Advanced Aripiprazole Manufacturing: Technical Breakthroughs and Commercial Scalability
Advanced Aripiprazole Manufacturing: Technical Breakthroughs and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic pathways for high-volume antipsychotic agents, and the production of Aripiprazole stands as a prime example of process evolution. Patent CN1576273A introduces a refined methodology that addresses critical bottlenecks found in earlier generations of synthesis, specifically targeting the stability and purity of key intermediates. This technical insight report analyzes the strategic advantages of this novel route, which utilizes a condensation reaction between specific piperazine derivatives and quinolinone precursors to achieve pharmaceutical-grade standards with reduced operational complexity. By shifting away from harsh halogenation methods towards controlled sulfonate ester coupling, the process mitigates the formation of stubborn by-products that traditionally plague purification stages. For R&D directors and procurement specialists, understanding the mechanistic underpinnings of this patent is essential for evaluating supply chain resilience and cost structures in the competitive API market. The data suggests that this approach not only enhances the chemical integrity of the final molecule but also streamlines the downstream processing requirements, offering a compelling value proposition for large-scale manufacturing partners seeking reliable Aripiprazole supplier capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Aripiprazole has relied heavily on the methodologies described in patent EP367141, which utilizes 1,4-dibromobutane as a primary alkylating agent to construct the critical butoxy side chain. While effective in a laboratory setting, this conventional route presents significant challenges when translated to industrial scale, primarily due to the high reactivity of the dibromide species in aqueous or alkaline environments. The presence of two bromine atoms creates a susceptibility to hydrolysis reactions, leading to the generation of hydroxyl-substituted by-products that are chemically similar to the desired intermediate and notoriously difficult to separate. As illustrated in the structural representation of the problematic intermediate below, the 7-bromobutoxy moiety is prone to degradation, which compromises the overall mass balance and necessitates extensive recrystallization cycles to meet purity specifications.
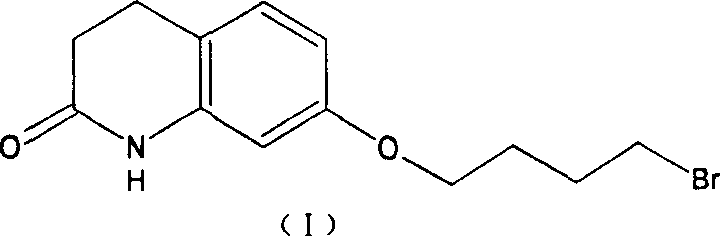
Furthermore, the purification of this brominated intermediate often requires column chromatography or multiple recrystallization steps, which drastically reduces the total recovery rate and increases solvent consumption. The accumulation of impurities at this early stage propagates through the synthesis, influencing the subsequent coupling reaction with the piperazine ring and ultimately affecting the impurity profile of the final Active Pharmaceutical Ingredient. For supply chain managers, these inefficiencies translate into longer lead times and higher waste treatment costs, as the removal of inorganic salts and organic by-products becomes a resource-intensive burden. The inability to guarantee consistent quality without aggressive purification makes the conventional method less attractive for manufacturers aiming for lean, cost-effective production models in the highly regulated pharmaceutical sector.
The Novel Approach
In contrast, the methodology outlined in CN1576273A proposes a strategic substitution of the alkylating agent, replacing 1,4-dibromobutane with 4-chloro-1-butanol or its sulfonate ester derivatives to construct the side chain with greater precision. This novel approach leverages the specific reactivity of the chloro-alcohol or sulfonate group, which offers a better balance between nucleophilic substitution efficiency and stability against hydrolysis under the reaction conditions. By decoupling the side chain synthesis from the final ring closure, the process allows for the isolation and characterization of high-purity intermediates, such as 4-[4-(2,3-dichlorophenyl)piperazinyl]-1-butanol, before they are committed to the final condensation step. This modularity ensures that any impurities generated during the side chain formation can be removed effectively before the most valuable coupling reaction takes place, thereby safeguarding the yield of the final Aripiprazole product.
The implementation of this route also simplifies the work-up procedures, as the by-products formed are often more polar or ionic, facilitating their removal through standard aqueous washes rather than complex chromatographic separations. The patent data indicates that this method can achieve crude product purity exceeding 97% with a single recrystallization step, a significant improvement over the multi-step purification required by older methods. For procurement teams, this translates to a more predictable manufacturing timeline and reduced dependency on expensive purification resins or solvents. The ability to control the quality of the intermediate effectively means that the final API can consistently meet stringent pharmacopeial standards without the need for excessive reprocessing, making this novel approach a superior choice for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Sulfonate-Mediated Condensation
The core of this improved synthesis lies in the mechanistic efficiency of the nucleophilic substitution reaction between the piperazine-containing side chain and the quinolinone core. In the preferred embodiment, the hydroxyl group of the butanol intermediate is activated by conversion into a sulfonate ester, typically using p-toluenesulfonyl chloride, which transforms a poor leaving group into an excellent one for the subsequent displacement reaction. This activation step is crucial because it allows the coupling with 7-hydroxy-3,4-dihydroquinolin-2(1H)-one to proceed under milder alkaline conditions, minimizing the risk of base-catalyzed degradation of the sensitive quinolinone ring system. The reaction mechanism involves the deprotonation of the phenolic hydroxyl on the quinolinone by a base such as potassium hydroxide, generating a phenoxide ion that attacks the electrophilic carbon of the sulfonate ester in an SN2 fashion.
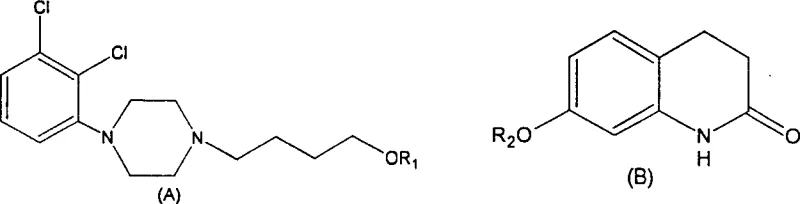
This mechanistic pathway is highly selective, as the sulfonate leaving group is displaced cleanly without the competing elimination reactions that often plague alkyl halide couplings at elevated temperatures. The use of polar aprotic solvents like acetonitrile or DMF further enhances the nucleophilicity of the phenoxide ion while stabilizing the transition state, leading to the high yields reported in the patent examples. From an impurity control perspective, this mechanism avoids the formation of dialkylated piperazine by-products or ether-linked dimers that are common in less controlled alkylation processes. The result is a cleaner reaction profile where the primary impurity is simply the unreacted starting material, which can be easily removed during the crystallization of the final product. For R&D directors, this level of mechanistic control provides confidence in the robustness of the process, ensuring that scale-up from kilogram to tonne quantities will not introduce unforeseen chemical complexities.
How to Synthesize Aripiprazole Efficiently
The synthesis of Aripiprazole via this optimized route involves a sequential three-step process that prioritizes the quality of intermediates to ensure final product integrity. The initial phase focuses on the construction of the piperazine-butanol side chain through the reaction of 1-(2,3-dichlorophenyl)piperazine with a halo-alcohol, followed by activation of the alcohol functionality. The subsequent steps involve the precise coupling of this activated side chain with the quinolinone core under controlled thermal and alkaline conditions. Detailed standard operating procedures for each stage, including specific molar ratios, temperature profiles, and work-up protocols, are essential for replicating the high yields and purity described in the technical literature. The following guide outlines the critical operational milestones required to execute this synthesis successfully in a GMP-compliant environment.
- Preparation of 4-[4-(2,3-dichlorophenyl)piperazinyl]-1-butanol via nucleophilic substitution of 1-(2,3-dichlorophenyl)piperazine with 4-halo-1-butanol.
- Conversion of the butanol intermediate into a sulfonate ester using p-toluenesulfonyl chloride to enhance leaving group ability.
- Final condensation reaction between the sulfonate ester and 7-hydroxy-3,4-dihydroquinolin-2(1H)-one under alkaline conditions to form Aripiprazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible benefits that extend beyond mere chemical yield, impacting the overall cost of goods sold and supply reliability. The elimination of 1,4-dibromobutane removes a significant source of raw material volatility and cost, as the alternative chloro-alcohol or sulfonate precursors are generally more abundant and stable in the global chemical market. This shift in raw material strategy reduces the risk of supply disruptions caused by the limited availability of high-purity dibromides, ensuring a more continuous flow of production batches. Furthermore, the simplified purification process reduces the consumption of organic solvents and energy required for distillation and recrystallization, contributing to substantial cost savings in manufacturing overheads without compromising on quality standards.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers production costs by minimizing the number of unit operations required to achieve pharmaceutical-grade purity. By avoiding the extensive chromatographic purification needed in conventional methods, manufacturers can reduce solvent waste and labor hours associated with complex work-ups. The higher crude purity achieved directly translates to less material loss during recrystallization, improving the overall mass efficiency of the plant. Additionally, the use of common alkaline reagents and solvents avoids the need for specialized or expensive catalysts, further driving down the variable cost per kilogram of the final API.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain reliability by reducing the sensitivity of the process to minor fluctuations in reaction conditions. The stability of the intermediates allows for longer hold times and more flexible scheduling, which is critical for managing large-scale production campaigns. The use of readily available starting materials mitigates the risk of raw material shortages, ensuring that production timelines can be met consistently. This reliability is essential for maintaining the continuity of supply for downstream formulation partners who depend on timely delivery of high-purity Aripiprazole.
- Scalability and Environmental Compliance: The process is inherently scalable, as the reaction conditions are mild and do not require extreme temperatures or pressures that pose engineering challenges at large volumes. The reduction in hazardous waste generation, particularly from brominated by-products, simplifies environmental compliance and waste treatment protocols. This aligns with modern green chemistry principles, making the manufacturing process more sustainable and acceptable to regulatory bodies. The ability to scale up complex pharmaceutical intermediates efficiently ensures that the production capacity can be expanded to meet growing market demand without significant capital investment in new equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Aripiprazole synthesis route. These answers are derived from the specific technical disclosures and experimental data provided in the patent documentation, offering clarity on process feasibility and quality outcomes. Understanding these details is crucial for stakeholders evaluating the potential for technology transfer or contract manufacturing partnerships. The responses highlight the key differentiators of this method compared to legacy processes, focusing on purity, yield, and operational safety.
Q: How does the new synthesis route improve impurity control compared to the EP367141 method?
A: The novel process replaces 1,4-dibromobutane with 4-chloro-1-butanol, significantly reducing hydrolysis side reactions and simplifying the purification of the key intermediate, leading to higher crude purity.
Q: What are the typical reaction conditions for the final condensation step?
A: The final coupling typically proceeds in polar aprotic solvents like acetonitrile or DMF with alkaline reagents such as potassium hydroxide at temperatures ranging from 80°C to 140°C.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes readily available raw materials and avoids extreme conditions, offering stable yields and easier waste treatment, which are critical for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aripiprazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of process robustness in the supply of life-saving medications like Aripiprazole. Our technical team has extensively analyzed the pathways described in CN1576273A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. We are committed to delivering high-purity Aripiprazole that meets stringent purity specifications through our rigorous QC labs, ensuring that every batch conforms to the highest international standards. Our facility is equipped to handle the specific solvent systems and reaction conditions outlined in this novel route, guaranteeing a seamless transition from pilot scale to full commercial manufacturing.
We invite global pharmaceutical partners to collaborate with us on optimizing their supply chain for this critical antipsychotic agent. By leveraging our expertise in process chemistry, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments that demonstrate our capability to be your long-term strategic partner. Let us help you secure a stable, high-quality supply of Aripiprazole that supports your mission to improve patient outcomes worldwide.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
