Advanced Synthesis of 3-Aminomethyl Tetrahydrofuran for High-Efficiency Dinotefuran Production
The global demand for next-generation neonicotinoid insecticides, particularly Dinotefuran, has created an urgent need for efficient and scalable synthesis routes for its key precursor, 3-aminomethyl tetrahydrofuran. Patent CN111303083A introduces a groundbreaking methodology that shifts the synthetic paradigm from traditional multi-step condensations to a streamlined hydroaminocarbonylation strategy. This technical breakthrough addresses critical bottlenecks in purity control and process safety that have long plagued the agrochemical intermediate sector. By utilizing 2,5-dihydrofuran as a primary feedstock, the invention eliminates the reliance on hazardous acyl chlorides and expensive precious metal catalysts found in legacy processes. For R&D Directors and Supply Chain Heads, this represents a significant opportunity to optimize the manufacturing of high-purity agrochemical intermediates. The protocol described herein not only enhances reaction selectivity but also aligns with modern green chemistry principles, ensuring that the production of 3-aminomethyl tetrahydrofuran is both economically viable and environmentally sustainable for large-scale commercial operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
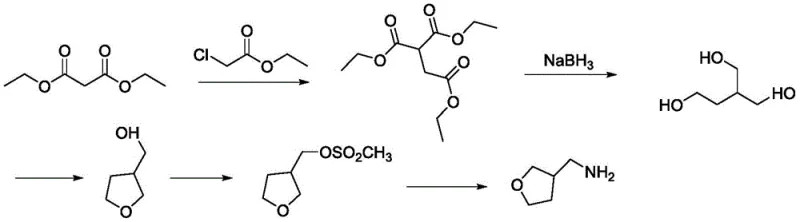
Historically, the industrial production of 3-aminomethyl tetrahydrofuran has been hindered by convoluted synthetic pathways that impose heavy burdens on both cost and environmental compliance. As illustrated in the conventional route, methods relying on diethyl malonate and ethyl chloroacetate require extensive reduction steps using sodium borohydride, followed by sulfonation and subsequent amination. This sequence is inherently inefficient, generating substantial quantities of chemical waste and requiring rigorous purification to remove inorganic salts. Furthermore, alternative routes involving hydroformylation often depend on Rhodium-based homogeneous catalysts, which are prohibitively expensive and difficult to recover from the reaction mixture. These legacy processes also frequently necessitate harsh reaction conditions, such as extreme low temperatures or the use of corrosive inorganic acyl chlorides, which accelerate equipment degradation and increase operational risks. For procurement managers, these factors translate into volatile production costs and unpredictable lead times, making the reliable sourcing of high-purity 3-aminomethyl tetrahydrofuran a persistent challenge in the competitive agrochemical market.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach detailed in patent CN111303083A leverages a direct hydroaminocarbonylation of 2,5-dihydrofuran to construct the core tetrahydrofuran skeleton with an amine functionality in fewer steps. This strategy bypasses the need for complex protecting group manipulations and expensive stoichiometric reducing agents. By integrating carbon monoxide and ammonium salts directly into the catalytic cycle, the process achieves a high atom economy that significantly reduces the volume of raw materials required per kilogram of product. The reaction conditions are notably milder, operating within a manageable temperature and pressure range that standard stainless-steel reactors can easily accommodate without specialized lining or cryogenic cooling. This simplification of the process flow not only accelerates the production cycle but also enhances the robustness of the manufacturing process against batch-to-batch variability. For supply chain stakeholders, this novel approach offers a pathway to cost reduction in agrochemical intermediate manufacturing by stabilizing the supply of critical precursors and minimizing the logistical complexities associated with hazardous reagent handling.
Mechanistic Insights into Pd-Catalyzed Hydroaminocarbonylation
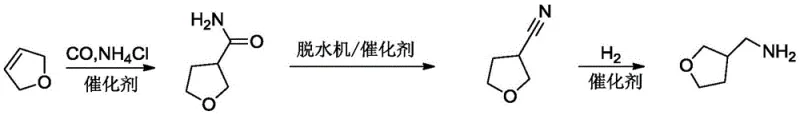
The core of this technological advancement lies in the palladium-catalyzed hydroaminocarbonylation mechanism, which facilitates the direct insertion of a carbonyl group and an amine moiety into the olefinic bond of 2,5-dihydrofuran. The catalytic cycle initiates with the coordination of the palladium species to the double bond, followed by the migratory insertion of carbon monoxide to form an acyl-palladium intermediate. Subsequent nucleophilic attack by the ammonium salt releases the 3-formamide tetrahydrofuran product and regenerates the active catalyst species. This mechanism is highly selective for the 3-position, minimizing the formation of regioisomers that typically complicate downstream purification. The use of various palladium precursors, such as palladium dichloride or palladium acetate, allows for fine-tuning of the electronic properties of the catalyst to maximize turnover numbers. Understanding this mechanistic pathway is crucial for R&D teams aiming to replicate the high yields reported in the patent, as it highlights the importance of maintaining precise carbon monoxide pressure and solvent polarity to sustain catalytic activity throughout the reaction duration.
Following the formation of the formamide intermediate, the process employs a dehydration cyanation step to convert the amide functionality into a nitrile group, which is subsequently reduced to the primary amine. This transformation is critical for establishing the aminomethyl side chain essential for the biological activity of the final pesticide. The dehydration is typically mediated by agents like phosphorus oxychloride or thionyl chloride in the presence of a catalyst, ensuring complete conversion while minimizing side reactions such as polymerization or ring-opening of the tetrahydrofuran moiety. The final catalytic hydrogenation step utilizes Raney nickel or supported transition metal catalysts to reduce the nitrile group under moderate hydrogen pressure. This step is designed to be highly chemoselective, reducing the nitrile without affecting the ether linkage in the ring. The cumulative effect of these mechanistic controls is a product profile with exceptional purity, often exceeding 96% as confirmed by gas chromatography, which meets the stringent specifications required for reliable agrochemical intermediate supplier certifications.
How to Synthesize 3-Aminomethyl Tetrahydrofuran Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and the control of gas pressures to ensure safety and reproducibility. The process begins with the hydroaminocarbonylation in a high-pressure reactor, where the stoichiometry of ammonium salt to olefin must be optimized to drive the equilibrium towards the formamide product. Following isolation, the dehydration step demands strict temperature control to prevent the decomposition of the sensitive nitrile intermediate. Finally, the hydrogenation phase requires thorough purging of the reactor to eliminate oxygen before introducing hydrogen, a standard safety protocol in fine chemical manufacturing. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures, are outlined in the structured guide below to assist technical teams in rapid technology transfer.
- Perform hydroaminocarbonylation on 2,5-dihydrofuran with CO and ammonium salt using a palladium catalyst to yield 3-formamide tetrahydrofuran.
- Conduct a dehydration cyanation reaction on the formamide intermediate using a dehydrating agent to produce 3-cyano tetrahydrofuran.
- Execute catalytic hydrogenation on the nitrile compound using a Raney-type or transition metal catalyst to obtain the final 3-aminomethyl tetrahydrofuran.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patent-protected synthesis route offers profound advantages for procurement managers and supply chain directors seeking to optimize their sourcing strategies for agrochemical intermediates. The primary benefit stems from the drastic simplification of the supply chain for raw materials; 2,5-dihydrofuran is a commercially available feedstock that does not suffer from the same price volatility as specialized organometallic reagents or complex esters used in older methods. By eliminating the need for stoichiometric amounts of expensive reducing agents like sodium borohydride or precious rhodium catalysts, the variable cost of goods sold is significantly reduced. This cost structure allows for more competitive pricing models without sacrificing margin, providing a buffer against market fluctuations in raw material costs. Furthermore, the reduced complexity of the process translates to shorter manufacturing cycles, enabling suppliers to respond more agilely to sudden spikes in demand for Dinotefuran precursors.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in auxiliary reagent consumption directly lower the operational expenditure per batch. Unlike previous methods that required costly metal removal steps to meet purity standards, this route utilizes catalysts that are easier to separate or are used in lower loadings, thereby reducing waste disposal costs. The overall process efficiency means that less energy is consumed per unit of product, contributing to substantial cost savings in agrochemical intermediate manufacturing. These efficiencies compound over large production volumes, making the economic case for switching to this new technology compelling for large-scale producers.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable raw materials mitigates the risk of supply disruptions that often plague specialized chemical supply chains. The milder reaction conditions reduce the wear and tear on production equipment, leading to higher asset availability and fewer unplanned maintenance shutdowns. This reliability is crucial for reducing lead time for high-purity agrochemical intermediates, ensuring that downstream pesticide formulators receive their materials on schedule. The robustness of the process also allows for easier scaling from pilot plant to commercial production, providing supply chain heads with confidence in long-term capacity planning.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional routes, simplifying compliance with increasingly strict environmental regulations. The absence of heavy metal contaminants in the final product reduces the burden on wastewater treatment facilities and lowers the cost of environmental remediation. This green chemistry profile enhances the marketability of the intermediate to eco-conscious global agrochemical companies. The scalability of the reaction, demonstrated by the wide range of viable pressures and temperatures, ensures that commercial scale-up of complex agrochemical intermediates can be achieved with minimal technical risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific technical advantages and operational parameters detailed in the patent documentation. Understanding these nuances is essential for technical procurement teams evaluating the feasibility of integrating this intermediate into their existing supply chains. The answers provide clarity on yield expectations, safety protocols, and the comparative benefits over legacy manufacturing methods.
Q: Why is the hydroaminocarbonylation route superior to traditional malonate methods?
A: Traditional methods using diethyl malonate involve complex multi-step reductions and generate significant three-waste pollution. The novel hydroaminocarbonylation route utilizes 2,5-dihydrofuran directly, simplifying the process, reducing auxiliary raw material consumption like sodium borohydride, and significantly lowering environmental impact while improving overall yield.
Q: What are the cost advantages of avoiding Rhodium catalysts in this synthesis?
A: Previous hydroformylation methods relied on expensive Rhodium homogeneous catalysts which drastically increased industrial costs and limited catalyst recovery. This patent utilizes Palladium-based catalysts which are more cost-effective and easier to manage in large-scale reactors, leading to substantial cost savings in agrochemical intermediate manufacturing without compromising reaction selectivity.
Q: How does this method improve supply chain reliability for Dinotefuran production?
A: The process operates under milder reaction conditions (50-200°C) compared to harsh low-temperature or high-corrosion alternatives. This reduces equipment maintenance downtime and safety risks, ensuring consistent production continuity. The use of readily available raw materials like 2,5-dihydrofuran further stabilizes the supply chain against raw material volatility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminomethyl Tetrahydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficacy and safety of final agrochemical products. Our technical team has extensively analyzed the synthetic route described in CN111303083A and possesses the expertise to implement this advanced chemistry at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-aminomethyl tetrahydrofuran meets the exacting standards required for Dinotefuran synthesis. We are committed to being a reliable 3-aminomethyl tetrahydrofuran supplier that partners with you to optimize your production costs and secure your supply chain.
We invite global agrochemical manufacturers to collaborate with us to leverage this innovative synthesis technology for their production lines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this route for your specific volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project timelines. Let us help you navigate the complexities of fine chemical sourcing and achieve a competitive edge in the market through superior process chemistry and supply chain reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
