, and cost-effective synthetic routes for its key intermediates. Patent CN112898299B discloses a groundbreaking preparation method for 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one, a critical building block in the synthesis of this life-saving breast cancer medication. This technical insight report analyzes the novel methodology which overcomes significant limitations in prior art, specifically addressing the high cost of precious metal catalysts and the complexity of wastewater treatment. By shifting from expensive organic amine acid-binding agents to inorganic bases and utilizing a trace palladium catalytic system enhanced by phase transfer catalysts, this innovation offers a compelling value proposition for pharmaceutical manufacturers seeking to optimize their supply chain resilience and reduce the total cost of ownership for API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this pivotal pyrido-pyrimidinone scaffold has been hindered by inefficient catalytic systems and environmentally burdensome solvent choices. As illustrated in the prior art route, conventional methods typically rely on triethylamine as an acid-binding agent in ethanol, which generates wastewater laden with triethylamine hydrochloride and ethanol mixtures that are notoriously difficult and costly to treat. Furthermore, the cyclization step traditionally requires a substantial loading of palladium acetate, often around 0.03 equivalents, to drive the Heck reaction with crotonic acid. This high catalyst loading not only inflates the raw material costs due to the volatility of palladium prices but also introduces significant challenges in removing residual heavy metals to meet stringent pharmaceutical purity standards. The accumulation of these inefficiencies creates a bottleneck for commercial scale-up, making the final API prohibitively expensive for broader market access.
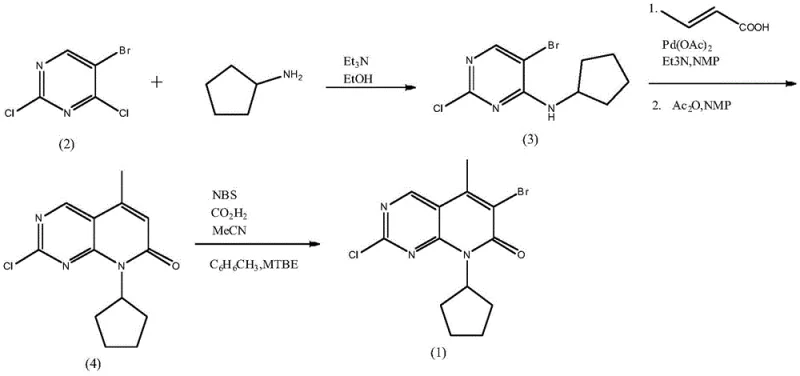
The Novel Approach
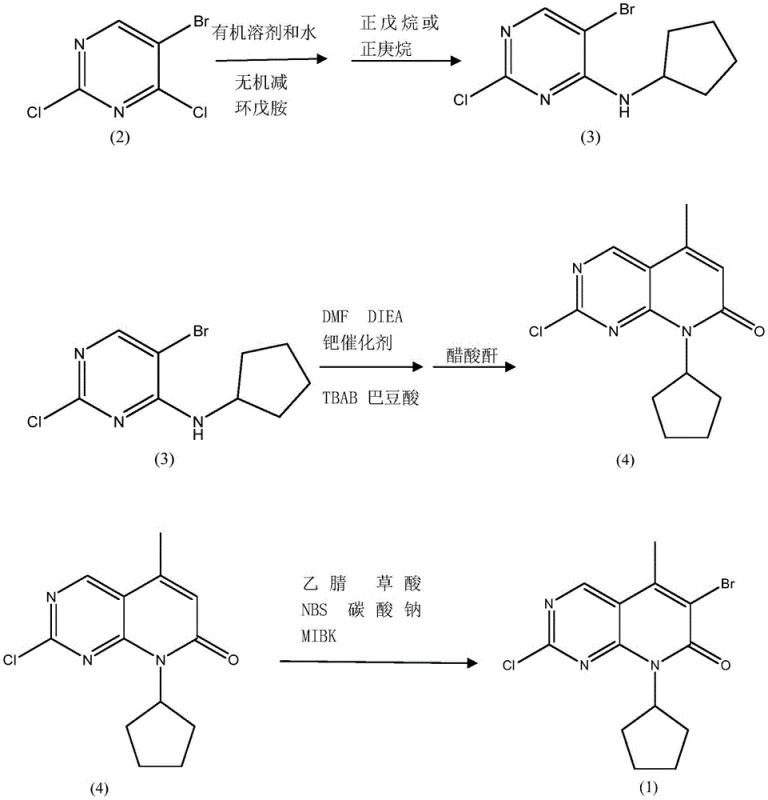
The innovative process detailed in CN112898299B fundamentally re-engineers the synthetic pathway to maximize efficiency and minimize environmental impact. Instead of the traditional organic base system, the new method employs a biphasic solvent system of dichloromethane and water with an inorganic base for the initial amination step, drastically simplifying downstream purification. The most significant breakthrough occurs during the cyclization phase, where the introduction of tetrabutylammonium bromide (TBAB) as a phase transfer catalyst and the controlled addition of trace water allows for a dramatic reduction in palladium usage. This optimized protocol enables the reaction to proceed smoothly with only 0.002 to 0.005 equivalents of palladium catalyst, representing a tenfold reduction compared to legacy methods. The result is a streamlined process that delivers high-purity intermediates while simultaneously lowering the barrier to industrial adoption through reduced operational complexity.
Mechanistic Insights into Trace Palladium-Catalyzed Cyclization
The core of this technological advancement lies in the synergistic interaction between the trace palladium catalyst and the phase transfer agent TBAB within the reaction matrix. In standard Heck coupling reactions, high catalyst loading is often required to overcome kinetic barriers and maintain turnover frequency, especially in heterogeneous or viscous media. However, the presence of TBAB facilitates the transport of ionic species across phase boundaries, effectively increasing the local concentration of reactive intermediates near the catalytic center. Additionally, the patent highlights the counter-intuitive yet critical role of trace water; rather than inhibiting the reaction, minute amounts of water appear to stabilize the active palladium species or facilitate the proton abstraction steps necessary for the catalytic cycle to turn over efficiently. This mechanistic nuance allows the reaction to achieve high conversion rates at 80-85°C without the need for excessive catalyst loading, thereby preserving the economic viability of the process.
Furthermore, the impurity profile of the final product is significantly improved through this refined mechanism. By minimizing the amount of palladium introduced into the system, the burden on downstream metal scavenging processes is greatly reduced, which directly correlates to higher overall yields and purity. The subsequent dehydration step using acetic anhydride is also optimized to occur in situ, preventing the isolation of unstable intermediates that could degrade or form side products. The final bromination step utilizes N-bromosuccinimide (NBS) promoted by oxalic acid in acetonitrile, a condition that ensures regioselective substitution at the 6-position while maintaining the integrity of the sensitive pyrimidine ring. This precise control over reaction selectivity is paramount for ensuring that the intermediate meets the rigorous quality specifications required for GMP manufacturing of oncology drugs.
How to Synthesize 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters, particularly temperature control during the exothermic amination and the precise stoichiometry of the phase transfer catalyst. The process is designed to be robust, tolerating slight variations in mixing while delivering consistent results, which is essential for technology transfer from laboratory to pilot plant scales. Operators should note the importance of the solvent recovery loops, particularly for DIEA and dichloromethane, as these contribute significantly to the green chemistry metrics of the overall process. For a detailed breakdown of the specific operational parameters, reagent grades, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Perform selective amination of 5-bromo-2,4-dichloropyrimidine with cyclopentylamine using an inorganic base in a biphasic solvent system to obtain the amino-pyrimidine precursor.
- Execute a Heck-type cyclization with crotonic acid using trace palladium catalyst and TBAB phase transfer catalyst, followed by dehydration with acetic anhydride to form the pyridone core.
- Conduct regioselective bromination of the pyridone intermediate using NBS in acetonitrile with oxalic acid promotion, followed by purification via MIBK recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic advantages beyond simple unit cost savings. The shift away from expensive palladium catalysts and difficult-to-treat organic amine wastes fundamentally alters the cost structure of the intermediate, making the supply chain more resilient to fluctuations in precious metal markets. By decoupling production economics from the volatile pricing of palladium, manufacturers can offer more stable long-term pricing contracts to their API customers. Additionally, the simplified wastewater profile reduces the operational expenditure associated with environmental compliance and effluent treatment, further enhancing the margin potential for large-volume production campaigns.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the drastic reduction in palladium catalyst loading. Since palladium is a high-value precious metal, reducing the requirement by an order of magnitude directly lowers the Bill of Materials (BOM) cost. Furthermore, the ability to recover and reuse solvents like DIEA and dichloromethane with high efficiency minimizes raw material consumption. This dual approach of reducing catalyst input and maximizing solvent recovery creates a leaner manufacturing process that is less sensitive to raw material price spikes, ensuring sustainable cost competitiveness in the global marketplace.
- Enhanced Supply Chain Reliability: The use of readily available inorganic bases and common organic solvents mitigates the risk of supply disruptions often associated with specialized reagents. The robustness of the reaction conditions, which operate at moderate temperatures and pressures, ensures that the process can be scaled up reliably across different manufacturing sites without requiring exotic equipment. This flexibility allows for diversified sourcing strategies, enabling pharmaceutical companies to qualify multiple suppliers who adopt this efficient technology, thereby securing the continuity of supply for critical oncology medications.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the replacement of triethylamine with inorganic bases significantly simplifies waste management. The resulting wastewater contains primarily inorganic salts, which are easier to neutralize and treat compared to complex organic amine salts. This reduction in hazardous waste volume not only lowers disposal costs but also aligns with increasingly stringent global environmental regulations. The process is inherently safer and cleaner, facilitating faster regulatory approvals for new manufacturing facilities and supporting the industry's move towards greener, more sustainable chemical production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms traditional approaches in terms of yield, purity, and operational efficiency. Understanding these nuances is critical for R&D teams evaluating technology transfer and for procurement teams assessing the long-term viability of this supply source.
Q: How does the new process reduce palladium catalyst costs compared to prior art?
A: The patented method reduces palladium loading from 0.03 equivalents in conventional routes to merely 0.002-0.005 equivalents. This drastic reduction, facilitated by the addition of TBAB and trace water, significantly lowers the raw material cost associated with precious metal catalysts.
Q: What are the environmental advantages regarding wastewater treatment?
A: Unlike traditional methods using triethylamine which generate complex organic salt wastewater, this process utilizes inorganic bases. This results in wastewater containing primarily inorganic salts, which are far easier and cheaper to treat, reducing the environmental burden and compliance costs.
Q: Is the solvent system suitable for large-scale industrial recovery?
A: Yes, the process employs solvents like dichloromethane and DIEA which are efficiently recovered. Specifically, DIEA can be recovered with high purity (over 99%) via pH adjustment and distillation, allowing for direct reuse in subsequent batches and minimizing solvent consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-bromo-2-chloro-8-cyclopentyl-5-methylpyrido[2,3-d]pyrimidin-7(8H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology therapies depends on the availability of high-quality intermediates produced via sustainable and scalable routes. Our technical team has extensively analyzed the methodology described in CN112898299B and possesses the expertise to implement this trace palladium catalysis strategy effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale optimization to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the exacting standards required for GMP API synthesis.
We invite global pharmaceutical partners to engage with us to explore how this advanced synthesis route can optimize your supply chain. By leveraging our technical capabilities, you can achieve significant efficiencies in your manufacturing operations while maintaining the highest standards of product quality. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our adoption of this innovative technology can drive value for your organization.
