Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Advanced Drug Discovery
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazine Compounds for Advanced Drug Discovery
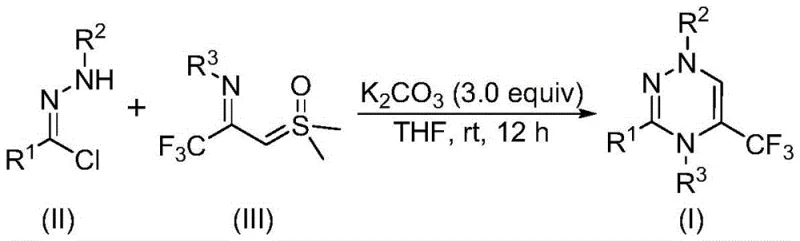
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic methodologies to access nitrogen-containing heterocycles with enhanced metabolic stability and bioavailability. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds, addressing critical bottlenecks in current manufacturing processes. This innovation leverages a synergistic [3+3] cycloaddition strategy between chlorohydrazones and trifluoroacetyl sulfur ylides, facilitated by inexpensive potassium carbonate. The significance of this technology lies in its ability to operate under mild, metal-free conditions while delivering high structural diversity, a key requirement for modern drug discovery campaigns targeting complex biological pathways.
The incorporation of trifluoromethyl groups into heterocyclic scaffolds is a proven strategy to modulate lipophilicity and electronegativity, directly impacting the pharmacokinetic profile of potential drug candidates. As illustrated in the biological context of related structures, these motifs are prevalent in potent inhibitors such as PI3Kα inhibitors and dual c-Met/VEGFR-2 inhibitors, underscoring the commercial value of efficient access to this chemical space.
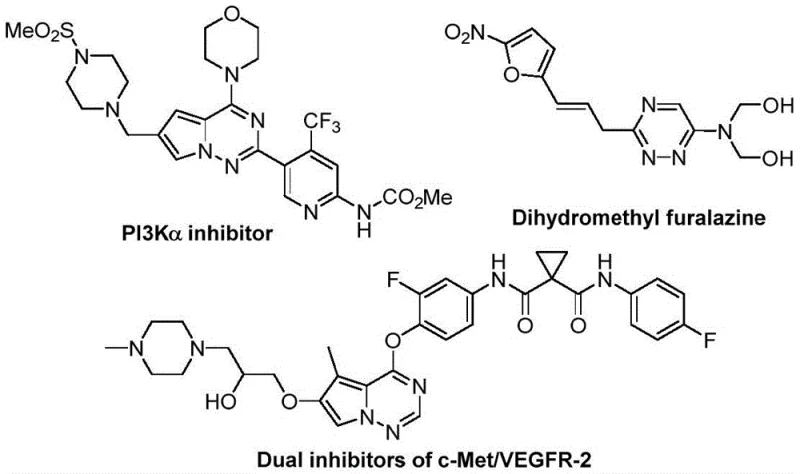
For R&D directors and process chemists, the ability to synthesize these cores without relying on precious metal catalysts represents a significant advancement in green chemistry and cost-effective process design. The method described in the patent not only simplifies the synthetic route but also ensures that the final products meet stringent purity specifications required for clinical applications, positioning it as a vital tool for reliable pharmaceutical intermediate supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2,4-triazine skeletons has relied on condensation reactions between amidrazones and 1,2-diketones or alkynes, often necessitating harsh reaction conditions and extended reaction times. Traditional multicomponent reactions involving hydrazides and dicarbonyl compounds frequently suffer from poor atom economy and limited substrate scope, restricting the chemical diversity accessible to medicinal chemists. Furthermore, many existing protocols require the use of transition metal catalysts or strong bases that generate significant amounts of hazardous waste, complicating downstream purification and increasing the environmental footprint of the manufacturing process. These conventional approaches often struggle with low reaction efficiency and the formation of difficult-to-remove impurities, which can severely impact the overall yield and quality of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN116253692A utilizes a novel cyclization strategy that bypasses the need for expensive catalysts and extreme conditions. By employing chlorohydrazone and trifluoroacetyl sulfur ylide as key building blocks, the reaction proceeds smoothly in common organic solvents like tetrahydrofuran at ambient temperatures. This approach eliminates the necessity for inert atmosphere protection, allowing the reaction to be conducted in air, which drastically simplifies operational complexity and equipment requirements. The use of potassium carbonate as a mild, non-toxic promoter ensures that the reaction environment remains benign, facilitating easier workup procedures and reducing the burden on waste treatment facilities. This novel route offers superior structural flexibility, enabling the rapid generation of diverse libraries of trifluoromethyl-substituted triazines for high-throughput screening.
Mechanistic Insights into Potassium Carbonate-Promoted Cycloaddition
The mechanistic pathway of this transformation involves a sophisticated sequence of elementary steps initiated by the deprotonation of the chlorohydrazone species. Under the promotion of potassium carbonate, the chlorohydrazone undergoes elimination of hydrogen chloride to generate a highly reactive nitrile imine intermediate in situ. This transient species acts as a 1,3-dipole, engaging in a concerted [3+3] cycloaddition reaction with the trifluoroacetyl sulfur ylide, which serves as the three-atom component. The synergy between these two precursors leads to the formation of the six-membered 1,2,4-triazine ring system with the concomitant extrusion of dimethyl sulfoxide as a byproduct. Alternatively, the reaction may proceed through a stepwise mechanism involving intermolecular nucleophilic addition followed by intramolecular nucleophilic substitution, ultimately converging on the same thermodynamic product. This mechanistic understanding is crucial for optimizing reaction parameters and predicting the behavior of sterically hindered or electronically diverse substrates.
From an impurity control perspective, the absence of transition metals eliminates the risk of metal-catalyzed side reactions such as homocoupling or over-oxidation, which are common pitfalls in traditional heterocycle synthesis. The mild basicity of potassium carbonate minimizes the degradation of sensitive functional groups, ensuring that the integrity of complex substituents is maintained throughout the cyclization process. The clean conversion profile observed in the patent examples suggests that the formation of regioisomers or polymeric byproducts is negligible, resulting in a crude reaction mixture that is amenable to straightforward purification. This high level of chemoselectivity is particularly advantageous for the synthesis of advanced intermediates where protecting group strategies add unnecessary cost and time to the overall synthetic sequence.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
The practical implementation of this synthesis protocol is designed for ease of execution in both laboratory and pilot plant settings. The procedure involves simply combining the stoichiometric amounts of chlorohydrazone, trifluoroacetyl sulfur ylide, and potassium carbonate in a suitable solvent, followed by stirring at room temperature. The robustness of the reaction allows for a wide range of substrate combinations without the need for rigorous exclusion of moisture or oxygen, making it an ideal candidate for process intensification. Detailed standardized synthesis steps for specific derivatives are outlined below to ensure reproducibility and consistency across different batches.
- Combine potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide in an organic solvent such as THF.
- Stir the reaction mixture at room temperature (20-40°C) under an air atmosphere for 10 to 14 hours.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free synthesis route offers tangible benefits in terms of cost structure and logistical reliability. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply chain risks associated with the fluctuating prices of rare earth elements. Furthermore, the use of commodity chemicals like potassium carbonate and readily available organic solvents ensures a stable and secure supply of raw materials, reducing the likelihood of production delays due to material shortages. The simplified operational requirements, such as the ability to run reactions in air at room temperature, lower the capital expenditure needed for specialized reactor infrastructure, thereby improving the overall return on investment for manufacturing facilities.
- Cost Reduction in Manufacturing: The removal of heavy metal catalysts from the process flow significantly reduces the cost associated with catalyst procurement and, more importantly, the extensive purification steps required to meet regulatory limits on metal residues. By utilizing inexpensive inorganic salts as promoters, the direct material costs are drastically lowered, leading to substantial cost savings in API manufacturing. The simplified workup procedure, which often requires only filtration and standard chromatography, minimizes solvent consumption and labor hours, further enhancing the economic viability of the process on a commercial scale.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including acyl chlorides, hydrazines, and aromatic amines, are widely available from multiple global suppliers, ensuring a resilient supply chain that is not dependent on single-source vendors. The robustness of the reaction conditions means that production can be maintained consistently without the need for highly specialized technical expertise or fragile reaction environments. This reliability translates to shorter lead times for high-purity pharmaceutical intermediates, allowing downstream customers to maintain leaner inventory levels and respond more agilely to market demands.
- Scalability and Environmental Compliance: The inherent safety of operating at ambient temperatures and pressures makes this process highly scalable from gram to multi-ton quantities without significant re-engineering of the reaction parameters. The absence of toxic heavy metals and the use of benign reagents align with increasingly stringent environmental regulations, reducing the compliance burden and waste disposal costs for manufacturing sites. This green chemistry approach not only improves the corporate sustainability profile but also future-proofs the manufacturing process against evolving regulatory landscapes regarding chemical safety and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazine synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance for industry stakeholders.
Q: Does this synthesis method require heavy metal catalysts?
A: No, the process utilizes potassium carbonate as a promoter, completely eliminating the need for expensive and toxic transition metal catalysts, which simplifies purification and reduces heavy metal residue risks in the final API.
Q: What are the optimal reaction conditions for scalability?
A: The reaction proceeds efficiently at room temperature (20-40°C) under an air atmosphere for 10-14 hours, making it highly suitable for large-scale production without the need for specialized inert gas equipment or cryogenic cooling.
Q: What is the substrate scope for this triazine synthesis?
A: The method demonstrates broad functional group tolerance, accommodating various substituted phenyl, naphthyl, and alkyl groups on the chlorohydrazone and sulfur ylide components, allowing for diverse structural modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in handling fluorinated compounds and nitrogen heterocycles allows us to optimize this specific patent technology for maximum yield and minimal impurity formation.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free protocol. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your unique project requirements, ensuring a collaborative approach to overcoming your most challenging synthetic hurdles.
