Breakthrough in Asymmetric Alpha-Difluoromethylation: Scalable PTC Technology for Complex Pharmaceutical Intermediates
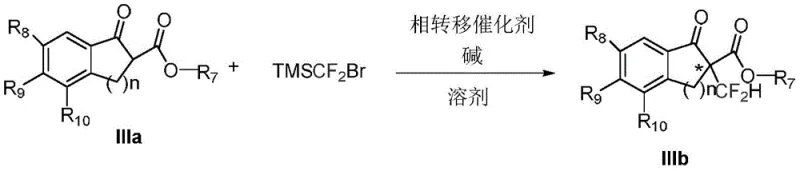
The pharmaceutical and agrochemical industries are increasingly reliant on fluorine-containing motifs to enhance the metabolic stability and lipophilicity of drug candidates. Patent CN110872232B introduces a groundbreaking methodology for the asymmetric alpha-difluoromethylation of beta-keto esters, addressing a long-standing challenge in organic synthesis. This invention utilizes a specialized phase transfer catalysis (PTC) strategy employing cinchona alkaloid-derived quaternary ammonium salts to achieve high enantioselectivity without the need for transition metals. The core innovation lies in the direct introduction of the difluoromethyl group using TMSCF2Br under mild biphasic conditions, providing a robust pathway to chiral building blocks essential for modern drug discovery.
Traditionally, the synthesis of difluoromethylated compounds has relied heavily on transition metal catalysis or harsh electrophilic reagents, which often suffer from limitations such as high cost, toxicity, and difficult purification processes. Conventional methods frequently require expensive ligands and inert atmosphere conditions that complicate large-scale manufacturing. Furthermore, achieving high enantiomeric excess (ee) in direct difluoromethylation reactions has historically been elusive, often necessitating multi-step resolution procedures that drastically reduce overall yield. The reliance on heavy metals also introduces significant regulatory hurdles for API manufacturers who must adhere to strict limits on residual metal impurities, adding layers of complexity and cost to the downstream processing.
In stark contrast, the novel approach disclosed in this patent leverages the power of asymmetric phase transfer catalysis to overcome these barriers. By employing easily accessible cinchona alkaloid derivatives as chiral catalysts, the process operates under green and sustainable conditions. The reaction proceeds efficiently in a biphasic system comprising an organic solvent and an aqueous base, eliminating the need for stringent anhydrous conditions typically required by organometallic reagents. This method not only simplifies the operational procedure but also significantly enhances the safety profile of the synthesis. The ability to directly access optically active alpha-difluoromethyl-beta-keto esters with high selectivity represents a paradigm shift, offering a streamlined route that bypasses the need for protective group manipulations or post-synthetic resolutions.
Mechanistic Insights into Cinchona Alkaloid-Catalyzed Asymmetric Difluoromethylation
The success of this transformation hinges on the precise design of the chiral phase transfer catalyst, specifically the cinchonine derivative series labeled as Ia. These catalysts function by facilitating the transport of the deprotonated beta-keto ester anion from the aqueous phase into the organic phase, where it reacts with the difluorocarbene precursor generated from TMSCF2Br. The rigid chiral environment provided by the cinchona backbone dictates the facial selectivity of the nucleophilic attack, ensuring the formation of the desired enantiomer. Structural modifications on the catalyst, such as the substitution patterns on the benzyl group (R2-R6) and the quinoline ring (R1), play a critical role in tuning the steric and electronic properties to maximize enantioselectivity.
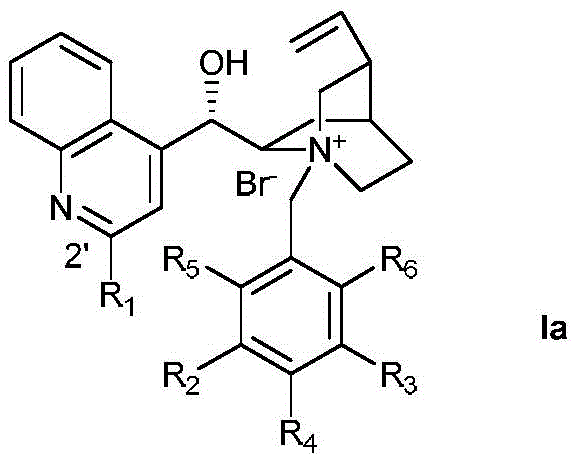
Impurity control is inherently managed through the high specificity of the catalytic cycle. The use of TMSCF2Br as the difluoromethylating agent minimizes side reactions commonly associated with more reactive difluorocarbene sources. The biphasic nature of the reaction allows for the continuous regeneration of the active catalyst species at the interface, maintaining a steady concentration of the reactive intermediate while suppressing non-selective background reactions. Experimental data indicates that optimizing the base strength and solvent polarity is crucial; for instance, the use of 30% aqueous potassium carbonate in a toluene/chloroform mixture was found to provide an optimal balance between reaction rate and stereocontrol. This mechanistic understanding allows for fine-tuning the process to accommodate a wide range of substrate electronic properties, ensuring consistent quality across different batches.
How to Synthesize Chiral Alpha-Difluoromethyl-Beta-Keto Esters Efficiently
The synthetic protocol outlined in the patent provides a clear roadmap for implementing this technology in a laboratory or pilot plant setting. The process begins with the preparation of the specific cinchona alkaloid catalyst, followed by the setup of the biphasic reaction system. Key parameters such as temperature control (-20°C to -5°C) and stirring efficiency are vital to maintain the integrity of the chiral induction. The following guide summarizes the standardized operational steps derived from the patent examples to ensure reproducibility and high yield.
- Prepare the reaction mixture by combining the beta-keto ester substrate, the chiral phase transfer catalyst (Ia series), and TMSCF2Br in a suitable organic solvent such as toluene or chloroform.
- Add an aqueous inorganic base solution, preferably 30% potassium carbonate, to the mixture and stir vigorously at low temperatures ranging from -40°C to 0°C to ensure high enantioselectivity.
- Monitor the reaction progress via TLC until completion, then separate the organic layer, dry the solvent, and purify the resulting chiral product through column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phase transfer catalysis technology offers substantial strategic benefits beyond mere chemical efficiency. The elimination of transition metals from the synthetic route translates directly into simplified purification workflows and reduced waste disposal costs. Traditional metal-catalyzed processes often require extensive scavenging steps to meet regulatory standards for residual metals, which adds time and expense to the manufacturing timeline. By switching to an organocatalytic PTC system, manufacturers can bypass these costly downstream operations, leading to a leaner and more cost-effective production process.
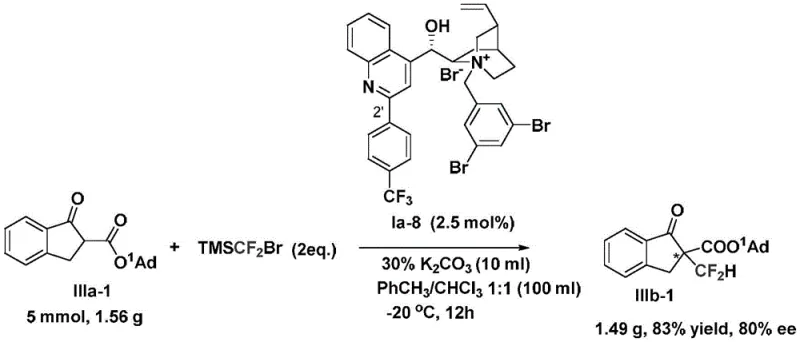
Supply chain reliability is further enhanced by the accessibility of the raw materials involved. Cinchona alkaloids are naturally occurring and commercially available in bulk quantities, reducing the risk of supply bottlenecks associated with specialized synthetic ligands. The patent explicitly demonstrates the scalability of this method through a gram-scale amplification experiment (Example 84), where the reaction maintained an 83% yield and 80% ee on a 5 mmol scale. This proof of concept suggests that the process is robust enough for commercial scale-up, mitigating the risks often associated with translating academic methodologies to industrial production. The ability to produce high-purity intermediates consistently ensures a stable supply for downstream API synthesis.
From an environmental compliance perspective, this method aligns perfectly with green chemistry principles. The use of aqueous bases and the avoidance of toxic heavy metals significantly reduce the environmental footprint of the manufacturing process. This not only simplifies regulatory compliance regarding hazardous waste but also enhances the sustainability profile of the final product, a factor that is increasingly important for global pharmaceutical partners. The operational simplicity of the biphasic system also means that existing reactor infrastructure can often be utilized without major modifications, facilitating rapid technology transfer and reducing capital expenditure requirements for new production lines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric difluoromethylation technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and limitations.
Q: What are the primary advantages of this phase transfer catalysis method over transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metals, utilizing cheap and readily available cinchona alkaloid derivatives instead. It operates under mild conditions and avoids heavy metal contamination, which is critical for pharmaceutical grade intermediates.
Q: Can this asymmetric difluoromethylation process be scaled up for industrial production?
A: Yes, the patent demonstrates successful gram-scale amplification (Example 84) with maintained high yield (83%) and enantioselectivity (80% ee), indicating strong potential for commercial scale-up without significant loss of efficiency.
Q: What is the scope of substrates compatible with this catalytic system?
A: The system exhibits broad substrate tolerance, successfully reacting with various beta-keto esters containing different substituents such as halogens, alkyl groups, and alkoxy groups on the aromatic ring, as well as different ester protecting groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phase Transfer Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this asymmetric difluoromethylation technology for the development of next-generation fluorinated pharmaceuticals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral intermediate delivered meets the highest industry standards for enantiomeric excess and chemical purity.
We invite you to collaborate with our technical team to explore how this innovative PTC method can optimize your specific synthesis route. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your project's unique requirements. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you accelerate your path to market with reliable, high-quality fluorinated intermediates.
