Advanced Synthesis of Substituted Benzoic Acids for Pharmaceutical and Agrochemical Applications
Introduction to Advanced Benzoic Acid Synthesis
The chemical industry constantly seeks more efficient pathways to access functionalized aromatic building blocks, particularly substituted benzoic acids which serve as critical scaffolds in pharmaceutical and agrochemical manufacturing. Patent CN1095823C introduces a robust and versatile methodology for synthesizing optionally substituted hydroxybenzoic, alkanoyloxybenzoic, formyloxybenzoic, and alkoxybenzoic acids starting from readily available 1-substituted 2,6-dihalobenzenes. This technology addresses long-standing challenges in regioselectivity and process safety by leveraging a combination of copper-catalyzed nucleophilic substitution and controlled Grignard carboxylation. Unlike conventional routes that rely on hazardous diazotization reactions or multi-step functional group interconversions, this approach offers a streamlined path to high-purity intermediates. The ability to selectively manipulate one halogen atom while preserving the other for subsequent metallation represents a significant leap forward in synthetic efficiency, providing a reliable foundation for the commercial scale-up of complex aromatic acids required in modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of meta-substituted benzoic acids, such as 3-methoxy-2-methylbenzoic acid, has relied heavily on diazotization of corresponding anilines followed by hydrolysis, a process fraught with operational hazards and environmental concerns. These traditional methods often necessitate the handling of unstable diazonium salts and generate substantial quantities of inorganic salt waste, complicating wastewater treatment and increasing the overall carbon footprint of the manufacturing process. Furthermore, alternative routes involving the hydrolysis of nitriles or oxidation of methyl groups frequently suffer from poor regiocontrol, leading to difficult-to-separate isomeric mixtures that compromise the purity profile essential for pharmaceutical applications. The reliance on stoichiometric oxidants or harsh acidic conditions also limits the substrate scope, making it challenging to introduce sensitive functional groups without extensive protection and deprotection strategies. Consequently, procurement teams often face volatile pricing and supply chain disruptions due to the regulatory pressures surrounding these older, less sustainable chemistries.
The Novel Approach
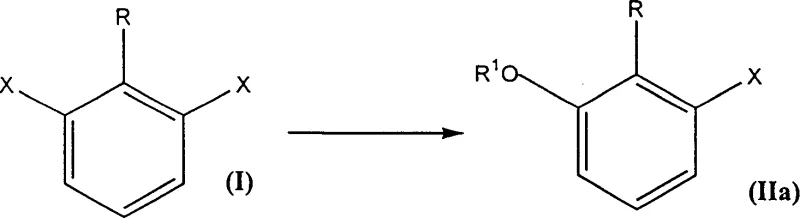
The methodology described in CN1095823C circumvents these bottlenecks by employing a strategic sequence of selective nucleophilic substitution followed by Grignard carboxylation. By utilizing 1-substituted 2,6-dihalobenzenes as starting materials, the process exploits the differential reactivity of the halogen positions, allowing for the precise installation of alkoxy or aryloxy groups using copper catalysis. This selectivity is paramount, as it ensures that the remaining halogen is available for the subsequent formation of the Grignard reagent without interference from the newly introduced oxygen functionality. The innovation extends to the direct conversion of the Grignard intermediate into either the carboxylic acid via carbon dioxide quenching or directly into the acyl chloride using phosgene. This flexibility allows manufacturers to tailor the output to specific downstream needs, whether it be for coupling reactions requiring acid chlorides or for further functionalization of the carboxylic acid moiety, thereby drastically simplifying the overall synthetic tree and enhancing process robustness.
Mechanistic Insights into Copper-Catalyzed Substitution and Grignard Carboxylation
The core of this synthetic strategy lies in the copper-catalyzed nucleophilic aromatic substitution, which facilitates the displacement of one halogen atom on the dihalobenzene ring with high fidelity. The mechanism likely involves the oxidative addition of the aryl halide to a low-valent copper species, followed by ligand exchange with the alkoxide or phenoxide nucleophile and subsequent reductive elimination to release the mono-substituted product. The choice of catalyst, such as cuprous cyanide or cuprous bromide, along with polar aprotic solvents like DMSO or NMP, is critical for stabilizing the transition state and suppressing bis-substitution side reactions. Experimental data indicates that under optimized conditions, selectivity for the mono-substituted intermediate can exceed 90%, even at high conversion rates, which is a remarkable achievement for nucleophilic aromatic substitutions on electron-rich systems. This high level of control minimizes the formation of diaryl ethers or other poly-substituted impurities, ensuring a cleaner reaction profile that simplifies isolation and purification steps.
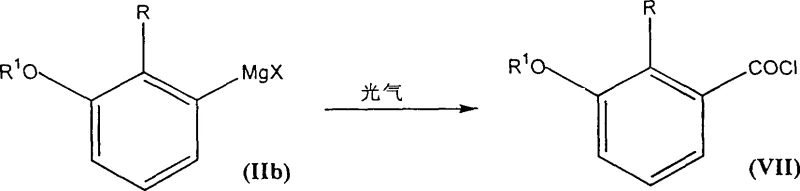
Following the substitution, the formation of the Grignard reagent requires meticulous attention to anhydrous conditions to prevent premature protonation of the organomagnesium species. The presence of water would lead to the formation of dehalogenated byproducts, such as 1,2-disubstituted benzenes, which are difficult to remove and reduce overall yield. The process utilizes initiators like glycol dibromide or ethyl iodide to activate the magnesium surface, ensuring rapid onset of the metallation reaction. Once formed, the Grignard intermediate reacts with carbon dioxide to form a carboxylate salt, which is subsequently hydrolyzed to the free acid. Alternatively, quenching with phosgene generates the acyl chloride directly, bypassing the need for separate chlorinating agents like thionyl chloride. This mechanistic versatility not only improves atom economy but also reduces the generation of corrosive gaseous byproducts typically associated with acid chloride synthesis, aligning with modern green chemistry principles.

How to Synthesize Substituted Benzoic Acids Efficiently
The practical implementation of this synthesis involves a carefully sequenced workflow that begins with the preparation of the mono-substituted aryl halide. Operators must maintain strict temperature control during the nucleophilic substitution phase, typically heating the reaction mixture of dihalobenzene, alkoxide, and copper catalyst in DMSO to temperatures between 140°C and 160°C to drive the reaction to completion while monitoring conversion via gas chromatography. Once the intermediate is isolated or used in situ, the transition to Grignard chemistry demands a switch to anhydrous ether solvents like THF and the rigorous exclusion of moisture. The detailed standardized synthesis steps, including specific molar ratios, addition rates, and workup procedures for maximizing yield and purity, are outlined in the guide below.
- Perform a selective nucleophilic substitution on a 1-substituted 2,6-dihalobenzene using an alkoxide or phenoxide in the presence of a copper catalyst to form a mono-substituted intermediate.
- React the resulting aryl halide intermediate with magnesium metal under strictly anhydrous conditions to generate the corresponding Grignard reagent.
- Quench the Grignard reagent with carbon dioxide followed by hydrolysis to yield the target substituted benzoic acid, or alternatively use phosgene for direct acyl chloride formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic benefits regarding cost stability and supply continuity. By eliminating the need for hazardous diazotization reagents and reducing the number of unit operations required to reach the final acid or acid chloride, the overall manufacturing cost structure is significantly optimized. The high selectivity of the copper-catalyzed step means that raw material consumption is more efficient, as less starting material is lost to poly-substituted byproducts, directly impacting the cost of goods sold. Furthermore, the ability to produce acyl chlorides directly from the Grignard reagent removes an entire processing step involving thionyl chloride or phosphorus pentachloride, which not only saves on reagent costs but also mitigates the logistical challenges associated with handling and disposing of large volumes of corrosive chlorinating agents and their acidic waste streams.
- Cost Reduction in Manufacturing: The streamlined nature of this process inherently lowers production costs by consolidating multiple synthetic transformations into fewer steps. The elimination of the separate acid-to-acid-chloride conversion step, achieved by direct phosgene quenching of the Grignard reagent, removes the capital and operational expenses associated with chlorination reactors and scrubbing systems. Additionally, the high selectivity of the initial substitution reaction reduces the burden on downstream purification units such as distillation columns or crystallizers, leading to lower energy consumption and higher throughput. This efficiency gain allows for a more competitive pricing model for high-purity intermediates, providing a buffer against raw material price fluctuations in the global chemical market.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like dihalobenzenes, magnesium, and carbon dioxide ensures a robust and resilient supply chain that is less susceptible to the bottlenecks often seen with specialized reagents. The process avoids the use of highly regulated precursors that might face export controls or stringent transportation restrictions, thereby smoothing the logistics of raw material inbound delivery. Moreover, the scalability of the Grignard carboxylation step is well-understood in the industry, allowing for seamless technology transfer from pilot plant to multi-ton commercial production without the need for exotic equipment. This reliability ensures consistent delivery schedules for downstream pharmaceutical customers who depend on just-in-time inventory models for their API synthesis.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers a superior profile compared to traditional methods by minimizing the generation of heavy metal waste and acidic effluents. The use of catalytic amounts of copper, which can potentially be recovered or immobilized on supports, aligns with increasingly strict environmental regulations regarding heavy metal discharge in pharmaceutical manufacturing. The direct formation of acyl chlorides also reduces the volume of sulfur or phosphorus-containing waste that typically requires neutralization and specialized disposal. These factors collectively simplify the permitting process for new manufacturing lines and reduce the long-term liability associated with waste management, making the facility more sustainable and compliant with global green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, derived from the specific embodiments and experimental data provided in the patent documentation. Understanding these nuances is critical for process engineers aiming to replicate the high yields and selectivity reported in the intellectual property. The answers reflect the preferred conditions and critical control points identified during the development of this technology.
Q: How does this process improve selectivity compared to traditional diazotization routes?
A: Traditional diazotization routes often require harsh acidic conditions and generate stoichiometric amounts of salt waste. This patent utilizes copper-catalyzed nucleophilic substitution on dihalobenzenes, achieving selectivity rates greater than 90% for the mono-substituted product, significantly reducing downstream purification burdens and improving overall atom economy.
Q: Can this method be adapted for direct acyl chloride production?
A: Yes, a unique embodiment of this invention allows for the direct conversion of the Grignard intermediate into an acyl chloride by quenching with phosgene. This eliminates the separate step of converting the carboxylic acid to the acid chloride using thionyl chloride or phosphorus chlorides, thereby streamlining the synthesis of amide or hydrazide derivatives.
Q: What are the critical control parameters for the Grignard formation step?
A: The formation of the Grignard reagent must be conducted under strictly anhydrous conditions to prevent protonation of the organometallic species, which would lead to dehalogenated byproducts. The process utilizes initiators like glycol dibromide or iodine and maintains reflux temperatures in ether solvents like THF to ensure complete conversion of the aryl halide before carboxylation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medicines and advanced agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1095823C can be translated into a robust industrial reality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our substituted benzoic acids meet the exacting standards required by global regulatory bodies. Our commitment to quality assurance extends beyond mere compliance; we actively collaborate with clients to optimize impurity profiles and ensure batch-to-batch consistency.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis for transitioning from a legacy diazotization process or need immediate access to specific COA data for 3-methoxy-2-methylbenzoic acid, our experts are ready to provide comprehensive route feasibility assessments. By partnering with us, you gain access to a supply chain that prioritizes innovation, sustainability, and reliability, empowering your R&D and manufacturing teams to focus on what they do best: creating value through chemistry.
