Advanced Chiral Quaternary Ammonium Catalysts for Scalable Pharmaceutical Intermediate Production
Advanced Chiral Quaternary Ammonium Catalysts for Scalable Pharmaceutical Intermediate Production
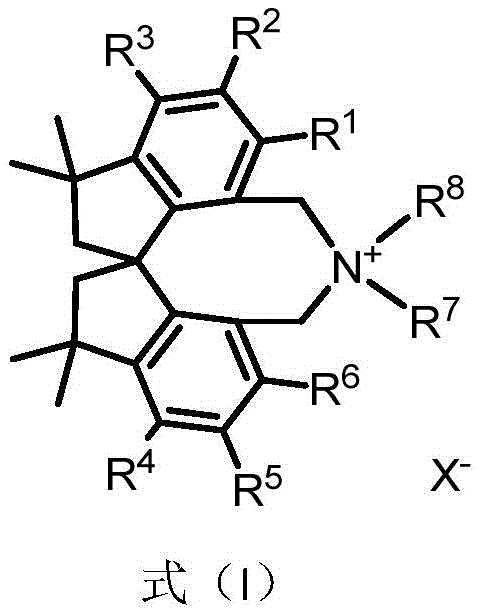
The field of asymmetric catalysis continues to evolve rapidly, driven by the relentless demand for optically active molecules in the pharmaceutical and agrochemical sectors. Patent CN112916042B introduces a significant breakthrough with the development of a novel chiral quaternary ammonium salt phase transfer catalyst based on a tetramethylspirodihydroindene skeleton. This innovation addresses critical limitations found in traditional catalyst systems, offering a robust platform for stereoselective synthesis. As the industry seeks more reliable pharmaceutical intermediate suppliers, the emergence of such structurally unique catalysts represents a pivotal shift towards more efficient and controllable manufacturing processes. The patent details not only the molecular architecture but also a practical preparation method, ensuring that this technology can be translated from the laboratory bench to commercial production lines with high fidelity.
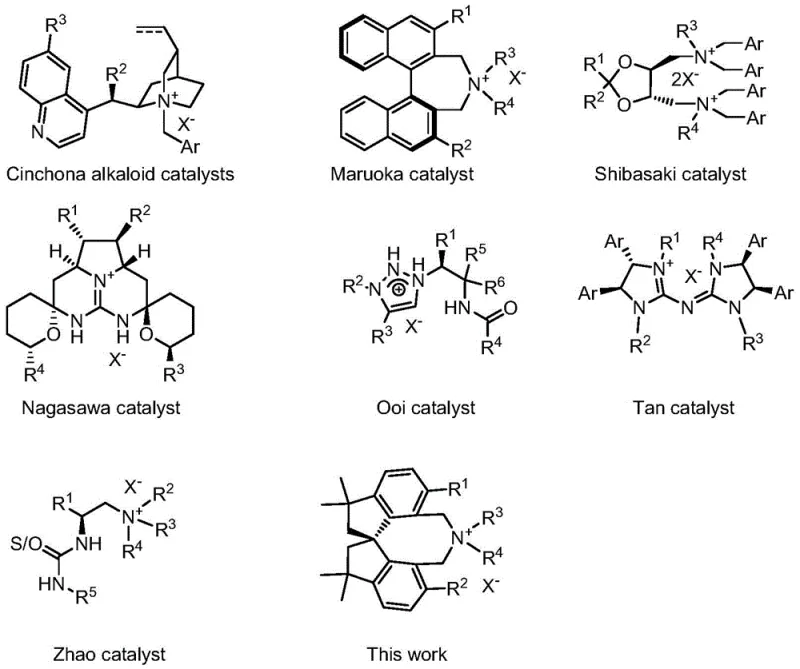
Historically, chiral phase transfer catalysis has relied heavily on derivatives of natural products like Cinchona alkaloids or axially chiral binaphthyl structures. While these have served the industry well, they often face challenges regarding substrate scope and stereoselectivity in complex transformations. The new catalyst described in CN112916042B leverages a rigid spiro-framework to create a highly defined chiral environment around the active quaternary ammonium center. This structural rigidity is key to achieving excellent enantioselectivity, particularly in challenging reactions such as the asymmetric alkylation of glycine Schiff bases. For R&D directors focused on purity and impurity profiles, this level of control is essential for minimizing downstream purification costs and ensuring regulatory compliance for final drug substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional chiral phase transfer catalysts, while effective in many scenarios, exhibit distinct weaknesses that can hinder large-scale manufacturing efficiency. For instance, chiral quaternary phosphonium salts, though potent, are notorious for their poor stability and tendency to deactivate under prolonged reaction conditions or elevated temperatures. Similarly, chiral crown ethers, another major class, often suffer from structural inflexibility that makes modification difficult, limiting their ability to be tuned for specific substrate classes. Furthermore, even the widely used Cinchona alkaloid-derived catalysts, despite their success in early industrial applications, sometimes fail to provide ideal stereoselectivities for newer, more complex reaction types such as intramolecular conjugate additions or aziridine desymmetrization. These limitations necessitate the screening of multiple catalyst generations, consuming valuable time and resources during process development.
The Novel Approach
The novel approach presented in this patent overcomes these hurdles by introducing a completely new skeletal framework based on tetramethylspirodihydroindene. This spiro-structure provides a unique three-dimensional arrangement that locks the conformation of the catalyst, thereby enhancing the transmission of chiral information to the reacting substrates. Unlike the flexible chains found in some amino acid-derived catalysts, this rigid backbone minimizes non-productive conformations that lead to racemic byproducts. The design allows for extensive modular variation at the aryl positions (R1-R6) and the nitrogen substituents (R7-R8), enabling chemists to fine-tune the steric and electronic properties for specific cost reduction in API manufacturing scenarios. This adaptability makes it a superior choice for developing robust processes that can withstand the rigors of industrial scale-up.
Mechanistic Insights into Asymmetric Phase Transfer Catalysis
The mechanism of action for this chiral quaternary ammonium salt involves the formation of a tight ion pair between the chiral cation and the enolate anion generated from the glycine Schiff base. In the organic phase, the bulky spiro-framework shields one face of the enolate, forcing the electrophile to approach from the less hindered side. This facial discrimination is the fundamental driver of high enantiomeric excess (ee). The rigidity of the tetramethylspirodihydroindene core ensures that this chiral pocket remains consistent throughout the catalytic cycle, preventing the loss of stereochemical integrity that can occur with more flexible catalysts. Understanding this mechanistic nuance is vital for R&D teams aiming to optimize reaction conditions for maximum yield and selectivity.
Furthermore, the impurity control mechanism is inherently built into the catalyst's design. By enforcing a single dominant transition state geometry, the formation of diastereomeric impurities is significantly suppressed. This is particularly important in the synthesis of pharmaceutical intermediates where strict limits on chiral impurities are enforced by regulatory bodies. The phase transfer nature of the catalyst also allows the reaction to proceed under mild basic conditions, often using solid bases like cesium hydroxide or potassium carbonate, which minimizes the risk of base-sensitive functional group degradation. This compatibility with sensitive substrates expands the utility of the catalyst beyond simple alkylations to more complex cascade reactions, providing a versatile tool for modern organic synthesis.
How to Synthesize Chiral Quaternary Ammonium Salt Efficiently
The synthesis of this advanced catalyst is designed for practicality and scalability, utilizing well-established organic transformations that are familiar to process chemists. The route begins with a readily available chiral spiro-dialdehyde precursor, which undergoes a double Suzuki-Miyaura coupling to install the necessary aryl groups. This is followed by a reduction-bromination sequence to convert the aldehydes into reactive benzyl bromide handles. Finally, quaternization with a secondary amine yields the target catalyst. This linear sequence avoids protecting group manipulations and uses commercially available reagents, streamlining the production workflow. Detailed standardized synthesis steps see the guide below.
- Perform Suzuki coupling of the bis-triflate spiro precursor with aryl boronic acid using a palladium catalyst to install the aryl groups.
- Reduce the dialdehyde intermediate with sodium borohydride followed by bromination with HBr in acetic acid to generate the dibromide species.
- React the dibromide with a secondary amine in the presence of a base to form the final chiral quaternary ammonium salt catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this new catalyst technology offers tangible benefits regarding cost stability and supply security. The synthetic route relies on commodity chemicals such as aryl boronic acids, palladium catalysts, and common amines, all of which are available from multiple global suppliers. This diversification of raw material sources mitigates the risk of supply chain disruptions that often plague specialized reagent markets. Additionally, the high catalytic efficiency means that lower loadings can be used to achieve desired conversion rates, directly reducing the cost per kilogram of the final product. The ability to operate under mild conditions also translates to lower energy consumption and reduced wear on reactor equipment, contributing to overall operational expenditure savings.
- Cost Reduction in Manufacturing: The elimination of expensive and unstable catalyst precursors significantly lowers the input cost profile. By utilizing a robust spiro-framework that maintains activity over longer periods, the frequency of catalyst replenishment is reduced. Furthermore, the high selectivity minimizes the formation of waste byproducts, reducing the burden on waste treatment facilities and lowering the environmental compliance costs associated with hazardous waste disposal. The process avoids the use of cryogenic conditions often required by organolithium or Grignard reagents, allowing reactions to run at manageable temperatures that are cheaper to maintain in large-scale reactors.
- Enhanced Supply Chain Reliability: The starting materials for this catalyst, particularly the spiro-indane backbone and various aryl boronic acids, are produced by established chemical manufacturers with proven track records. This ensures a steady flow of inputs without the bottlenecks associated with exotic natural product extraction. The synthetic steps are robust and tolerant to minor variations in reagent quality, which enhances the reliability of batch-to-batch production. For supply chain planners, this predictability is crucial for maintaining inventory levels and meeting delivery commitments to downstream pharmaceutical clients without unexpected delays.
- Scalability and Environmental Compliance: The reaction conditions described in the patent, such as the use of acetonitrile and chloroform mixtures or DMF, are standard solvents in the fine chemical industry with established recovery and recycling protocols. The absence of heavy metal contaminants in the final catalyst structure (after purification) simplifies the purification of the API intermediate, aligning with strict ICH guidelines on elemental impurities. The scalability is further supported by the exothermic nature of the quaternization step being manageable, allowing for safe operation in large vessels. This ease of scale-up ensures that the technology can grow with the demand of the drug product from clinical trials to commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this novel catalyst system. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation. Understanding these aspects helps technical teams evaluate the fit of this catalyst for their specific process needs.
Q: What are the stability advantages of this spiro-framework catalyst compared to Cinchona alkaloids?
A: Unlike chiral crown ethers which are difficult to modify or phosphonium salts which suffer from poor stability, this tetramethylspirodihydroindene-based quaternary ammonium salt offers superior thermal and chemical stability while maintaining a rigid chiral environment for high stereoselectivity.
Q: Can this catalyst be used for reactions other than glycine alkylation?
A: Yes, the patent indicates broad applicability for asymmetric phase transfer catalytic reactions including asymmetric conjugate addition, asymmetric Mannich reactions, and asymmetric aldol condensation reactions, making it a versatile tool for diverse API intermediate synthesis.
Q: Is the synthesis of this catalyst scalable for industrial production?
A: The synthetic route utilizes standard organic transformations such as Suzuki coupling and nucleophilic substitution under relatively mild conditions, avoiding exotic reagents, which facilitates commercial scale-up and ensures supply chain reliability for large-volume manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phase Transfer Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the tetramethylspirodihydroindene-based catalyst in advancing asymmetric synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of characterizing complex chiral molecules to the highest standards. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated teams work tirelessly to ensure every batch meets the exacting requirements of global regulatory agencies.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments for the synthesis of your target chiral intermediates. Together, we can accelerate your development timeline and bring life-saving medicines to market faster and more efficiently.
