Scalable Synthesis of Dihydroquercetin via THP Protection for Industrial Pharmaceutical Applications
Scalable Synthesis of Dihydroquercetin via THP Protection for Industrial Pharmaceutical Applications
The global demand for high-purity flavonoids, particularly dihydroquercetin (also known as taxifolin), has surged due to its potent antioxidant properties and therapeutic potential in treating cardiovascular and metabolic disorders. As a reliable dihydroquercetin supplier, understanding the nuances of its production is critical for meeting pharmaceutical grade standards. Patent CN102070592B introduces a transformative synthetic methodology that addresses the historical bottlenecks of low yield and hazardous reagents associated with conventional production. This technical insight report analyzes the patented convergent synthesis route, which leverages tetrahydropyranyl (THP) ether protection to achieve a robust, scalable, and environmentally compliant manufacturing process suitable for the rigorous demands of the fine chemical industry.
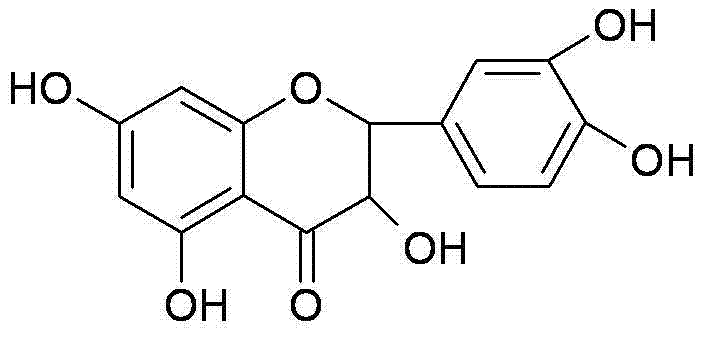
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of dihydroquercetin relied heavily on extraction from natural sources such as larch wood or Engelhardia roxburghiana leaves, a process inherently plagued by low content levels in biomass and massive solvent waste, rendering it economically unviable for large-scale industrial production. Alternative chemical synthesis routes often employed chloromethyl methyl ether (MOM-Cl) for hydroxyl protection, a reagent classified as a strong carcinogen that necessitates expensive containment systems and complex waste treatment protocols, thereby drastically inflating operational costs. Furthermore, classical routes frequently utilized expensive starting materials like pre-protected 2,4,6-trihydroxyacetophenone, which limits the ability to achieve cost reduction in pharmaceutical intermediates manufacturing. Other methods involving sharpless reagents or photochemical extensions introduce exotic catalysts like ruthenium salts that are not only costly but also difficult to source consistently, creating supply chain vulnerabilities for commercial scale-up of complex flavonoid intermediates.
The Novel Approach
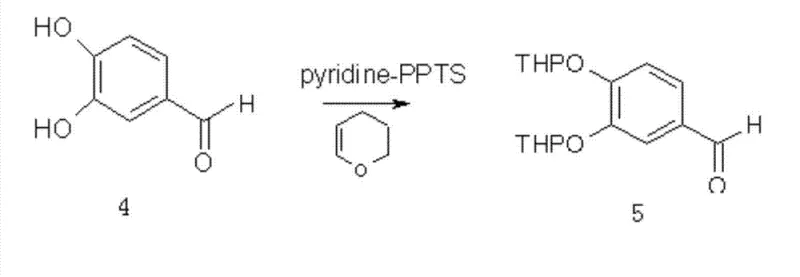
The patented methodology outlined in CN102070592B circumvents these issues by initiating synthesis from commodity chemicals, specifically anhydrous phloroglucinol and acetonitrile, via a classic Hoesch reaction to generate the acetophenone core in situ. A pivotal innovation lies in the substitution of hazardous MOM groups with THP ethers using 3,4-dihydropyran and pyridinium p-toluenesulfonate (PPTS), which offers superior stability during subsequent base-catalyzed condensations while being easily removable under mild acidic conditions. This route strategically merges the final deprotection and cyclization steps into a single unit operation, significantly shortening the production timeline and reducing solvent inventory requirements. By optimizing reaction conditions such as temperature ranges of 20-50°C for protection and utilizing aqueous hydrogen peroxide for epoxidation, the process ensures high atom economy and minimizes the generation of toxic byproducts, aligning perfectly with modern green chemistry principles.
Mechanistic Insights into THP-Protection and Convergent Cyclization
The core of this synthesis relies on a convergent strategy where two key fragments, the acetophenone derivative and the benzaldehyde derivative, are independently protected before coupling. The mechanism begins with the acid-catalyzed formation of the oxocarbenium ion from 3,4-dihydropyran, which is then attacked by the phenolic hydroxyls of the substrates to form stable THP ethers. This protection is crucial as it prevents unwanted side reactions at the ortho- and para- positions during the subsequent aldol condensation. The condensation itself proceeds via a Claisen-Schmidt mechanism where the alpha-methyl group of the protected acetophenone is deprotonated by a base such as sodium hydroxide (10%-40% concentration) to form an enolate, which attacks the carbonyl carbon of the protected aldehyde to yield the chalcone intermediate with high stereoselectivity.
Following chalcone formation, the introduction of the oxygen bridge is achieved through epoxidation using hydrogen peroxide in an alcoholic solvent under basic conditions, generating an epoxy chalcone intermediate. The final transformation is a cascade reaction triggered by an acidic catalyst like p-toluenesulfonic acid in anhydrous methanol. In this step, the THP protecting groups are hydrolyzed to regenerate the free phenols, while simultaneously, the intramolecular nucleophilic attack of the A-ring phenol on the epoxide occurs, closing the C-ring to form the dihydroflavonol skeleton. This tandem deprotection-cyclization mechanism is highly efficient, minimizing the isolation of unstable intermediates and ensuring that the final product, dihydroquercetin, is obtained with a purity specification exceeding 95% after simple recrystallization, demonstrating the robustness of the chemical design.
How to Synthesize Dihydroquercetin Efficiently
The synthesis of dihydroquercetin via this patented route involves five distinct operational stages that transform simple raw materials into a high-value flavonoid. The process begins with the preparation of 2,4,6-trihydroxyacetophenone followed by dual protection of both the acetophenone and 3,4-dihydroxybenzaldehyde fragments. These protected intermediates are then condensed to form a chalcone, which is subsequently epoxidized and finally cyclized. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and temperature profiles required for GMP-compliant manufacturing, are outlined below for technical reference.
- Prepare 2,4,6-trihydroxyacetophenone via Hoesch reaction using phloroglucinol and acetonitrile with ZnCl2/HCl.
- Protect hydroxyl groups of the acetophenone and 3,4-dihydroxybenzaldehyde using 3,4-dihydropyran (THP) with PPTS catalyst.
- Perform Claisen-Schmidt condensation between the protected intermediates to form the chalcone derivative.
- Oxidize the chalcone using hydrogen peroxide (H2O2) under basic conditions to generate the epoxy chalcone.
- Execute simultaneous deprotection and cyclization using an acidic catalyst (e.g., p-toluenesulfonic acid) in methanol to yield dihydroquercetin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this synthetic route offers profound strategic benefits beyond mere technical feasibility. By shifting away from extraction-dependent models, manufacturers can decouple production volumes from agricultural harvest cycles and climatic variables, ensuring a consistent and predictable supply of raw materials throughout the fiscal year. The utilization of bulk commodity chemicals like phloroglucinol and acetonitrile, rather than specialized fine chemical building blocks, significantly mitigates the risk of price volatility and supplier monopolies, thereby enhancing the overall resilience of the supply chain against market fluctuations.
- Cost Reduction in Manufacturing: The elimination of carcinogenic reagents like chloromethyl methyl ether removes the need for specialized hazardous material handling infrastructure and expensive waste disposal protocols, leading to direct operational expenditure savings. Additionally, the convergence of the deprotection and ring-closing steps reduces the total number of unit operations, which lowers energy consumption, labor hours, and solvent recovery costs, resulting in a leaner and more cost-effective production profile without compromising yield.
- Enhanced Supply Chain Reliability: Since the synthesis relies on widely available industrial solvents such as methylene chloride, methanol, and ethanol, along with common catalysts like zinc chloride and p-toluenesulfonic acid, the risk of raw material stockouts is minimized. This accessibility allows for flexible sourcing strategies where multiple vendors can be qualified for key inputs, reducing lead time for high-purity dihydroquercetin batches and preventing production stoppages due to single-source dependencies.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory glassware to multi-ton reactor systems without requiring exotic high-pressure or cryogenic equipment. The reduced solvent usage and the avoidance of heavy metal catalysts simplify wastewater treatment and environmental compliance reporting, facilitating faster regulatory approvals and smoother audits in strictly regulated pharmaceutical markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process safety, purity control, and scalability for potential partners evaluating this technology for their supply chains.
Q: Why is the THP protection method superior to traditional MOM protection for dihydroquercetin synthesis?
A: Traditional methods often utilize chloromethyl methyl ether (MOM-Cl), a potent carcinogen that poses severe safety and regulatory risks in large-scale manufacturing. The patented THP (tetrahydropyranyl) protection route employs 3,4-dihydropyran and mild acid catalysts like PPTS, eliminating carcinogenic reagents while maintaining high stability during the aldol condensation and epoxidation steps.
Q: How does this synthetic route improve cost efficiency compared to plant extraction?
A: Plant extraction suffers from low natural abundance of dihydroquercetin and significant solvent waste. This synthetic approach utilizes inexpensive, commodity-grade starting materials like phloroglucinol and acetonitrile. Furthermore, merging the deprotection and cyclization steps reduces unit operations, solvent consumption, and overall processing time, leading to substantial cost reductions.
Q: What are the critical quality control parameters for the epoxy chalcone intermediate?
A: The formation of the epoxy chalcone via hydrogen peroxide oxidation requires precise temperature control (0-40°C) and stoichiometric balance to prevent over-oxidation or ring opening prior to cyclization. TLC monitoring is essential to ensure complete conversion before proceeding to the final acid-catalyzed ring closure, which dictates the final purity and optical activity of the dihydroquercetin product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydroquercetin Supplier
The synthesis method detailed in patent CN102070592B represents a significant leap forward in the manufacturing of flavonoid intermediates, offering a pathway to high-purity products with a reduced environmental footprint. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial realities. Our facility is equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of dihydroquercetin meets the exacting standards required for pharmaceutical and nutraceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic route for your product pipeline. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce overall procurement costs.
