Advanced Manufacturing of Aripiprazole Intermediates via Optimized Cyclization Routes
Advanced Manufacturing of Aripiprazole Intermediates via Optimized Cyclization Routes
The pharmaceutical landscape for atypical antipsychotics continues to evolve, driven by the need for more efficient and environmentally sustainable manufacturing processes. Patent CN1634889A introduces a significant breakthrough in the synthesis of Aripiprazole, a critical medication for treating schizophrenia and schizoaffective disorders. This technology addresses longstanding challenges in the production of the key intermediate, specifically focusing on reducing reaction times and eliminating hazardous solvents. By optimizing the coupling of piperazine derivatives with halogenated phenols, the disclosed method offers a streamlined pathway that enhances overall process safety and throughput. For global supply chain leaders, this represents a pivotal shift towards greener chemistry without compromising the structural integrity or purity of the final active pharmaceutical ingredient.
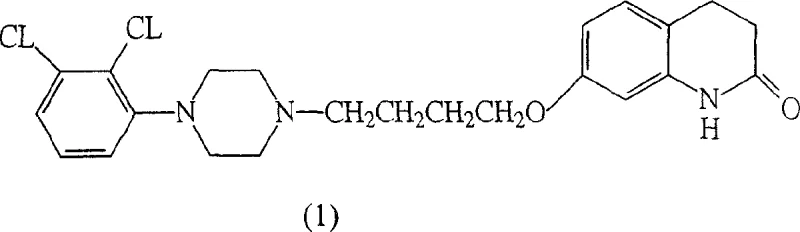
Aripiprazole acts as a partial agonist at dopamine D2 and serotonin 5-HT1A receptors, and its complex molecular architecture demands precise synthetic control. The traditional reliance on chlorinated solvents and extended reaction durations has historically created bottlenecks in production capacity and increased waste disposal costs. The innovations presented in this patent provide a robust alternative, utilizing a versatile array of six different processes to generate the crucial intermediate (4). This flexibility allows manufacturers to adapt raw material sourcing based on availability and cost, ensuring business continuity even during market fluctuations. As a reliable API intermediate supplier, understanding these nuanced synthetic variations is essential for maintaining a competitive edge in the generic pharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Aripiprazole has been hindered by inefficient reaction kinetics and the use of environmentally detrimental reagents. Prior art, such as the methods described in related patents, often necessitates reaction times extending beyond 9 hours, which severely limits reactor turnover rates and overall plant productivity. Furthermore, the heavy reliance on chloroform as a primary solvent poses significant occupational health risks and complicates waste management protocols due to its high toxicity and regulatory scrutiny. These factors collectively inflate the cost of goods sold (COGS) and introduce unnecessary volatility into the supply chain. For procurement managers, the dependency on such hazardous materials also increases liability insurance premiums and requires specialized infrastructure for safe handling and storage, creating a substantial barrier to entry for new manufacturers.
The Novel Approach
In stark contrast, the methodology outlined in CN1634889A leverages a series of optimized nucleophilic substitution reactions that drastically shorten the production cycle. By employing safer, low-toxicity solvents such as ethyl acetate, petroleum ether, and dehydrated alcohol, the new process aligns with modern green chemistry principles while maintaining high reaction efficiency. The reaction time is compressed to a window of merely 5 to 6 hours, effectively doubling the potential throughput of existing manufacturing assets. This approach not only mitigates environmental impact but also simplifies the downstream purification steps, as the removal of less toxic solvents is generally more straightforward and energy-efficient. The versatility of the method allows for the use of various halogenated precursors, providing a strategic advantage in raw material procurement.
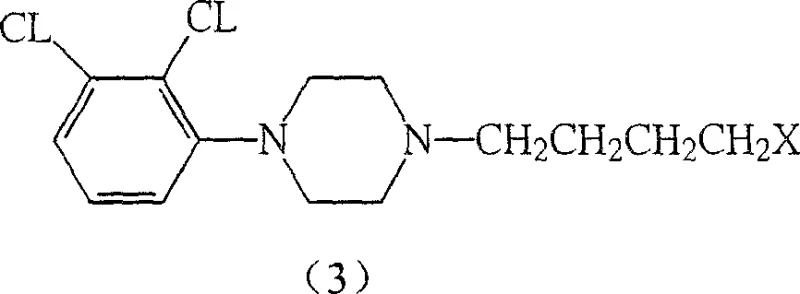
The core of this innovation lies in the formation of intermediate (4), which serves as the direct precursor to the final drug substance. The patent details multiple pathways to achieve this, including the reaction of specific phenol derivatives with diethylenediamine compounds under controlled thermal conditions. This modularity ensures that if one specific raw material faces a shortage, alternative routes can be activated without halting production. For a reliable agrochemical intermediate supplier or pharma partner, this redundancy is invaluable. It demonstrates a deep understanding of process chemistry that goes beyond simple recipe following, offering a resilient framework for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Lewis Acid Catalyzed Cyclization
The transformation of intermediate (4) into the final Aripiprazole molecule (1) is a critical ring-closing step that defines the quality and yield of the entire process. This cyclization is facilitated by strong Lewis acids, which activate the carbonyl group and promote intramolecular electrophilic aromatic substitution. The patent specifies a range of effective catalysts, including Aluminum Chloride (AlCl3), Tin Tetrachloride (SnCl4), and Zinc Chloride (ZnCl2), operating at temperatures ranging from room temperature up to 200°C, with an optimal range of 160-180°C. The choice of catalyst influences the reaction rate and the profile of by-products, making the selection of the appropriate Lewis acid a key variable for process optimization. Understanding this mechanism allows R&D teams to fine-tune reaction parameters to minimize impurities and maximize the yield of the target compound.
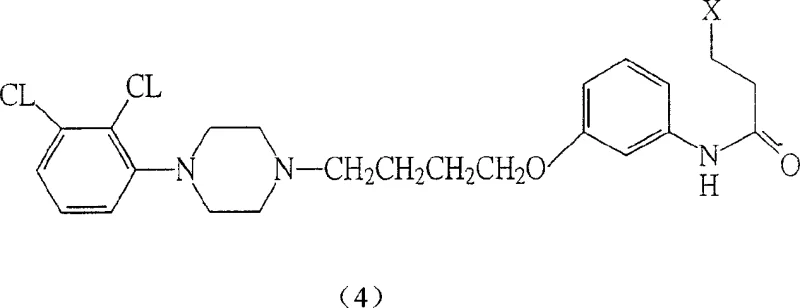
Impurity control is paramount in the synthesis of psychotropic medications, where even trace contaminants can have significant biological effects. The described mechanism inherently limits the formation of side products by favoring the thermodynamic stability of the fused ring system under the specified acidic conditions. The use of molten salt systems or specific solvent combinations further aids in dissolving the intermediate and ensuring homogeneous reaction conditions, which is crucial for consistent batch-to-batch quality. By strictly controlling the temperature and the stoichiometry of the Lewis acid, manufacturers can achieve a purity profile that meets stringent regulatory standards. This level of mechanistic control is what distinguishes a high-purity OLED material or pharmaceutical intermediate from a commodity chemical, adding significant value to the final product.
How to Synthesize Aripiprazole Intermediate Efficiently
The synthesis of the key intermediate involves a carefully orchestrated sequence of nucleophilic attacks and subsequent workup procedures. The process begins with the activation of the phenolic hydroxyl group or the halogenated alkyl chain, followed by coupling with the piperazine nitrogen. Detailed operational parameters, including specific molar ratios and temperature ramps, are critical for success. The patent emphasizes the importance of base selection, recommending inorganic bases like sodium carbonate or organic amines to neutralize the acid by-products generated during the substitution. For a comprehensive guide on executing this synthesis with maximum efficiency and safety, please refer to the standardized protocol below.
- React a halogenated phenol derivative with a piperazine compound in the presence of a base and appropriate solvent at temperatures between 50-150°C.
- Isolate the resulting intermediate compound (4) through filtration and purification techniques such as recrystallization.
- Perform a ring-closing reaction on intermediate (4) using Lewis acids like AlCl3 or SnCl4 at elevated temperatures (160-180°C) to yield Aripiprazole.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting the synthesis route described in CN1634889A offers tangible benefits that extend far beyond the laboratory bench, directly impacting the bottom line and operational resilience of manufacturing organizations. The shift away from chlorinated solvents eliminates the need for expensive containment systems and specialized waste treatment facilities, leading to substantial cost savings in facility maintenance and regulatory compliance. Moreover, the reduced reaction time translates directly into increased asset utilization, allowing producers to meet surging market demand without significant capital expenditure on new reactors. This efficiency gain is a powerful driver for cost reduction in pharmaceutical manufacturing, enabling more competitive pricing strategies in the generic drug sector.
- Cost Reduction in Manufacturing: The elimination of toxic chloroform and the adoption of common industrial solvents like ethyl acetate significantly lower raw material procurement costs and waste disposal fees. By removing the need for complex solvent recovery systems dedicated to halogenated compounds, the overall energy consumption of the plant is drastically reduced. This streamlined solvent profile simplifies the supply chain, as these materials are readily available from multiple global vendors, reducing the risk of supply disruptions and price volatility associated with specialty chemicals.
- Enhanced Supply Chain Reliability: The flexibility to utilize six different synthetic routes for the intermediate provides a robust buffer against raw material shortages. If a specific halogenated precursor becomes unavailable due to geopolitical or logistical issues, the process can be swiftly adapted to use an alternative starting material without revalidating the entire workflow. This adaptability ensures continuous production schedules and reliable delivery timelines for downstream customers, reinforcing the manufacturer's reputation as a dependable partner in the global healthcare supply chain.
- Scalability and Environmental Compliance: The reaction conditions, which operate effectively at atmospheric pressure and moderate temperatures, are inherently safer and easier to scale from pilot plants to multi-ton commercial production. The reduced toxicity of the reagents aligns with increasingly strict environmental regulations, minimizing the risk of fines and shutdowns. This proactive approach to environmental stewardship not only protects the company's license to operate but also appeals to socially responsible investors and customers who prioritize sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the technology's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own production lines.
Q: How does this new method improve upon conventional Aripiprazole synthesis?
A: The patented method significantly reduces reaction time from over 9 hours to approximately 5-6 hours and replaces toxic chloroform with safer solvents like ethyl acetate and ethanol, enhancing both operational safety and environmental compliance.
Q: What catalysts are suitable for the final cyclization step?
A: The process allows for flexibility in catalyst selection, utilizing Lewis acids such as Aluminum Chloride (AlCl3), Tin Tetrachloride (SnCl4), Boron Trifluoride (BF3), or Zinc Chloride (ZnCl2) to facilitate the ring-closure efficiently.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the use of common industrial solvents and robust reaction conditions (room temperature to 200°C) makes the process highly adaptable for large-scale manufacturing, ensuring consistent supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aripiprazole Intermediate Supplier
The technological advancements detailed in CN1634889A underscore the complexity and precision required to produce high-quality psychiatric medications. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-volume supply. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for final API synthesis. We understand that in the pharmaceutical industry, consistency is key, and our commitment to quality assurance is unwavering.
We invite you to collaborate with us to leverage these advanced synthetic routes for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain and reduce your time to market.
