Optimized Synthetic Route for Remdesivir Intermediates: Enhancing Purity and Commercial Scalability
Optimized Synthetic Route for Remdesivir Intermediates: Enhancing Purity and Commercial Scalability
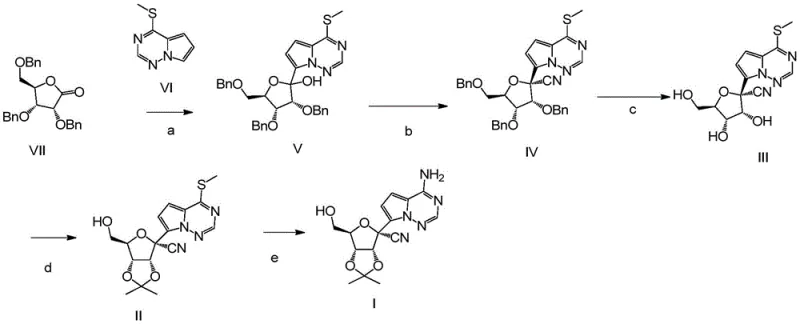
The global pharmaceutical landscape has witnessed an unprecedented demand for antiviral therapeutics, with Remdesivir standing out as a critical intervention for coronavirus treatments. At the heart of manufacturing this complex nucleoside analog lies the challenge of synthesizing its key intermediates efficiently and economically. Patent CN115703796A introduces a groundbreaking methodology for preparing Compound I, a pivotal precursor in the Remdesivir value chain. This innovation addresses long-standing bottlenecks in nucleoside chemistry by replacing unstable amino-substituted starting materials with robust methylthio-substituted heterocycles. For R&D directors and procurement strategists, this patent represents a significant leap forward, offering a pathway that not only improves chemical yields but also streamlines the supply chain for high-purity pharmaceutical intermediates. By fundamentally altering the order of functional group introduction, this technology mitigates side reactions that have historically plagued the production of this vital medicine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Remdesivir intermediates, as documented in earlier literature such as J. Med. Chem. 2017, relied heavily on the direct coupling of amino-substituted heterocycles with sugar moieties. This approach presented severe chemical limitations primarily due to the presence of active hydrogen atoms on the exocyclic amino group. These active hydrogens acted as interfering nucleophiles or acidic sites during the critical glycosylation and subsequent cyanation steps. Consequently, manufacturers faced consistently low yields in the initial coupling reactions, which created a cascading effect of inefficiency throughout the entire synthesis. The need to protect and deprotect these sensitive amino groups added unnecessary complexity, increased the number of unit operations, and introduced additional opportunities for impurity generation. For a reliable pharmaceutical intermediates supplier, these inefficiencies translate directly into higher production costs and longer lead times, making the conventional route less viable for meeting the surging global demand for antiviral medications.
The Novel Approach
The methodology disclosed in CN115703796A circumvents these historical challenges by employing a methylthio-substituted heterocycle, specifically Compound VI, as the starting nucleophile. This strategic substitution replaces the problematic amino group with a chemically inert methylthio group during the early, harsh reaction conditions. This change allows the nucleophilic addition to the ribonolactone (Compound VII) to proceed with exceptional efficiency, achieving yields upwards of 95% in the initial step. The methylthio group remains stable through the subsequent cyanation and debenzylation steps, ensuring that the core scaffold is built with high fidelity. Only in the final stage is the methylthio group displaced by an amino group via aminolysis. This "late-stage functionalization" strategy drastically simplifies the process flow, reduces the total number of purification steps, and significantly enhances the overall mass balance of the synthesis, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Stereoselective Nucleophilic Addition and Cyanation
The core of this synthetic breakthrough lies in the precise control of stereochemistry during the formation of the C-C bond between the heterocycle and the sugar ring. In the first step, a bulky strong base, such as lithium diisopropylamide (LDA) or lithium bis(trimethylsilyl)amide (LiHMDS), is utilized to deprotonate the C4 position of the methylthio-substituted pyrrolo-triazine at cryogenic temperatures ranging from -70°C to -78°C. This generates a highly reactive nucleophilic species that attacks the carbonyl carbon of the protected ribonolactone. The steric bulk of the base and the low temperature are critical for minimizing epimerization and ensuring the formation of the desired beta-anomer. Following the addition, the resulting hemiketal intermediate is immediately subjected to cyanation using a Lewis acid catalyst like boron trifluoride etherate or TMSOTf in the presence of trimethylsilyl cyanide (TMSCN). This step proceeds via an oxocarbenium ion intermediate, where the cyanide ion attacks from the less hindered face, dictated by the neighboring group participation of the sugar protecting groups, to install the quaternary nitrile center with high diastereoselectivity.
Furthermore, the impurity profile of this route is inherently cleaner due to the absence of reactive amino protons during the coupling phase. In conventional routes, the free amine can lead to oligomerization or N-alkylation byproducts that are structurally similar to the target molecule and difficult to remove. By masking the amine functionality as a thioether until the very end, the reaction mixture remains homogeneous and predictable. The subsequent debenzylation step utilizes a dual-catalyst system of palladium on carbon and palladium hydroxide on carbon under hydrogen pressure. This specific combination ensures complete removal of the benzyl protecting groups without reducing the sensitive nitrile or the heterocyclic ring system. The final aminolysis step, utilizing ammonium acetate or ammonia in methanol at elevated temperatures, cleanly displaces the methylthio group. This sequence ensures that the final Compound I meets stringent purity specifications required for downstream phosphorylation and drug substance manufacturing, providing a robust solution for producing high-purity Remdesivir intermediates.
How to Synthesize Remdesivir Intermediate Compound I Efficiently
The synthesis of Compound I is achieved through a logical five-step sequence that prioritizes yield and operational simplicity. The process begins with the cryogenic lithiation of the heterocyclic base followed by coupling with the sugar lactone, establishing the core nucleoside skeleton. Subsequent steps involve the installation of the nitrile group, removal of benzyl protecting groups, protection of the diol system as an acetonide, and finally, the conversion of the thioether to the primary amine. Each step has been optimized to use common industrial solvents and reagents, facilitating easy technology transfer from the laboratory to the pilot plant. The detailed standardized synthesis steps for implementing this route are provided in the guide below.
- Perform nucleophilic addition of methylthio-substituted heterocycle VI to ribonolactone VII using a bulky strong base like LDA at cryogenic temperatures to form Compound V.
- Execute stereoselective cyanation of Compound V using Lewis acids such as BF3·Et2O and TMSCN to generate the nitrile intermediate Compound IV.
- Conduct catalytic hydrogenation for debenzylation to yield Compound III, followed by acetonide protection to form Compound II, and finalize with aminolysis to obtain Compound I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the substantial improvement in overall process yield. By avoiding the yield losses associated with the active amino group in early stages, the total amount of raw materials required to produce a kilogram of the final intermediate is significantly reduced. This efficiency gain directly correlates to a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market. Additionally, the use of commodity chemicals such as acetone for the protection step and standard hydrogenation catalysts reduces dependency on specialized, high-cost reagents. This simplification of the bill of materials enhances supply chain resilience, ensuring that production schedules are not disrupted by the scarcity of exotic chemicals.
- Cost Reduction in Manufacturing: The elimination of complex amino-protection and deprotection cycles in the early stages of synthesis removes several unit operations from the manufacturing timeline. This reduction in processing time lowers energy consumption and labor costs per batch. Moreover, the high yields reported in the patent examples, often exceeding 90% for individual steps, mean that less solvent is required for purification and less waste is generated for disposal. The economic impact of these efficiencies is profound, enabling manufacturers to achieve significant cost savings without compromising on the quality of the final product.
- Enhanced Supply Chain Reliability: The robustness of the methylthio-intermediate strategy ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and customer trust. Since the route relies on well-established chemical transformations such as nucleophilic addition, cyanation, and catalytic hydrogenation, it can be easily scaled up using existing infrastructure in most fine chemical facilities. This scalability reduces the lead time for high-purity pharmaceutical intermediates, allowing suppliers to respond rapidly to fluctuations in market demand. The stability of the intermediates also simplifies logistics, as they do not require extreme storage conditions, further reducing the risk of supply chain disruptions.
- Scalability and Environmental Compliance: From an environmental perspective, this route aligns well with green chemistry principles by maximizing atom economy and minimizing waste generation. The high conversion rates reduce the volume of mother liquors containing unreacted starting materials, thereby lowering the burden on wastewater treatment facilities. The use of acetone, a relatively benign solvent, for the acetonide protection step further improves the environmental profile of the process compared to routes using more hazardous reagents. These factors make the commercial scale-up of complex nucleoside analogs more sustainable and easier to permit, ensuring long-term viability for manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for potential partners and technical evaluators. Understanding these details is essential for assessing the feasibility of integrating this technology into existing production lines.
Q: Why is the methylthio-substituted route superior to the traditional amino-substituted route?
A: The traditional route utilizes an amino-substituted heterocycle which possesses active hydrogens that interfere with subsequent glycosylation and cyanation reactions, leading to significantly lower yields. The novel method employs a methylthio-substituted precursor which is chemically stable during these critical steps, allowing for much higher conversion rates and easier purification before the final aminolysis step.
Q: What are the key cost-saving factors in this new synthetic pathway?
A: This pathway eliminates the need for complex protecting group strategies on the heterocycle nitrogen early in the synthesis. Furthermore, the use of inexpensive reagents like acetone for the acetonide protection step and standard catalysts like Pd/C for deprotection reduces raw material costs. The overall higher yields across all five steps minimize waste disposal costs and maximize throughput per batch.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the industrial suitability of this route. The reaction conditions utilize standard solvents like THF, DCM, and methanol, and the workup procedures involve conventional techniques such as extraction, filtration, and silica gel chromatography. The robustness of the methylthio intermediate ensures consistent quality, which is critical for scaling up to metric ton quantities required for global antiviral supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Remdesivir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving antiviral medications. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN115703796A and is fully prepared to implement this advanced methodology for our global clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the pharmaceutical industry. Our state-of-the-art facilities are equipped with the necessary cryogenic reactors and hydrogenation units to execute this specific route safely and effectively, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality.
We invite procurement leaders and R&D directors to collaborate with us to leverage this innovative synthesis technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of Remdesivir intermediates that drives down costs and accelerates your time to market.
