Advanced Zinc-Catalyzed Synthesis of Multi-Substituted Quinoline Derivatives for Pharma
Advanced Zinc-Catalyzed Synthesis of Multi-Substituted Quinoline Derivatives for Pharma
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing privileged scaffolds such as quinoline derivatives. These structures are ubiquitous in bioactive molecules, exhibiting potent antimalarial, antiepileptic, and antitumor activities, yet their synthesis has historically been plagued by economic and environmental inefficiencies. A groundbreaking development detailed in patent CN111116466B, published on February 10, 2023, introduces a transformative catalytic synthesis method that leverages inexpensive zinc iodide (ZnI2) to drive the cyclization of aryl propargyl substituted anilines. This innovation represents a paradigm shift away from reliance on scarce precious metals, offering a pathway to high-purity pharmaceutical intermediates that aligns with modern green chemistry principles while drastically improving process economics for large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinoline core has relied heavily on two distinct but problematic strategies: classical strong acid-mediated cyclizations and transition metal catalysis using noble metals. Traditional methods often necessitate harsh acidic conditions that not only limit functional group tolerance but also result in significant formation of tarry by-products and lower overall isolated yields, complicating downstream purification. Alternatively, while noble metal catalysts such as rhodium complexes have demonstrated the ability to facilitate these transformations under milder conditions, their prohibitive cost creates a severe bottleneck for commercial viability. For instance, literature precedents utilizing rhodium catalysts report costs exceeding 700 yuan for the catalyst alone per gram of 2-phenylquinoline produced, rendering such routes economically unfeasible for commodity-level production of active pharmaceutical ingredients or agrochemical intermediates where margin compression is a constant pressure.
The Novel Approach
The methodology disclosed in the referenced patent circumvents these economic barriers by employing zinc iodide, a readily available and inexpensive Lewis acid, as the primary catalyst. This novel approach operates under relatively mild thermal conditions (100°C) in a common organic solvent like toluene, eliminating the need for exotic ligands or high-pressure equipment. The versatility of this system is exemplified by its successful application across a diverse range of substrates, including those bearing electron-donating groups like methoxy and methyl, as well as electron-withdrawing halogens such as chlorine and bromine. 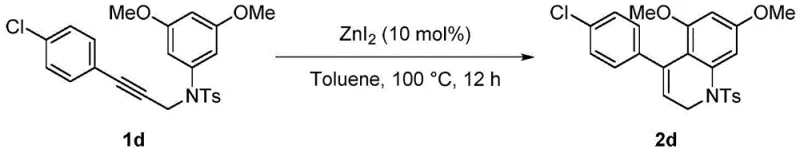 As illustrated in the reaction schemes, the process consistently delivers excellent yields, often surpassing 90%, which indicates a highly efficient conversion that minimizes raw material waste and maximizes throughput for supply chain planning.
As illustrated in the reaction schemes, the process consistently delivers excellent yields, often surpassing 90%, which indicates a highly efficient conversion that minimizes raw material waste and maximizes throughput for supply chain planning.
Mechanistic Insights into ZnI2-Catalyzed Cyclization
The efficacy of this synthesis lies in the specific Lewis acidic nature of zinc iodide, which activates the alkyne moiety of the aryl propargyl substituted aniline towards intramolecular nucleophilic attack by the nitrogen atom. Unlike stronger Brønsted acids that might protonate the amine and deactivate it, the zinc center coordinates selectively with the pi-system of the triple bond, lowering the energy barrier for the 6-endo-dig cyclization without compromising the nucleophilicity of the aniline nitrogen. This mechanistic pathway ensures a clean transformation that proceeds with high regioselectivity, avoiding the formation of isomeric by-products that often plague acid-catalyzed variants. The reaction profile suggests a concerted or stepwise electrophilic activation that is sufficiently robust to tolerate various substituents on the aromatic rings, thereby providing a reliable platform for generating diverse libraries of quinoline analogues for drug discovery programs.
Furthermore, the choice of zinc iodide contributes significantly to impurity control and product purity, which are critical parameters for R&D directors overseeing API development. The mild reaction temperature of 100°C prevents thermal degradation of sensitive functional groups, while the use of toluene as a solvent facilitates easy removal and recycling, reducing the residual solvent burden in the final crude product. 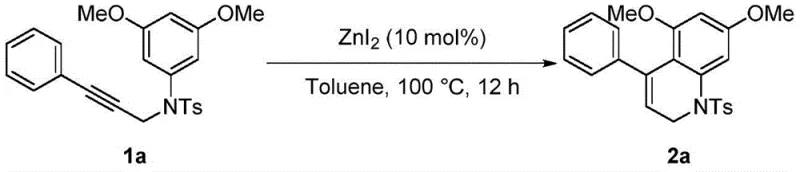 The absence of heavy transition metals like rhodium or iridium eliminates the need for expensive and technically demanding metal scavenging steps, which are typically required to meet strict regulatory limits on residual metals in pharmaceutical substances. This inherent cleanliness of the reaction profile simplifies the purification workflow, often allowing for straightforward silica gel chromatography or crystallization to achieve the stringent purity specifications required for clinical grade materials.
The absence of heavy transition metals like rhodium or iridium eliminates the need for expensive and technically demanding metal scavenging steps, which are typically required to meet strict regulatory limits on residual metals in pharmaceutical substances. This inherent cleanliness of the reaction profile simplifies the purification workflow, often allowing for straightforward silica gel chromatography or crystallization to achieve the stringent purity specifications required for clinical grade materials.
How to Synthesize Multi-Substituted Quinoline Derivatives Efficiently
Implementing this catalytic protocol in a laboratory or pilot plant setting requires adherence to specific stoichiometric and thermal parameters to ensure optimal performance. The process begins with the dissolution of the aryl propargyl substituted aniline starting material in toluene, maintaining a precise molar volume ratio of 1:1 mmol/mL to ensure adequate solubility and reaction kinetics. Following this, a catalytic amount of zinc iodide, specifically 10 mol% relative to the substrate, is introduced to the mixture to initiate the catalytic cycle.
- Dissolve aryl propargyl substituted aniline in toluene at a molar volume ratio of 1: 1 mmol/mL under normal pressure.
- Add zinc iodide (10 mol%) to the toluene solution to form a mixed solution.
- Heat the mixture to 100°C and stir constantly for 12 hours to generate the multi-substituted quinoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-catalyzed route offers profound strategic advantages that extend far beyond simple reagent substitution. The most immediate impact is the drastic reduction in direct material costs associated with catalysis; shifting from a rhodium-based system to a zinc-based one reduces the catalyst cost component by three orders of magnitude, fundamentally altering the cost structure of the final molecule. This economic efficiency allows for greater flexibility in pricing strategies and improves the overall margin profile of the product portfolio, making it highly competitive in the global market for fine chemical intermediates. Additionally, the reliance on commodity chemicals like zinc iodide and toluene mitigates supply risk, as these materials are produced at massive scales globally and are not subject to the geopolitical volatility often associated with platinum group metals.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a significant line item from the bill of materials, while simultaneously reducing the operational expenditure related to metal recovery and waste treatment. By avoiding the use of rhodium or iridium complexes, manufacturers save substantially on both the initial purchase of the catalyst and the downstream processing costs required to remove trace metal residues to meet regulatory standards. This dual saving mechanism ensures that the total cost of goods sold (COGS) is significantly lowered, enhancing the commercial viability of producing these quinoline derivatives at multi-ton scales without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: Sourcing zinc iodide is inherently more stable and predictable than sourcing specialized noble metal catalysts, which often have long lead times and limited supplier bases. The use of standard solvents like toluene further simplifies logistics, as these are stocked by virtually every chemical distributor worldwide, ensuring continuous production capability even during periods of market disruption. This robustness in the supply chain translates to shorter lead times for customers and a higher degree of confidence in meeting delivery schedules, which is critical for pharmaceutical clients managing tight development timelines and inventory levels.
- Scalability and Environmental Compliance: The reaction conditions described—normal pressure and moderate temperatures—are ideally suited for scale-up in standard stainless steel reactors, removing the need for specialized high-pressure equipment. The high yields observed, ranging from 90.5% to 96.2%, indicate a highly atom-economical process that generates minimal waste, aligning with increasingly stringent environmental regulations and sustainability goals. This scalability ensures that the transition from gram-scale laboratory synthesis to hundred-kilogram or ton-scale commercial production is seamless, allowing for rapid response to market demand surges without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the experimental data and comparative analysis provided in the patent documentation. These insights are intended to clarify the operational benefits and technical feasibility for potential partners evaluating this technology for their own supply chains.
Q: How does the ZnI2 catalyzed method compare to Rhodium catalysts in terms of cost?
A: The ZnI2 catalyzed method reduces catalyst costs by 3 orders of magnitude compared to traditional Rhodium complex methods, dropping from approximately 700 yuan per gram of product to less than 1 yuan.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction is conducted under normal pressure in toluene solvent at 100°C for 12 hours, using 10 mol% zinc iodide as the catalyst.
Q: What yields can be expected with this novel catalytic approach?
A: The process achieves high yields ranging from 90.5% to 96.2%, significantly outperforming many conventional strong acid methods which often suffer from lower efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes that balance technical excellence with commercial pragmatism. Our team of expert chemists has thoroughly evaluated the zinc-catalyzed cyclization method described in patent CN111116466B and confirmed its potential for delivering high-purity quinoline derivatives with exceptional consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is executed with precision. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the exacting standards required by the global pharmaceutical industry, providing our partners with a secure and reliable source of critical intermediates.
We invite you to collaborate with us to leverage this cost-effective technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the market for multi-substituted quinoline derivatives through our advanced manufacturing capabilities.
