Advanced Fluazinam Manufacturing: Leveraging Mixed Solvent Technology for High Purity and Yield
The global demand for high-efficacy fungicides continues to drive innovation in agrochemical manufacturing, with fluazinam standing out as a critical active ingredient for disease control in various crops. Patent CN103626695A introduces a transformative approach to the synthesis of 3-chloro-N-(3-chloro-5-trifluoromethyl-2-pyridyl)-alpha,alpha,alpha-trifluoro-2,6-dinitro-p-toluidine, commonly known as fluazinam. This intellectual property details a sophisticated method utilizing a mixed solvent system composed of alkyl-substituted aromatic hydrocarbons and methyl isobutyl ketone (MIBK) to overcome longstanding challenges in yield and purity. By strategically engineering the reaction medium, this technology effectively inhibits side reactions that typically plague traditional synthesis routes, resulting in a final product with purity exceeding 99%. For R&D directors and procurement specialists alike, this represents a significant leap forward in process chemistry, offering a pathway to more reliable and cost-effective production of this vital agrochemical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
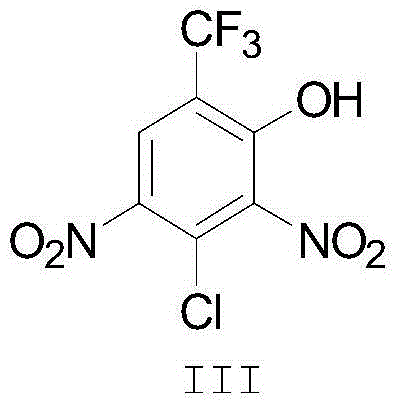
Historically, the synthesis of fluazinam has been fraught with inefficiencies when relying on conventional single-solvent systems. Early iterations of this process utilized dimethylformamide (DMF), which unfortunately yielded poor results with conversion rates hovering around only 22%, rendering it economically unviable for large-scale manufacturing. Subsequent attempts shifted towards tetrahydrofuran (THF), which improved yields to approximately 75% due to better hydrophobicity; however, THF introduced severe safety liabilities regarding flammability and the formation of explosive peroxides, complicating its handling in industrial settings. Even the adoption of methyl isobutyl ketone (MIBK) as a standalone solvent, while an improvement, still faced limitations regarding the suppression of hydrolysis byproducts and the efficiency of solvent recovery. In alkaline conditions, the dinitro intermediate is susceptible to hydrolysis, leading to the formation of unwanted phenolic byproducts that degrade overall yield and complicate downstream purification efforts.
The Novel Approach
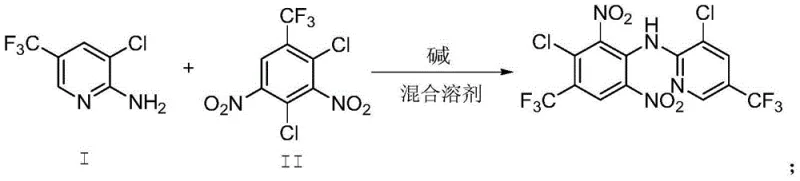
The breakthrough detailed in the patent lies in the synergistic combination of alkyl-substituted aromatic hydrocarbons, such as toluene or xylene, with methyl isobutyl ketone. This binary solvent system creates a reaction environment with superior hydrophobic characteristics compared to any single component alone. By maintaining an extremely low water content within the reaction medium, the process drastically minimizes the hydrolysis of the sensitive dinitro intermediate. Furthermore, this mixed solvent system facilitates a much simpler workup procedure where the solvents can be distilled and recycled with exceptional efficiency. The result is a robust manufacturing protocol that consistently delivers yields greater than 96% and purity levels surpassing 99%, effectively solving the yield-purity trade-off that has hindered previous methods.
Mechanistic Insights into Solvent-Mediated Nucleophilic Aromatic Substitution
The core chemical transformation in fluazinam synthesis is a nucleophilic aromatic substitution (SNAr) reaction between an aminopyridine derivative and a polychlorinated dinitrobenzene derivative. The success of this reaction is heavily dependent on the solvation of the transition state and the stability of the reactants under basic conditions. In the presence of moisture, the electron-deficient aromatic ring of the dinitro intermediate becomes a target for hydroxide ions, leading to the displacement of a chlorine atom and the formation of a phenolic impurity. The mixed solvent system acts as a thermodynamic barrier to this side reaction by sequestering water and maintaining a non-polar environment that favors the desired amine attack over hydrolysis. This precise control over the reaction microenvironment ensures that the kinetic pathway leads almost exclusively to the desired C-N bond formation.
From an impurity control perspective, the mechanism relies on the differential solubility and partitioning of byproducts. The hydrophobic nature of the toluene-MIBK mixture ensures that inorganic salts and polar hydrolysis byproducts remain in the aqueous phase during the acidic quench, while the highly lipophilic fluazinam product stays in the organic layer. This inherent selectivity simplifies the purification train, reducing the need for extensive chromatography or multiple recrystallization steps. The ability to recycle the solvent with a water content of less than 0.5% further reinforces this mechanism, as each subsequent batch begins with a reaction medium that is already optimized to suppress hydrolytic degradation, ensuring batch-to-batch consistency in the impurity profile.
How to Synthesize Fluazinam Efficiently
Implementing this advanced synthesis route requires careful attention to solvent ratios and temperature control to maximize the benefits of the mixed solvent system. The process begins with the dissolution of the amine and dinitro intermediates in the pre-mixed solvent blend, followed by the controlled addition of a base such as potassium hydroxide. Maintaining the reaction temperature between 20°C and 35°C is critical to balance reaction rate with selectivity. Following the reaction completion, a straightforward acidic workup and solvent removal sequence allows for the isolation of the crude product, which is then purified via methanol recrystallization. For a detailed breakdown of the specific operational parameters and stoichiometry required for this process, please refer to the standardized synthesis guide below.
- Charge the reactor with Intermediate I, Intermediate II, and the mixed solvent system comprising alkyl-substituted aromatic hydrocarbons and methyl isobutyl ketone.
- Add solid potassium hydroxide in batches at 20-25°C over 2-3 hours, then warm to 30-35°C to complete the reaction.
- Quench with dilute hydrochloric acid, separate layers, remove solvents under reduced pressure, and recrystallize from methanol to obtain pure fluazinam.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this mixed solvent methodology offers tangible strategic advantages beyond mere chemical yield. The primary benefit lies in the substantial reduction of raw material waste and the optimization of solvent consumption. By utilizing common, industrially available solvents like toluene and MIBK in a recyclable loop, manufacturers can significantly decouple their production costs from the volatility of specialized solvent markets. The simplified purification process also translates to reduced energy consumption and shorter cycle times, as the need for complex distillation columns or extensive drying steps is minimized. This operational efficiency directly contributes to a more resilient supply chain capable of meeting high-volume demands without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive, hard-to-recycle solvents like DMF and the replacement with a recoverable toluene-MIBK system drives down the variable cost per kilogram of production. The high recovery rate of the mixed solvent means that fresh solvent purchases are limited to makeup quantities rather than full batch replacements, leading to significant long-term savings. Additionally, the higher yield means that less starting material is wasted on byproducts, effectively lowering the cost of goods sold (COGS) for the final active ingredient.
- Enhanced Supply Chain Reliability: Toluene and MIBK are commodity chemicals with robust global supply chains, reducing the risk of procurement bottlenecks associated with niche solvents. The ability to recycle the solvent mixture internally creates a buffer against external supply shocks, ensuring continuous operation even during market fluctuations. This reliability is crucial for maintaining steady production schedules and fulfilling long-term contracts with agrochemical formulators who depend on consistent API availability.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial reactors without requiring fundamental changes to the chemistry. The reduced generation of aqueous waste streams containing high levels of organic solvents simplifies wastewater treatment and helps facilities meet increasingly stringent environmental regulations. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the sustainability profile of the manufactured product.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is essential for stakeholders evaluating its adoption for commercial production. The following questions address common inquiries regarding the practical implementation and benefits of the mixed solvent technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear picture of what partners can expect when integrating this process into their manufacturing portfolio.
Q: Why is a mixed solvent system superior to single solvents like DMF or THF for fluazinam synthesis?
A: Single solvents like DMF result in low yields (around 22%), while THF poses safety risks due to flammability. The mixed solvent system of alkyl-substituted aromatics and MIBK offers superior hydrophobicity, effectively suppressing hydrolysis side reactions and enabling easier solvent recovery with lower water content.
Q: What is the primary impurity controlled by this new solvent method?
A: The primary impurity is the hydrolysis product of Intermediate II (the dinitro compound), which forms under alkaline conditions in the presence of water. The low water content of the recycled mixed solvent significantly reduces this side reaction.
Q: Can the solvents used in this process be recycled effectively?
A: Yes, the patent highlights that the mixed solvent system allows for efficient recycling. The recovered solvent mixture maintains a water content of less than 0.5%, which is significantly lower than recovered single-component MIBK, ensuring consistent reaction performance in subsequent batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluazinam Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this mixed solvent process are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of fluazinam meets the highest international standards for agrochemical intermediates. Our commitment to quality assurance means that clients receive a product that is not only chemically pure but also consistent in its physical properties, facilitating seamless formulation downstream.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain visibility into the potential economic impact of switching to this more efficient manufacturing method. We encourage you to reach out for specific COA data and route feasibility assessments to verify how our capabilities align with your project requirements. Let us collaborate to engineer a supply solution that balances performance, cost, and reliability for your agrochemical portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
