Advanced Synthesis of 3-Substituted Vinyl Cephalosporin Derivatives for Oral Antibiotics
The pharmaceutical landscape for beta-lactam antibiotics continues to evolve, driven by the urgent need for agents with superior oral bioavailability and broader antimicrobial spectra. Patent CN1048706A introduces a significant breakthrough in this domain by disclosing novel 3-substituted vinyl cephalosporin derivatives. These compounds represent a strategic advancement over earlier generations, specifically addressing the limitations of poor absorption associated with traditional 3-position substituents. The core innovation lies in the precise modification of the cephem nucleus at the 3-position with various vinyl groups, coupled with sophisticated 7-position side chains containing aminothiazole and oximino moieties. This dual modification strategy results in compounds that exhibit remarkable antagonistic action against various pathogenic bacteria, positioning them as potent candidates for next-generation oral antibiotic therapies. For industry stakeholders, understanding the synthetic accessibility and structural versatility of these derivatives is crucial for evaluating their potential as reliable cephalosporin intermediate supplier offerings.
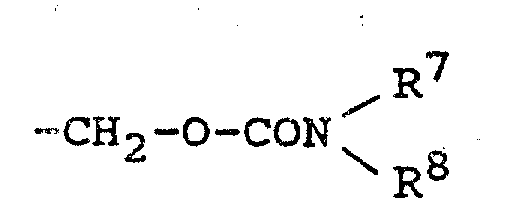
The development of these derivatives addresses a critical gap in existing cephalosporin chemistry. Conventional methods for modifying the 3-position often involve harsh conditions that can compromise the integrity of the sensitive beta-lactam ring or result in mixtures of stereoisomers that are difficult to separate. Traditional approaches using simple nucleophilic displacement at the 3-acetoxymethyl position frequently suffer from low yields and require extensive purification to remove toxic heavy metal catalysts or unstable intermediates. Furthermore, achieving the specific Z-configuration of the oximino group at the 7-position, which is essential for high biological activity, has historically been a challenging bottleneck in process chemistry. These limitations not only inflate production costs but also pose significant risks to supply chain continuity due to the complexity of waste management and the scarcity of high-purity starting materials required for older synthetic routes.
In contrast, the novel approach detailed in the patent leverages a robust Wittig olefination strategy to install the vinyl functionality at the 3-position with high stereocontrol. By utilizing specific phosphoranyl methyl cephalosporin precursors and reacting them with tailored aldehydes under mild conditions, the process achieves superior regioselectivity and minimizes degradation of the beta-lactam core. This method allows for the introduction of diverse functional groups, such as carbamoyloxy and heterocyclic vinyl moieties, without compromising the stability of the molecule. Additionally, the acylation steps at the 7-position employ activated ester methods that proceed efficiently at low temperatures, preserving the delicate stereochemistry required for optimal pharmacological performance. This streamlined synthetic pathway not only enhances overall yield but also simplifies downstream processing, making it an ideal candidate for cost reduction in antibiotic manufacturing.
Mechanistic Insights into Wittig Olefination and Beta-Lactam Acylation
The core mechanistic advantage of this synthesis lies in the careful orchestration of the Wittig reaction at the C-3 position. The process begins with a 3-phosphoranyl methyl cephalosporin intermediate, which acts as the ylide precursor. Upon reaction with specific aldehydes, such as t-butyldimethylsilyloxy acetaldehyde or heterocyclic aldehydes, the reaction proceeds through a four-membered oxaphosphetane transition state. This mechanism is highly favorable for generating the desired Z-alkene geometry, which is critical for the biological activity of the final cephalosporin derivative. The use of silyl protecting groups during this stage ensures that reactive hydroxyl functionalities do not interfere with the olefination, allowing for subsequent deprotection and functionalization steps to be carried out orthogonally. This level of control over the reaction trajectory is essential for maintaining high purity specifications and minimizing the formation of E-isomer impurities that could complicate regulatory approval.
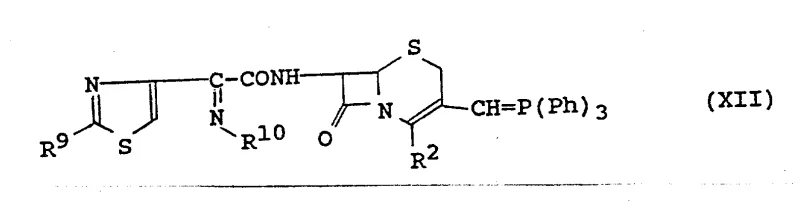
Following the establishment of the 3-vinyl side chain, the synthesis focuses on the acylation of the 7-amino group. This step typically involves the coupling of the cephalosporin nucleus with an activated thiazolyl-acetic acid derivative. The activation is often achieved using reagents like dicyclohexylcarbodiimide (DCC) in the presence of hydroxybenzotriazole (HOBt), which facilitates the formation of an active ester intermediate in situ. This activated species then reacts rapidly with the 7-amino group of the cephem nucleus. The reaction conditions are meticulously controlled, often requiring temperatures as low as -50°C to 50°C depending on the specific reagents, to prevent racemization at the chiral centers of the beta-lactam ring. The resulting amide bond is stable and positions the pharmacophore correctly for interaction with bacterial penicillin-binding proteins. Understanding these mechanistic nuances is vital for any team aiming for the commercial scale-up of complex pharmaceutical intermediates, as slight deviations in temperature or stoichiometry can lead to significant variations in product quality.
How to Synthesize 3-Vinyl Cephalosporin Derivatives Efficiently
The synthesis of these high-value intermediates requires a disciplined approach to reaction monitoring and purification. The process generally initiates with the preparation of the phosphorane precursor, followed by the key olefination step which sets the stereochemistry for the entire molecule. Subsequent steps involve the strategic installation of the 7-side chain and the final deprotection of carboxyl and amino groups to reveal the active pharmacophore. Each stage demands rigorous control over solvent quality and moisture content to ensure consistent yields. For a comprehensive understanding of the operational parameters, including specific molar ratios, solvent systems, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical path from raw materials to the final sodium salt.
- Preparation of the 3-phosphoranyl methyl cephalosporin precursor followed by Wittig reaction with specific aldehydes to introduce the vinyl side chain.
- Acylation of the 7-amino group using activated thiazolyl-acetic acid derivatives under controlled low-temperature conditions to maintain stereochemistry.
- Deprotection of amino and carboxyl groups using trifluoroacetic acid or formic acid, followed by salt formation to obtain the final active pharmaceutical ingredient.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route described in patent CN1048706A offers distinct logistical and economic benefits over legacy methods. The reliance on Wittig chemistry, while requiring specific phosphorus reagents, utilizes starting materials that are widely available in the global fine chemical market. This reduces the risk of supply bottlenecks that often plague processes dependent on exotic or single-source catalysts. Furthermore, the ability to crystallize intermediates at various stages of the synthesis provides natural purification checkpoints, reducing the burden on final purification steps and lowering the overall consumption of chromatography media. This inherent process robustness translates directly into enhanced supply chain reliability, ensuring that production schedules can be met consistently without unexpected delays caused by difficult-to-remove impurities.
- Cost Reduction in Manufacturing: The streamlined nature of the synthetic pathway eliminates the need for multiple protection and deprotection cycles that are common in older cephalosporin syntheses. By reducing the total number of unit operations, the process significantly lowers energy consumption and solvent usage, which are major cost drivers in pharmaceutical manufacturing. Additionally, the high selectivity of the acylation steps minimizes the formation of regioisomers, thereby increasing the effective yield of the desired product and reducing the waste disposal costs associated with off-spec material. This efficiency creates substantial cost savings opportunities without compromising the stringent quality standards required for antibiotic intermediates.
- Enhanced Supply Chain Reliability: The modular design of the synthesis allows for the decoupling of certain steps, meaning that key intermediates can be stockpiled or sourced from different facilities if necessary. This flexibility is crucial for maintaining continuity of supply in a volatile global market. The use of stable protecting groups, such as trityl and silyl ethers, ensures that intermediates have a long shelf life, allowing manufacturers to build strategic inventory buffers. This stability mitigates the risk of production stoppages due to raw material spoilage or transportation delays, providing a more resilient supply chain for downstream drug formulation partners.
- Scalability and Environmental Compliance: The reaction conditions described are amenable to large-scale batch processing, with thermal profiles that can be safely managed in standard stainless steel reactors. The avoidance of highly toxic heavy metal catalysts simplifies the environmental compliance landscape, reducing the complexity of wastewater treatment and hazardous waste disposal. This aligns with modern green chemistry principles and regulatory expectations, facilitating smoother permitting processes for new manufacturing lines. The ability to scale this process from pilot plant to multi-ton production ensures that growing market demand for these potent oral antibiotics can be met sustainably and efficiently.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these 3-substituted vinyl cephalosporin derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for making informed decisions about sourcing and process integration.
Q: What is the primary advantage of the 3-vinyl substitution in these cephalosporins?
A: The 3-vinyl substitution significantly enhances oral bioavailability and antimicrobial spectrum compared to traditional 3-methyl or 3-acetoxymethyl cephalosporins, making them highly effective for perorally administrable treatments.
Q: How does the patented process ensure high purity for commercial scale-up?
A: The process utilizes specific crystallization techniques and column chromatography purification steps after key intermediates are formed, effectively removing triphenylphosphine oxide byproducts common in Wittig reactions.
Q: Are the starting materials for this synthesis readily available?
A: Yes, the synthesis relies on standard cephalosporin nuclei and commercially accessible aldehydes and phosphoranes, ensuring a stable supply chain for large-scale manufacturing without exotic reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephalosporin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving antibiotics. Our expertise in beta-lactam chemistry allows us to replicate and optimize complex synthetic pathways like the one described in CN1048706A with precision. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets rigorous international standards. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities down to ppm levels, guaranteeing that every batch of cephalosporin intermediate we deliver supports the efficacy and safety of your final drug product.
We invite you to collaborate with us to leverage these advanced synthetic technologies for your pipeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain partner committed to innovation, quality, and the reliable delivery of complex pharmaceutical intermediates essential for the next generation of antimicrobial therapies.
