Advanced Synthesis of Selective Pyrazole Anticholinergic Intermediates for Commercial Scale-up
Introduction to Novel Anticholinergic Technology
The pharmaceutical landscape for treating disorders such as irritable bowel syndrome and chronic obstructive pulmonary disease is constantly evolving, driven by the need for agents with superior safety profiles. Patent CN103275007A introduces a groundbreaking class of pyrazole derivatives that function as selective muscarinic acetylcholine receptor antagonists. Unlike traditional anticholinergics which often suffer from poor organ selectivity leading to cardiac side effects, these novel compounds demonstrate a marked preference for smooth muscle receptors in the ileum and trachea over cardiac receptors. This technological leap addresses a critical unmet medical need by minimizing adverse events such as dizziness and heart rate acceleration while maintaining potent therapeutic efficacy. For R&D teams and procurement specialists, this represents a significant opportunity to access high-value intermediates that enable the production of next-generation therapeutics with improved patient compliance and safety margins.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of anticholinergic agents has been hindered by the structural limitations of early generations of drugs, such as quaternary ammonium salts and certain alkaloids. These conventional molecules often act as non-selective antagonists, blocking muscarinic receptors indiscriminately across various organ systems. This lack of specificity results in a narrow therapeutic window where the dose required to treat bladder or airway dysfunction inevitably triggers severe cardiovascular complications. Furthermore, the synthetic routes for modifying these older scaffolds to improve selectivity have often been complex, requiring harsh conditions or expensive chiral catalysts that drive up manufacturing costs. The inability to finely tune the receptor affinity profile has long been a bottleneck in bringing safer antispasmodic and bronchodilator medications to the market, necessitating a fundamental shift in molecular design strategy.
The Novel Approach
The methodology outlined in the patent presents a robust and scalable solution by integrating a pyrazole heterocycle into a 2,2-diphenylbutanamide scaffold. This structural modification is not merely cosmetic; it fundamentally alters the electronic and steric properties of the molecule, enhancing its binding affinity for M3 receptors in smooth muscle while sparing M2 receptors in the heart. The synthetic pathway is elegantly designed, utilizing readily available starting materials like 4-bromo-2,2-diphenylbutanamide and pyrazole. As illustrated in the reaction scheme below, the process avoids the use of precious metal catalysts, relying instead on standard nucleophilic substitution and alkylation techniques that are well-understood in industrial chemistry. This approach not only simplifies the supply chain but also ensures that the resulting intermediates possess the precise stereochemical and functional attributes required for high-selectivity biological activity.
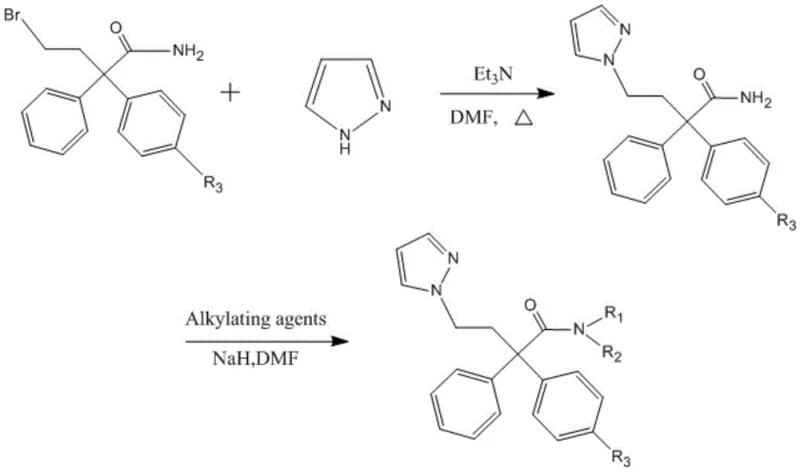
Mechanistic Insights into Nucleophilic Substitution and Alkylation
The core of this synthesis lies in a two-stage transformation that builds molecular complexity with high efficiency. The first stage involves the nucleophilic displacement of a bromide leaving group on the butanamide chain by the nitrogen atom of the pyrazole ring. This reaction is facilitated by triethylamine as a base in dimethylformamide (DMF) at elevated temperatures around 140°C. The thermal energy provided is crucial for overcoming the activation barrier of the substitution, ensuring complete conversion of the bromo-precursor into the pyrazole-substituted intermediate. Following this, the reaction mixture undergoes a rigorous workup involving aqueous quenching and organic extraction, followed by purification via silica gel chromatography. This step is critical for removing unreacted starting materials and byproducts, establishing a high-purity foundation for the subsequent modification.
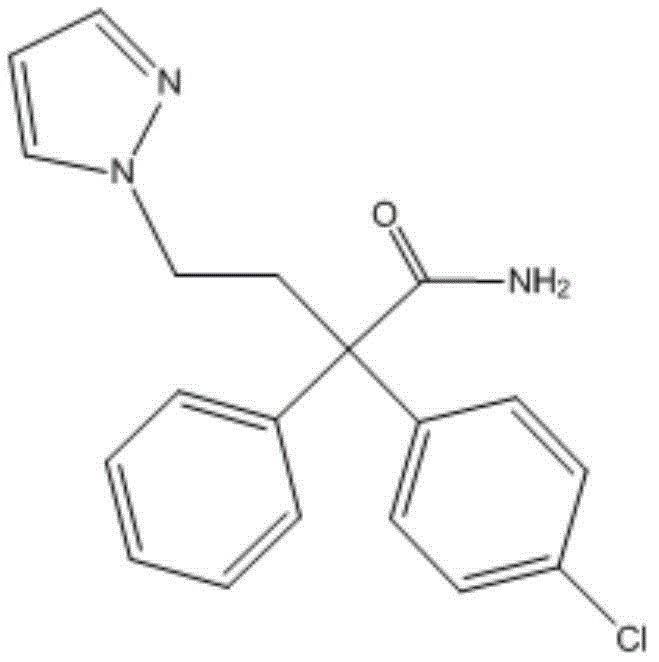
The second stage focuses on the functionalization of the amide nitrogen to fine-tune the pharmacological profile. Using sodium hydride (NaH) as a strong base in an ice bath at 0-5°C, the amide proton is deprotonated to form a reactive nucleophile. This species then attacks various alkylating agents, such as methyl iodide or ethyl bromide, to introduce diverse alkyl groups (R1 and R2). The low temperature is essential here to control the exothermic nature of the deprotonation and prevent side reactions like over-alkylation or decomposition. The patent data indicates that introducing specific substituents, particularly halogens on the phenyl ring (R3) as seen in compounds like Compound 7, further amplifies the biological potency. This modular approach allows for the rapid generation of a library of analogs, enabling medicinal chemists to optimize the balance between potency, selectivity, and metabolic stability without reinventing the synthetic wheel for each new candidate.
How to Synthesize Pyrazole Derivatives Efficiently
The synthesis of these high-value pharmaceutical intermediates is designed for reproducibility and scalability, making it an ideal candidate for technology transfer from lab to plant. The process leverages common industrial solvents and reagents, minimizing the need for specialized equipment or hazardous exotic chemicals. By adhering to the specific temperature gradients and stoichiometric ratios defined in the patent, manufacturers can achieve consistent yields ranging from 40% to over 60% across different analogs. The detailed标准化 synthesis steps见下方的指南 ensure that critical parameters such as reaction time and purification methods are optimized for maximum recovery and purity. This level of procedural clarity is vital for maintaining batch-to-batch consistency, a key requirement for regulatory approval in the pharmaceutical sector.
- React 4-bromo-2,2-diphenylbutanamide with pyrazole and triethylamine in DMF at 140°C to form the intermediate.
- Purify the intermediate via silica gel column chromatography and recrystallization in ethyl acetate.
- Alkylate the intermediate using alkylating agents and NaH in DMF at 0-5°C to obtain the final target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial strategic benefits for supply chain managers and procurement officers looking to optimize their API sourcing strategies. The reliance on commodity chemicals such as pyrazole, triethylamine, and simple alkyl halides means that raw material costs are inherently stable and predictable, shielding the production budget from the volatility often associated with specialized reagents. Furthermore, the absence of transition metal catalysts eliminates the need for expensive and time-consuming metal scavenging steps, which are typically required to meet stringent residual metal limits in final drug substances. This simplification of the downstream processing workflow translates directly into reduced operational expenditures and shorter cycle times, allowing for faster response to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of standard solvent systems like DMF and dichloromethane significantly lower the direct material costs associated with production. Additionally, the purification strategy relies on conventional silica gel chromatography and recrystallization, techniques that are easily scalable and do not require capital-intensive preparative HPLC infrastructure. This streamlined process flow reduces the overall cost of goods sold (COGS), providing a competitive pricing advantage for the final active pharmaceutical ingredient. By avoiding complex multi-step sequences or cryogenic conditions beyond the initial alkylation, the energy consumption of the plant is also optimized, contributing to a leaner manufacturing footprint.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks that can disrupt production schedules. The robustness of the reaction conditions, particularly the tolerance for standard heating and stirring protocols, ensures that the process can be reliably executed in diverse manufacturing environments without requiring highly specialized operator training. This accessibility of inputs and simplicity of execution enhances the resilience of the supply chain, ensuring continuous availability of these critical intermediates even during periods of global logistical stress. Consequently, partners can maintain higher safety stocks and shorter lead times, securing their position in the competitive pharmaceutical market.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been demonstrated effectively from gram-scale laboratory experiments to potential multi-kilogram production runs without loss of efficiency. The waste streams generated are primarily composed of standard organic solvents and inorganic salts, which can be managed through established recycling and treatment protocols, aligning with modern green chemistry principles. The high selectivity of the reaction minimizes the formation of difficult-to-separate impurities, reducing the volume of solvent required for purification and lowering the overall environmental burden. This alignment with sustainability goals not only meets regulatory expectations but also appeals to environmentally conscious stakeholders, adding a layer of corporate social responsibility value to the procurement decision.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these pyrazole derivatives. The answers are derived directly from the experimental data and mechanistic explanations provided in the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating these intermediates into your existing development pipelines. We encourage further discussion with our technical team to explore specific customization options tailored to your project requirements.
Q: What is the primary advantage of these pyrazole derivatives over traditional anticholinergics?
A: These derivatives exhibit significantly higher selectivity for muscarinic receptors in smooth muscle (ileum and trachea) compared to cardiac tissue, thereby reducing severe side effects like tachycardia.
Q: What are the key reaction conditions for the synthesis?
A: The synthesis involves a high-temperature nucleophilic substitution at 140°C followed by a low-temperature alkylation step at 0-5°C using sodium hydride in DMF.
Q: How does the halogen substitution affect biological activity?
A: Introducing halogen substituents such as chlorine or bromine on the phenyl ring further enhances the antagonistic activity and selectivity of the compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of safer and more effective pharmaceuticals. Our facility is equipped with state-of-the-art reactors and purification systems capable of handling the specific thermal and chemical requirements of this pyrazole synthesis route. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every batch, guaranteeing that the material you receive meets the highest industry standards for clinical and commercial use.
We invite you to collaborate with us to leverage this innovative technology for your next-generation anticholinergic projects. Our team is ready to provide a Customized Cost-Saving Analysis that details how switching to this synthetic route can optimize your budget without compromising on quality. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. We are dedicated to supporting your R&D and supply chain goals with transparent communication and superior service, positioning your organization for success in the competitive global marketplace.
