Advanced Synthesis of Selective Indole Derivatives for High-Efficacy Muscarinic Antagonists
Advanced Synthesis of Selective Indole Derivatives for High-Efficacy Muscarinic Antagonists
The pharmaceutical industry continuously seeks novel therapeutic agents that offer superior efficacy with minimized adverse effects, particularly in the realm of anticholinergic treatments for conditions such as irritable bowel syndrome and chronic obstructive pulmonary disease. Patent CN103242214A introduces a groundbreaking class of indole derivatives represented by general formula (I), which function as highly selective antagonists of muscarinic acetylcholine receptors. These compounds address a critical unmet medical need by demonstrating a pronounced preference for smooth muscle receptors in the ileum and trachea over cardiac receptors, thereby drastically reducing the risk of tachycardia and other cardiovascular complications associated with earlier generation drugs. The synthesis methodology outlined in this patent provides a robust and scalable route for producing these high-value pharmaceutical intermediates, utilizing readily available starting materials and standard organic transformation techniques that are well-suited for industrial application.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, anticholinergic agents such as oxybutynin and propantheline bromide have been widely utilized, yet their clinical utility is often compromised by a lack of organ selectivity. These conventional molecules, often based on quaternary ammonium salt structures, exhibit antagonistic activity against muscarinic receptors in the heart that is similar to or even greater than their activity in the target tissues like the bladder or gastrointestinal tract. This non-selective binding profile leads to significant systemic side effects, including severe dizziness and accelerated heart rate, which can limit patient compliance and therapeutic dosage. Furthermore, while newer agents like darifenacin have improved selectivity, there remains a persistent demand for compounds with even higher specificity and potency to treat complex disorders effectively without compromising patient safety profiles.
The Novel Approach
The innovative strategy presented in the patent data involves the construction of a specific indole scaffold substituted with diphenyl butyramide moieties, which fundamentally alters the receptor binding dynamics. By systematically varying the substituents at the R1, R2, R3, and R4 positions, chemists can fine-tune the steric and electronic properties of the molecule to maximize affinity for M3 receptors while minimizing interaction with M2 cardiac receptors. The synthetic route is designed to be modular, allowing for the introduction of halogens or alkyl groups at key positions to enhance biological activity. This approach not only yields compounds with superior pharmacological profiles but also employs a streamlined chemical process that avoids the use of exotic reagents, facilitating easier purification and higher overall yields compared to more complex heterocyclic syntheses.
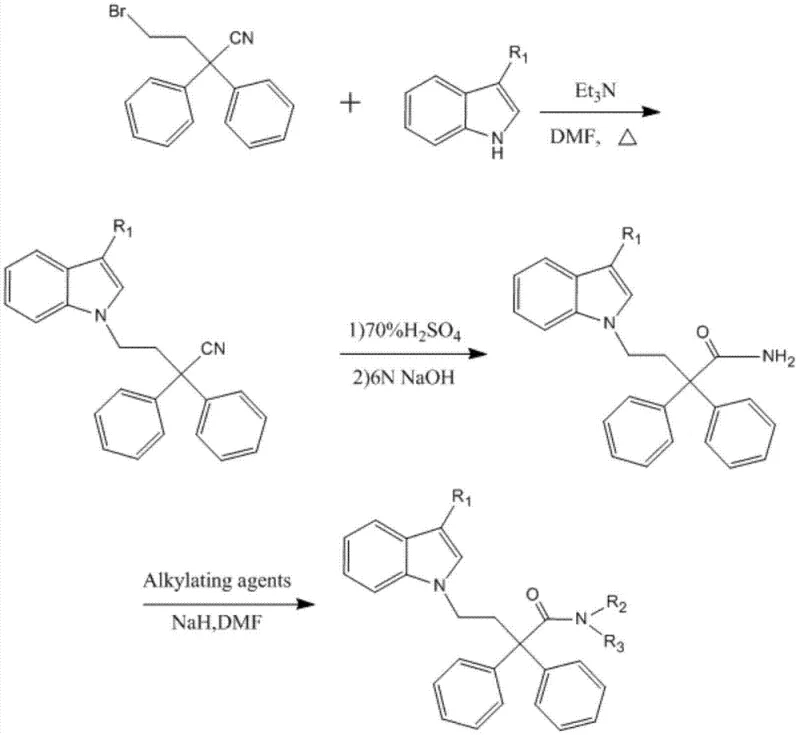
As illustrated in the reaction scheme above, the synthesis for derivatives where R4 is a hydrogen atom begins with the nucleophilic substitution of 4-bromo-2,2-diphenylbutyronitrile with an indole nucleus. This initial alkylation is conducted in dimethylformamide (DMF) with triethylamine as a base at elevated temperatures around 150°C, ensuring efficient coupling of the bulky diphenyl group to the indole nitrogen. Following this, the nitrile group undergoes acidic hydrolysis using 70% sulfuric acid at 140°C to convert it into the corresponding primary amide, a critical functional group for subsequent modification. The final step involves the N-alkylation of the amide nitrogen using alkylating agents in the presence of sodium hydride, allowing for the precise installation of methyl, ethyl, or isopropyl groups to optimize the drug's metabolic stability and receptor binding affinity.
Mechanistic Insights into Indole N-Alkylation and Amide Modification
The core chemical transformation driving this synthesis is the nucleophilic attack of the indole nitrogen on the electrophilic carbon of the bromo-butyronitrile chain. This reaction is facilitated by the use of a polar aprotic solvent like DMF, which stabilizes the transition state and enhances the nucleophilicity of the indole anion generated in situ by triethylamine. The high temperature of 150°C is necessary to overcome the activation energy barrier associated with displacing the bromide ion from a sterically hindered tertiary carbon center adjacent to two phenyl rings. Monitoring via thin-layer chromatography (TLC) is essential to ensure complete consumption of the starting bromide, as residual halide can complicate downstream purification and affect the purity profile of the final active pharmaceutical ingredient.
Following the formation of the nitrile intermediate, the conversion to the amide involves a classic acid-catalyzed hydration mechanism. The nitrile nitrogen is protonated by the concentrated sulfuric acid, making the carbon susceptible to nucleophilic attack by water molecules present in the acid matrix or added during workup. This proceeds through an imidic acid intermediate which tautomerizes to the stable amide form. The subsequent alkylation of the amide nitrogen requires strong basic conditions provided by sodium hydride to deprotonate the relatively weakly acidic amide proton. This generates a resonance-stabilized anion that attacks the alkyl halide (such as methyl iodide or isopropyl bromide) in an SN2 fashion. Careful control of the reaction temperature at 0-5°C during this step is crucial to prevent side reactions such as elimination or over-alkylation, ensuring the formation of the desired mono-alkylated product with high regioselectivity.
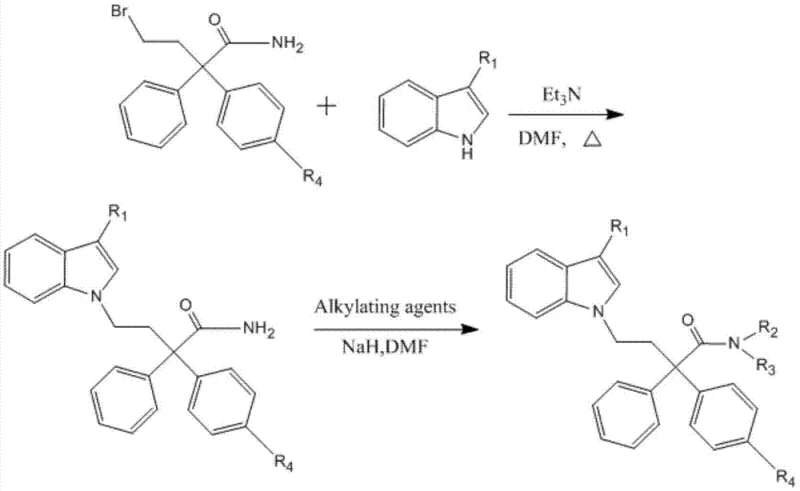
For derivatives containing halogen substituents on the phenyl ring (where R4 is Cl or Br), the synthetic strategy adapts by utilizing pre-halogenated starting materials, specifically 4-bromo-2-phenyl-2-(chlorophenyl or bromophenyl) butanamide. As shown in the second reaction pathway, this allows the halogen functionality to be preserved throughout the synthesis, avoiding the need for harsh halogenation conditions that could damage the sensitive indole ring. The coupling reaction proceeds similarly to the non-halogenated series, utilizing triethylamine in DMF at 150°C to attach the indole moiety. The presence of the halogen atom significantly enhances the lipophilicity and potentially the receptor binding affinity of the final molecule, as evidenced by the pharmacological data indicating higher activity for compounds 8 and 16. This modular approach demonstrates the versatility of the platform technology in generating a diverse library of analogs for structure-activity relationship (SAR) studies.
How to Synthesize Selective Indole Derivatives Efficiently
The synthesis of these high-purity pharmaceutical intermediates requires strict adherence to the optimized reaction parameters detailed in the patent to ensure consistent quality and yield. The process is divided into three distinct operational stages: the initial N-alkylation of the indole, the hydrolysis of the nitrile to the amide, and the final functionalization of the amide nitrogen. Each step necessitates specific workup procedures, including aqueous quenching, organic extraction with dichloromethane, and purification via silica gel column chromatography followed by recrystallization from ethyl acetate. Detailed standardized operating procedures for these steps are essential for maintaining batch-to-batch consistency in a GMP environment.
- Perform N-alkylation of substituted or unsubstituted indoles with 4-bromo-2,2-diphenylbutyronitrile using triethylamine in DMF at 150°C.
- Hydrolyze the resulting nitrile intermediate using 70% sulfuric acid at 140°C, followed by neutralization and extraction to obtain the primary amide.
- React the primary amide with alkylating agents in the presence of NaH in DMF under ice bath conditions to yield the final N-alkylated indole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the synthetic route described in patent CN103242214A offers substantial advantages over traditional methods for producing muscarinic antagonists. The reliance on commodity chemicals such as indole, triethylamine, and common alkyl halides ensures a stable and cost-effective raw material supply base, mitigating the risks associated with sourcing specialized or controlled precursors. Furthermore, the avoidance of precious metal catalysts like palladium or platinum eliminates the need for expensive metal scavenging steps and rigorous testing for heavy metal residues, which significantly simplifies the regulatory filing process and reduces overall manufacturing costs. This streamlined process flow translates directly into improved margin potential for generic drug manufacturers and contract development organizations.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of standard organic solvents like DMF and dichloromethane significantly lower the direct material costs associated with production. Additionally, the robust nature of the reaction conditions allows for simpler equipment requirements, avoiding the need for specialized high-pressure or cryogenic reactors, which further drives down capital expenditure and operational overheads in large-scale facilities.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted indoles and diphenyl butyronitrile derivatives, are commercially available from multiple global suppliers, ensuring a diversified supply chain that is resilient to regional disruptions. The synthetic steps are chemically robust with high tolerance for minor variations in reagent quality, reducing the likelihood of batch failures and ensuring consistent on-time delivery of critical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The process utilizes standard unit operations such as liquid-liquid extraction and crystallization, which are easily scalable from pilot plant to multi-ton commercial production without significant re-engineering. The waste streams primarily consist of aqueous saline solutions and organic solvents that can be efficiently recovered and recycled, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indole derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals evaluating this technology for potential licensing or procurement opportunities.
Q: How does this indole derivative improve upon existing anticholinergic drugs?
A: Unlike traditional quaternary ammonium salts which exhibit poor organ selectivity and cause severe cardiac side effects, these novel indole derivatives demonstrate significantly higher affinity for smooth muscle muscarinic receptors (ileum/trachea) compared to cardiac receptors, thereby minimizing heart rate acceleration and dizziness.
Q: What are the critical reaction conditions for the hydrolysis step?
A: The hydrolysis of the nitrile intermediate to the amide requires heating with 70% sulfuric acid at 140°C. Precise temperature control is essential to ensure complete conversion without degrading the sensitive indole ring structure.
Q: Can this synthesis be scaled for commercial API production?
A: Yes, the process utilizes standard organic solvents like DMF and dichloromethane and avoids expensive transition metal catalysts, making it highly suitable for commercial scale-up from kilogram to multi-ton production levels with robust quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates, guaranteeing that every batch of indole derivative delivered meets the highest international standards. We understand the critical importance of supply continuity in the pharmaceutical value chain and are committed to providing a seamless partnership that supports your drug development and commercialization goals.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your project's economic viability. Partner with us to leverage our technical expertise and secure a reliable supply of high-quality indole derivatives for your next-generation anticholinergic therapies.
