Advanced Synthetic Route for Florfenicol: Enhancing Purity and Scalability for Veterinary Pharmaceutical Manufacturing
The global demand for effective veterinary antibiotics continues to drive innovation in the synthesis of broad-spectrum agents like Florfenicol. Patent CN102070497A introduces a transformative methodology that addresses the longstanding inefficiencies in producing this critical active pharmaceutical ingredient (API). Unlike traditional pathways that rely on cumbersome resolution processes or excessively long linear sequences, this invention leverages a strategic aziridine intermediate to streamline the construction of the fluoromethyl side chain. By focusing on a concise four-step sequence starting from a protected aziridine precursor, the technology offers a robust framework for achieving high optical purity and yield. For R&D directors and procurement specialists, understanding this shift from resolution-dependent methods to asymmetric synthesis via aziridine ring manipulation is crucial for evaluating future supply chain stability and cost structures in the veterinary pharmaceutical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Florfenicol has been plagued by synthetic inefficiencies that inflate costs and complicate supply chains. As illustrated in prior art such as Chinese Patent CN1743308, traditional routes often involve more than ten discrete chemical transformations, including asymmetric cyanohydrin formation, multiple protection and deprotection steps, and hazardous reagent usage.
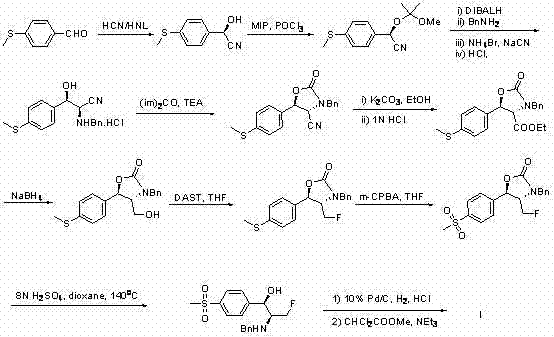
These lengthy sequences not only accumulate impurities at every stage, necessitating rigorous and costly purification protocols, but also result in significant material loss, drastically reducing the overall mass balance. Furthermore, methods relying on the resolution of racemic mixtures, as seen in US Patent US5382673, inherently discard up to half of the starting material, creating substantial waste disposal challenges and raw material inefficiencies. The reliance on toxic reagents and extreme reaction conditions in these legacy processes further exacerbates environmental compliance burdens, making them increasingly unsustainable for modern green chemistry mandates.
The Novel Approach
In stark contrast, the methodology disclosed in CN102070497A presents a paradigm shift by utilizing a pre-formed chiral aziridine scaffold to introduce the critical fluoromethyl motif with high fidelity. This approach condenses the synthesis into just four high-yielding steps: fluorination, acid-catalyzed ring opening, catalytic hydrogenation, and final acylation. By bypassing the need for early-stage resolution and minimizing the number of unit operations, this route significantly enhances the atom economy of the process. The use of mild fluorinating agents like DAST or Ishikawa reagent allows for precise control over the introduction of the fluorine atom without compromising the sensitive sulfone moiety. This streamlined strategy not only accelerates the time-to-market for batch production but also simplifies the downstream processing requirements, offering a clear pathway to cost reduction in veterinary drug manufacturing.
Mechanistic Insights into Aziridine Fluorination and Ring Opening
The core innovation of this synthesis lies in the stereoselective fluorination of the aziridine intermediate (II) and its subsequent regioselective ring opening. The process begins with the conversion of the hydroxymethyl group on the aziridine ring to a fluoromethyl group using diethylaminosulfur trifluoride (DAST) or related reagents. This transformation proceeds with retention of configuration at the chiral centers, a critical factor for maintaining the biological potency of the final Florfenicol molecule. The aziridine ring serves as a rigid template that locks the stereochemistry, preventing epimerization during the harsh fluorination conditions. Following fluorination, the strained three-membered ring is subjected to acid-catalyzed hydrolysis, typically using p-toluenesulfonic acid in a mixed solvent system.
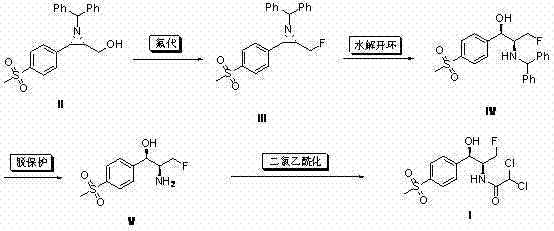
This ring-opening step is mechanistically fascinating as the nucleophilic attack by water occurs regioselectively at the benzylic position, driven by the stability of the developing carbocation character and the electronic influence of the p-methylsulfonyl group. This ensures the formation of the desired (1R,2S) amino alcohol skeleton with high diastereomeric excess. The subsequent removal of the benzhydryl protecting group via palladium-catalyzed hydrogenation is conducted under acidic conditions to prevent any potential base-catalyzed degradation of the fluorinated side chain. Finally, the dichloroacetylation is performed under mild basic conditions to install the pharmacophore, completing the synthesis with minimal risk of side reactions such as elimination or over-acylation.
How to Synthesize Florfenicol Efficiently
The implementation of this synthetic route requires careful attention to reagent quality and reaction monitoring to ensure consistent batch-to-batch reproducibility. The fluorination step, while robust, demands anhydrous conditions to prevent reagent decomposition, while the ring-opening step benefits from precise temperature control to optimize the ratio of regioisomers. Detailed standard operating procedures regarding quenching protocols and extraction methods are essential for maximizing isolation yields. For a comprehensive breakdown of the specific molar ratios, solvent choices, and workup procedures validated in the patent examples, please refer to the standardized guide below.
- Fluorinate the aziridine intermediate (II) using DAST or Ishikawa reagent to form the fluoromethyl aziridine (III).
- Perform acid-catalyzed ring opening of compound (III) using p-toluenesulfonic acid to yield the amino alcohol (IV).
- Execute palladium-carbon hydrogenation under acidic conditions to remove the benzhydryl protecting group, affording compound (V).
- Conduct dichloroacetylation of compound (V) with dichloroacetate esters in the presence of a base to finalize Florfenicol (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic advantages beyond mere technical elegance. The reduction in step count directly correlates to a decrease in capital expenditure on reactor time and a reduction in the inventory of intermediate materials that must be stored and managed. By eliminating the need for chiral resolution, the process removes the inherent 50% yield ceiling associated with racemic starting materials, effectively doubling the theoretical output from the same amount of raw material input. This efficiency gain is compounded by the use of widely available commodity chemicals rather than bespoke, high-cost chiral catalysts, stabilizing the cost of goods sold (COGS) against market volatility.
- Cost Reduction in Manufacturing: The streamlined four-step sequence drastically reduces the consumption of solvents, reagents, and energy compared to the ten-step legacy processes. Eliminating the resolution step alone represents a massive saving in raw material costs, as no valuable enantiomer is discarded as waste. Furthermore, the high yields reported in the patent examples for each individual step compound to create a superior overall process yield, meaning less starting material is required to produce one kilogram of final API. The avoidance of cryogenic conditions and the use of ambient pressure hydrogenation further lower the utility costs associated with heating, cooling, and specialized high-pressure equipment maintenance.
- Enhanced Supply Chain Reliability: Dependence on complex, multi-step syntheses often introduces multiple points of failure in the supply chain, where a bottleneck in any single step can halt entire production campaigns. This robust route minimizes such risks by utilizing stable intermediates and reagents that are readily sourced from multiple global suppliers. The simplicity of the workup procedures, involving standard extractions and crystallizations rather than complex chromatographic separations, ensures that production throughput is not limited by purification capacity. This operational resilience allows for more accurate forecasting of delivery timelines and reduces the likelihood of stockouts for critical veterinary medications.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a cleaner profile by reducing the total volume of chemical waste generated per unit of product. The absence of heavy metal catalysts in the key bond-forming steps simplifies the purification of the final drug substance to meet stringent residual metal specifications. The process is inherently scalable, as demonstrated by the use of standard organic solvents and reaction conditions that translate easily from laboratory glassware to industrial steel reactors. This scalability ensures that supply can be rapidly ramped up to meet surges in demand without requiring significant re-engineering of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Florfenicol synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders assess the feasibility of integrating this route into existing manufacturing portfolios.
Q: How does the new aziridine route improve upon traditional Florfenicol synthesis methods?
A: The patented route significantly reduces the number of synthetic steps compared to prior art like CN1743308, eliminating the need for multiple protection/deprotection cycles and toxic reagents, thereby improving overall yield and reducing environmental impact.
Q: What are the critical control points for maintaining stereochemistry in this synthesis?
A: Stereochemical integrity is primarily maintained during the fluorination of the aziridine ring and the subsequent regioselective acid-catalyzed ring opening, which preserves the (1R,2S) configuration essential for biological activity.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions, commercially available reagents like DAST and Pd/C, and avoids cryogenic temperatures or extreme pressures, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Florfenicol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Florfenicol intermediate meets the highest global regulatory standards. Our commitment to quality assurance ensures that the impurity profiles are tightly controlled, facilitating smoother regulatory filings for our clients.
We invite you to collaborate with us to leverage this advanced synthesis technology for your veterinary pharmaceutical projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of high-purity Florfenicol intermediates.
