Optimizing 7-ANCA Production: A Technical Breakthrough in Cephalosporin Intermediate Manufacturing
Introduction to Patented 7-ANCA Synthesis Technology
The pharmaceutical industry's demand for third-generation cephalosporins, such as ceftizoxime and ceftibuten, relies heavily on the efficient and cost-effective production of their core parent nucleus, 7-amino-3-non-3-cephem-4-carboxylic acid (7-ANCA). A significant technological advancement in this domain is detailed in Chinese Patent CN102180888B, which discloses a novel preparation method that fundamentally alters the traditional synthetic landscape. This patent introduces a streamlined pathway starting from 7-phenylacetamide-3-hydroxy cephalosporanic acid esters, utilizing a sequence of sulfonylation, elimination, Lewis acid-mediated deprotection, and enzymatic hydrolysis. Unlike conventional methods that often suffer from harsh reaction conditions and complex purification requirements, this invention emphasizes mild operating parameters, typically ranging from -10°C to room temperature, ensuring high chemical stability and yield. For R&D directors and process chemists, this represents a critical opportunity to re-evaluate existing manufacturing lines for improved efficiency and reduced environmental footprint.
The strategic value of this patent lies in its ability to bypass the limitations of earlier synthetic routes that relied on expensive catalysts or hazardous reagents. By employing aluminum chloride and anisole for the removal of the 4-position carboxyl protective group, the process eliminates the need for high-pressure hydrogenation equipment or toxic heavy metal reductants. This shift not only lowers the barrier to entry for manufacturing facilities but also enhances the overall safety profile of the production line. As global regulatory bodies tighten restrictions on heavy metal residues in active pharmaceutical ingredients (APIs), adopting a heavy-metal-free synthesis route becomes a competitive necessity rather than just an option. This report analyzes the technical merits and commercial implications of this patented methodology for stakeholders in the fine chemical and pharmaceutical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 7-ANCA has been plagued by several significant technical bottlenecks that impact both cost and scalability. One prominent prior art method, disclosed in CN 101357927A, utilizes a nitrobenzyl protecting group at the 4-position. While chemically stable, removing this group typically requires catalytic hydrogenation, a process that demands substantial capital investment in high-pressure reactors and expensive palladium catalysts. Furthermore, hydrogenation reactions can be sensitive to impurities, leading to inconsistent yields and potential safety hazards in large-scale operations. Another common approach involves the use of phosphorus pentachloride and pyridine at low temperatures to remove the 7-position phenylacetylamino group, a procedure known for generating large volumes of acidic waste and requiring rigorous temperature control that increases energy consumption.
Additionally, alternative routes reported in literature, such as those utilizing tributyltin hydride or zinc powder for reduction steps, introduce severe environmental liabilities. The use of organotin compounds results in toxic wastewater that is difficult and costly to treat to meet modern environmental standards. Similarly, zinc powder reduction generates significant amounts of solid metal waste. These legacy methods not only inflate the operational expenditure due to waste disposal costs but also complicate the supply chain by relying on reagents that are increasingly scrutinized under green chemistry initiatives. The cumulative effect of these drawbacks is a manufacturing process that is fragile, expensive, and environmentally burdensome, necessitating a robust alternative for sustainable production.
The Novel Approach
The patented method offers a transformative solution by replacing these problematic steps with a more elegant and efficient chemical strategy. The core innovation involves a sequential transformation where the 3-hydroxyl group is first sulfonylated and then eliminated to form the double bond, followed by a Lewis acid-mediated deprotection. Specifically, the use of aluminum chloride in the presence of anisole allows for the removal of diphenylmethyl or paramethoxybenzyl protecting groups under mild conditions without the need for hydrogen gas. This chemical switch drastically reduces equipment requirements, allowing production in standard glass-lined or stainless steel reactors without high-pressure ratings.
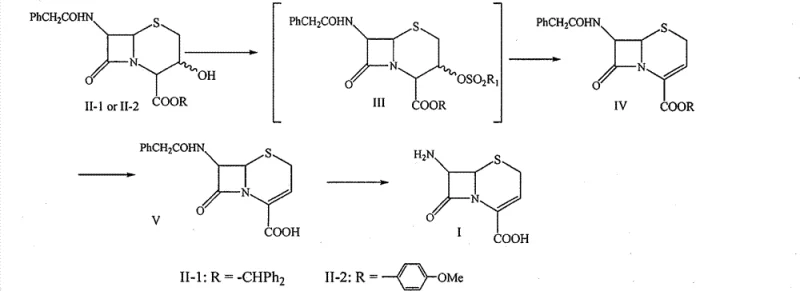
Furthermore, the final deprotection of the 7-position amino group is achieved using immobilized penicillin acylase, an enzymatic process that offers high regioselectivity and operates under neutral to slightly alkaline conditions. This biocatalytic step ensures that the sensitive beta-lactam ring remains intact, minimizing the formation of degradation impurities that often plague chemical hydrolysis methods. The combination of chemical and enzymatic steps creates a hybrid process that leverages the robustness of organic synthesis with the precision of biotechnology. For procurement managers, this means a supply chain that is less dependent on volatile precious metal markets and more resilient to regulatory changes regarding heavy metal usage.
Mechanistic Insights into Aluminum Chloride Mediated Deprotection
The mechanistic elegance of this synthesis lies primarily in the deprotection step, which serves as the pivot point for the entire process efficiency. In the traditional context, removing a diphenylmethyl ester usually requires strong acids like trifluoroacetic acid or catalytic hydrogenation. However, this patent utilizes a Friedel-Crafts type alkylation mechanism where aluminum chloride acts as a Lewis acid to coordinate with the carbonyl oxygen of the ester, increasing its electrophilicity. Anisole, acting as a scavenger, captures the resulting carbocation, driving the equilibrium towards the free carboxylic acid. This reaction proceeds smoothly in halogenated hydrocarbons like dichloromethane or aromatic solvents like toluene at temperatures as low as 0°C to 5°C. The mildness of this condition is crucial for preserving the integrity of the cephem nucleus, which is prone to ring-opening under harsh acidic or thermal stress.
From an impurity control perspective, this mechanism offers distinct advantages over oxidative or reductive deprotection methods. Chemical hydrogenation can sometimes lead to over-reduction of the double bond at the 3-position or the saturation of other sensitive functionalities if not carefully monitored. In contrast, the Lewis acid pathway is highly specific to the benzylic ester linkage. The subsequent enzymatic hydrolysis using immobilized penicillin acylase further refines the purity profile. Enzymes are inherently selective, targeting only the amide bond at the 7-position while leaving the beta-lactam ring and the newly formed double bond untouched. This dual-selectivity—chemical for the ester and biological for the amide—results in a crude product with a significantly cleaner impurity profile, reducing the burden on downstream purification processes like crystallization and chromatography.
How to Synthesize 7-ANCA Efficiently
The synthesis of 7-ANCA via this patented route is designed for operational simplicity and high throughput. The process begins with the activation of the 3-hydroxyl group using a sulfonyl chloride, such as methanesulfonyl chloride or benzenesulfonyl chloride, in the presence of an organic base like triethylamine. This is immediately followed by an elimination reaction using a secondary amine, such as diethylamine, to generate the delta-3 double bond. The resulting intermediate can then be subjected to the aluminum chloride deprotection step, which can potentially be telescoped to minimize solvent swaps. Finally, the enzymatic cleavage yields the target acid. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Perform 3-hydroxyl sulfonylation of the starting ester (II-1 or II-2) using sulfonyl chloride and an organic base at temperatures between -10°C and room temperature.
- Execute an elimination reaction using a secondary amine base to form the double bond at the 3-position, yielding compound IV.
- Remove the 4-position carboxyl protective group (diphenylmethyl or paramethoxybenzyl) using aluminum chloride and anisole in a halogenated hydrocarbon solvent.
- Hydrolyze the 7-position phenylacetylamino group using immobilized penicillin acylase under mild alkaline conditions to obtain the final 7-ANCA product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates into tangible strategic benefits beyond mere technical feasibility. The primary advantage is the drastic simplification of the raw material portfolio. By eliminating the need for palladium catalysts, tributyltin reagents, or high-pressure hydrogen gas, the purchasing department can source commodities that are widely available and price-stable. Aluminum chloride and anisole are bulk chemicals with mature supply chains, insulating the manufacturer from the volatility often seen in the precious metals market. This stability allows for more accurate long-term cost forecasting and contract negotiation with suppliers, ensuring a steady flow of materials even during global supply disruptions.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of capital-intensive unit operations. Traditional hydrogenation requires specialized high-pressure autoclaves and rigorous safety systems, representing a significant depreciation cost. By shifting to atmospheric pressure Lewis acid chemistry, facilities can utilize standard reactor trains, thereby lowering the barrier to entry and reducing fixed asset costs. Additionally, the avoidance of toxic heavy metals like tin and zinc removes the need for expensive wastewater treatment infrastructure dedicated to metal ion removal. The enzymatic step, while requiring specific biocatalysts, operates under mild conditions that reduce energy consumption for heating or cooling, contributing to lower overall utility costs per kilogram of product.
- Enhanced Supply Chain Reliability: Reliability in the pharmaceutical supply chain is paramount, and this process enhances it by reducing dependency on single-source or geographically concentrated reagents. Precious metal catalysts often face supply bottlenecks due to mining constraints or geopolitical factors. In contrast, the reagents used in this patented method—sulfonyl chlorides, organic amines, and aluminum salts—are produced by a wide range of chemical manufacturers globally. This diversification of the supplier base mitigates the risk of stockouts. Furthermore, the mild reaction conditions reduce the likelihood of batch failures due to thermal runaways or pressure excursions, leading to more consistent production schedules and reliable delivery times to downstream API customers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, but this route is inherently scalable due to its benign nature. The absence of exothermic hydrogenation events and toxic metal waste simplifies the scale-up protocol from pilot plant to commercial tonnage. From a regulatory standpoint, producing an intermediate with lower heavy metal content simplifies the registration dossier for the final drug product. Regulatory agencies worldwide are increasingly stringent about elemental impurities (ICH Q3D guidelines). By designing a process that avoids introducing these impurities in the first place, the manufacturer reduces the analytical burden and the risk of regulatory rejection, ensuring smoother market access for the final cephalosporin antibiotics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 7-ANCA synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and licensees. Understanding these nuances is essential for evaluating the fit of this technology within your existing manufacturing infrastructure.
Q: Why is the aluminum chloride deprotection method superior to traditional hydrogenation for 7-ANCA production?
A: Traditional methods often rely on catalytic hydrogenation to remove nitrobenzyl protecting groups, which requires high-pressure equipment and expensive palladium catalysts. The patented aluminum chloride method operates at atmospheric pressure and mild temperatures (-10°C to room temperature), significantly reducing facility investment and eliminating the safety risks associated with high-pressure hydrogenation while maintaining high yields.
Q: What are the key environmental advantages of this synthesis route compared to tributyltin or zinc powder methods?
A: Legacy synthetic routes frequently utilize toxic heavy metals like tributyltin or large quantities of zinc powder for reduction steps, generating hazardous metal-ion wastewater that is costly to treat. This novel process avoids heavy metal reagents entirely, utilizing organic amines and aluminum chloride, which simplifies waste treatment protocols and aligns with stricter global environmental compliance standards for API manufacturing.
Q: Can this process be scaled for commercial production of third-generation cephalosporins?
A: Yes, the process is specifically designed for industrialization. It utilizes readily available raw materials like 7-phenylacetamide-3-hydroxy cephalosporanic acid esters and common solvents such as dichloromethane and toluene. The reaction conditions are mild, and the one-pot potential for the first three steps reduces operational complexity, making it highly suitable for large-scale commercial manufacturing of intermediates for drugs like ceftizoxime.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-ANCA Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO specializing in complex pharmaceutical intermediates, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of this patented process, including the safe handling of Lewis acids and the integration of biocatalytic steps. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 7-ANCA meets the exacting standards required for third-generation cephalosporin synthesis.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you secure not just a supplier, but a strategic ally committed to driving efficiency and quality in the global pharmaceutical market.
