Advanced Semi-Synthesis of Dihydroquercetin: A Strategic Breakthrough for Global Pharmaceutical Supply Chains
Advanced Semi-Synthesis of Dihydroquercetin: A Strategic Breakthrough for Global Pharmaceutical Supply Chains
The global demand for high-purity flavonoids, particularly dihydroquercetin (also known as taxifolin), has surged due to its potent antioxidant and anti-inflammatory properties applicable in both pharmaceutical and nutraceutical sectors. Traditionally sourced through extraction from natural plants like Larix gmelinii, the supply chain has been plagued by seasonal variability, low yields, and extensive solvent waste. However, a pivotal technological shift is emerging through semi-synthetic routes that offer superior consistency and scalability. Specifically, the methodology disclosed in patent CN113185485A introduces a robust semi-synthesis method utilizing thiourea dioxide as a key reducing agent. This innovation addresses critical bottlenecks in pharmaceutical intermediates manufacturing by replacing hazardous reagents with safer, more efficient alternatives, thereby establishing a new benchmark for a reliable pharmaceutical intermediates supplier seeking to optimize their production portfolios.
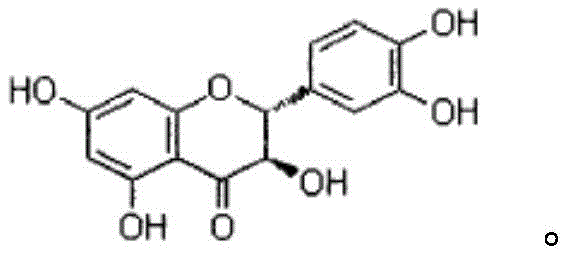
Dihydroquercetin acts as a hydride of quercetin, possessing both lipid and water solubility which enhances its bioavailability compared to its precursor. The structural integrity of the molecule, characterized by its specific hydroxylation pattern on the A, B, and C rings, is crucial for its biological activity. The challenge in synthesizing this molecule lies in the selective reduction of the C2-C3 double bond in the flavonol C-ring without affecting the sensitive phenolic hydroxyl groups. Conventional extraction methods often fail to meet the stringent purity requirements of modern drug development, leading to batch-to-batch inconsistencies. The semi-synthetic approach detailed in the referenced patent overcomes these limitations by leveraging precise chemical control, ensuring that the final product meets the rigorous specifications required for high-purity pharmaceutical intermediates intended for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of dihydroquercetin has relied heavily on phytochemical extraction, a process fraught with inefficiencies that strain supply chain reliability. The natural content of dihydroquercetin in plant sources is inherently low, necessitating the processing of massive quantities of biomass to yield negligible amounts of the target compound. This not only drives up the cost reduction in API manufacturing efforts but also creates significant environmental burdens due to the vast volumes of organic solvents required for extraction and separation. Furthermore, chemical synthesis via total synthesis routes, while capable of large-scale production, involves lengthy multi-step sequences that utilize toxic reagents and generate substantial hazardous waste. Even existing semi-synthetic methods using quercetin as a starting material have faced hurdles; many rely on sodium hydrosulfite-based reducing agents which are classified as dangerous goods, posing safety risks during transportation and storage, and often require complex binary reagent systems that are economically inefficient and difficult to control on an industrial scale.
The Novel Approach
The innovative method described in patent CN113185485A fundamentally reengineers the reduction step by employing thiourea dioxide in an alkaline aqueous environment. This approach capitalizes on the unique solubility characteristics of quercetin, which dissolves readily in alkaline water but is insoluble in neutral or acidic media. By conducting the reduction in this homogeneous alkaline phase, the reaction kinetics are significantly enhanced, allowing for a single-step conversion of quercetin dihydrate to dihydroquercetin. The use of thiourea dioxide is particularly strategic; unlike traditional reducing agents, it decomposes under alkaline heating to generate sulfenic acid, a species with a high reduction potential capable of selectively targeting the C-ring double bond. This eliminates the need for hazardous binary combinations and simplifies the downstream processing, directly contributing to cost reduction in fine chemical manufacturing by streamlining the operational workflow and minimizing safety compliance overheads.
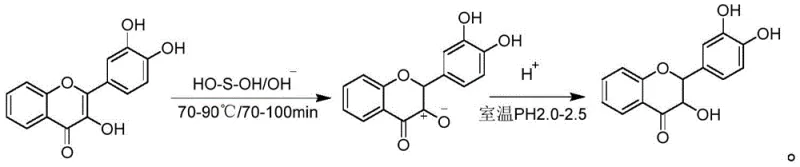
Mechanistic Insights into Thiourea Dioxide-Mediated Alkaline Reduction
The core of this technological advancement lies in the precise mechanistic interaction between the flavonol substrate and the in situ generated reducing species. When thiourea dioxide is introduced into the alkaline reaction medium at elevated temperatures (typically 70-90°C), it undergoes a rapid decomposition to form urea and sulfenic acid (H-O-S-O-H). This sulfenic acid acts as a potent hydride donor with a reduction potential ranging from -1000 to -1200 mV, which is superior to that of sodium sulfite. The reaction is meticulously controlled by maintaining the pH within a narrow window of 9.5 to 11.5, ensuring that the quercetin remains in its soluble phenolate form while maximizing the reactivity of the reducing agent. To prevent the oxidative degradation of the sensitive phenolic groups, the process mandates the use of inert gas shielding, such as nitrogen or argon, throughout the heating cycle. This protective atmosphere is critical for suppressing the formation of quinone-like by-products, thereby preserving the structural fidelity of the dihydroflavonol skeleton.
Impurity control is further engineered through a sophisticated two-stage addition protocol for the reducing agent. By adding approximately two-thirds to four-fifths of the thiourea dioxide initially at a lower temperature range (70-85°C) and the remainder at a slightly higher temperature (85-95°C), the reaction maintains a consistent reducing power without overwhelming the system with excess reagent that could lead to side reactions. Following the reduction, the reaction mixture is diluted and acidified to a pH of 2.0-2.5. This acidification step serves a dual purpose: it triggers the ring-closing stabilization of the dihydroquercetin and simultaneously precipitates any unreacted quercetin dihydrate, which is insoluble at this acidity. This clever physicochemical separation allows for the filtration and recycling of the starting material, ensuring that the subsequent extraction and crystallization steps deal with a significantly cleaner crude matrix, ultimately facilitating the production of commercial scale-up of complex pharmaceutical intermediates with minimal impurity burden.
How to Synthesize Dihydroquercetin Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating this high-efficiency transformation in a GMP-compliant environment. The process begins with the preparation of an alkaline aqueous solution, followed by the dissolution of quercetin dihydrate and the controlled addition of thiourea dioxide under inert gas. The reaction proceeds through defined temperature stages to ensure complete conversion, after which the mixture is cooled and acidified to isolate the product. The crude material is then subjected to a rigorous purification regimen involving solvent extraction and dual-step recrystallization. For R&D teams looking to implement this technology, the detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and optimal yield.
- Dissolve quercetin dihydrate in alkaline water (pH 9.5-11.5) and heat to 70-90°C under inert gas protection.
- Add thiourea dioxide reducing agent in two stages to ensure complete reduction of the C-ring double bond.
- Acidify the reaction mixture to pH 2.0-2.5 to precipitate unreacted starting material, then extract and recrystallize the filtrate to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this thiourea dioxide-based semi-synthesis route offers transformative advantages that extend beyond mere technical feasibility. The primary value proposition lies in the drastic simplification of the raw material supply chain. By replacing hazardous, regulated chemicals like sodium hydrosulfite with thiourea dioxide—a stable, non-hazardous solid—the logistical complexities and costs associated with the storage and transport of dangerous goods are effectively eliminated. This stability ensures a continuous supply of reagents without the risk of degradation or regulatory hold-ups, directly enhancing supply chain reliability for long-term production contracts. Furthermore, the ability to recover and recycle unreacted quercetin from the acidification filter cake creates a closed-loop material flow, significantly reducing the net consumption of the expensive starting material and driving down the overall cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive transition metal catalysts and the reduction of solvent usage. Since the reaction occurs in water, the need for large volumes of organic solvents during the reaction phase is removed, lowering both material costs and waste disposal fees. The simplified workup procedure, which avoids complex chromatographic separations in favor of crystallization, reduces labor hours and equipment occupancy time. Additionally, the high selectivity of the thiourea dioxide reduction minimizes the formation of difficult-to-remove impurities, which reduces the number of recrystallization cycles needed to reach pharmacopeial standards, thereby increasing overall throughput and yield efficiency.
- Enhanced Supply Chain Reliability: Sourcing dihydroquercetin from plant extraction is inherently vulnerable to agricultural variables such as weather, harvest seasons, and geopolitical instability in sourcing regions. In contrast, this semi-synthetic route relies on quercetin, which is widely available and can be produced consistently year-round. The use of stable, shelf-stable reagents like thiourea dioxide further mitigates the risk of supply disruptions caused by the expiration or degradation of sensitive chemicals. This predictability allows supply chain planners to forecast production schedules with greater accuracy, reducing lead time for high-purity pharmaceutical intermediates and ensuring that downstream drug manufacturers receive their materials on time, every time.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method represents a significant improvement over traditional processes. The shift to an aqueous reaction medium drastically reduces the emission of volatile organic compounds (VOCs), aligning with increasingly stringent global environmental regulations. The waste stream is primarily aqueous and contains biodegradable by-products like urea, simplifying wastewater treatment requirements. The process is inherently scalable, as demonstrated by the successful enlargement of reaction batches in the patent examples without loss of efficiency, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates from pilot plant to multi-ton annual production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this semi-synthetic technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: Why is thiourea dioxide preferred over sodium hydrosulfite for this reduction?
A: Thiourea dioxide is significantly more stable during storage and transportation compared to sodium hydrosulfite, which is classified as a dangerous good. Furthermore, thiourea dioxide generates sulfenic acid in situ under alkaline conditions, providing a higher reduction potential that minimizes by-product formation without requiring complex binary reagent systems.
Q: How does this method improve the purity profile of dihydroquercetin?
A: The process utilizes a specific two-stage temperature and feeding protocol combined with inert gas shielding to suppress oxidation side reactions. Subsequent acidification allows for the physical removal of unreacted quercetin before extraction, while a dual-step recrystallization in alcohol-water solvents ensures final purity levels exceeding 99% with minimal impurity carryover.
Q: Is this process scalable for industrial production?
A: Yes, the method relies on water as the primary reaction solvent and uses commercially available, low-cost reagents. The elimination of hazardous organic solvents during the reaction phase and the ability to recycle unreacted starting material make it highly suitable for large-scale commercial manufacturing with reduced environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydroquercetin Supplier
As the pharmaceutical industry continues to demand higher purity and more sustainable sourcing for active ingredients, the technology described in patent CN113185485A represents a critical evolution in flavonoid manufacturing. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this advanced semi-synthesis method to market. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of dihydroquercetin we produce meets the exacting standards required for global regulatory submissions. We understand that consistency is key, and our process controls are designed to deliver batch-to-batch uniformity that natural extraction simply cannot match.
We invite procurement leaders and R&D directors to engage with us to explore how this optimized synthesis route can benefit your specific product pipeline. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this technology can lower your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that secure your supply chain and enhance your competitive advantage in the marketplace.
