Advanced Synthesis of Amidinothioacetamide Cephalosporins for Enhanced Antibiotic Potency
Advanced Synthesis of Amidinothioacetamide Cephalosporins for Enhanced Antibiotic Potency
The escalating crisis of antimicrobial resistance, particularly among Gram-positive pathogens such as Methicillin-Resistant Staphylococcus Aureus (MRSA) and Penicillin-Resistant Streptococcus Pneumoniae (PRSP), has necessitated the continuous evolution of beta-lactam antibiotic architectures. In response to this critical global health challenge, patent CN1315845C discloses a groundbreaking class of methylene-containing aminoheterocycle substituted cephalosporins featuring an amidinothioacetamide moiety. This technological breakthrough represents a significant leap forward in semi-synthetic antibiotic design, specifically targeting the dual modification of the C-3 and C-7 positions of the cephem nucleus to broaden the antimicrobial spectrum. By integrating a nitrogen-containing heterocyclic ring at the C-3 position and introducing a specialized thiourea-derived side chain at the C-7 amino group, this innovation delivers compounds with superior stability against beta-lactamases and markedly improved efficacy against resistant bacterial strains. For pharmaceutical manufacturers and R&D teams seeking to diversify their antibiotic pipelines, understanding the synthetic nuances and commercial viability of these intermediates is paramount for developing next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to modifying cephalosporin antibiotics have often been constrained by the inherent instability of the beta-lactam ring under harsh reaction conditions and the limited availability of versatile C-3 leaving groups. Historically, many third-generation cephalosporins, such as cefotaxime and ceftriaxone, rely on specific acetoxy or heterocyclic thiomethyl groups at the C-3 position that, while effective, can limit further structural diversification without compromising metabolic stability. Conventional acylation methods at the C-7 position frequently suffer from low regioselectivity, leading to complex mixtures of N-acylated and O-acylated byproducts that require extensive and costly purification protocols. Furthermore, older synthetic routes often utilize hazardous reagents or multi-step protection-deprotection sequences that drastically reduce overall atom economy and increase the environmental footprint of the manufacturing process. These inefficiencies create significant bottlenecks in the supply chain, resulting in higher production costs and longer lead times for critical antibiotic intermediates, thereby hindering the rapid deployment of new formulations to combat emerging resistant pathogens.
The Novel Approach
The methodology outlined in the patent data introduces a streamlined and highly efficient synthetic strategy that overcomes these historical hurdles through precise chemical engineering. This novel approach leverages the unique reactivity of 7-amino-3-methylene-containing aminoheterocycle substituted cephalosporanic acid derivatives, such as 7-ACT (7-amino-3-((2,5-dihydro-6-hydroxy-2-methyl-5-oxo-as-triazine-3-yl) methyl thio))-cephalosporanic acid. 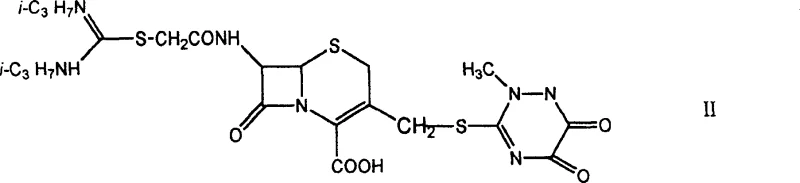 By utilizing a direct bromoacetylation strategy followed by a nucleophilic substitution with disubstituted thioureas, the process achieves high yields with exceptional purity profiles. The introduction of the amidinothioacetamide group at the C-7 position is particularly innovative, as it enhances the lipophilicity and membrane permeability of the molecule, facilitating better penetration into bacterial cells. This route eliminates the need for complex protecting groups and allows for the use of readily available, cost-effective reagents like bromoacetyl bromide and diisopropyl thiourea. The result is a robust manufacturing process that not only improves the biological profile of the final antibiotic but also aligns with modern green chemistry principles by minimizing waste and simplifying downstream processing.
By utilizing a direct bromoacetylation strategy followed by a nucleophilic substitution with disubstituted thioureas, the process achieves high yields with exceptional purity profiles. The introduction of the amidinothioacetamide group at the C-7 position is particularly innovative, as it enhances the lipophilicity and membrane permeability of the molecule, facilitating better penetration into bacterial cells. This route eliminates the need for complex protecting groups and allows for the use of readily available, cost-effective reagents like bromoacetyl bromide and diisopropyl thiourea. The result is a robust manufacturing process that not only improves the biological profile of the final antibiotic but also aligns with modern green chemistry principles by minimizing waste and simplifying downstream processing.
Mechanistic Insights into Bromoacetylation and Thiourea Substitution
The core of this synthetic innovation lies in a two-stage mechanistic sequence that meticulously functionalizes the cephalosporin nucleus. The first stage involves the nucleophilic attack of the primary amine at the C-7 position on the carbonyl carbon of bromoacetyl bromide. This reaction is typically conducted in a biphasic or mixed solvent system, such as acetone-water or dichloromethane-water, maintained at a controlled temperature range of 0°C to 50°C to prevent degradation of the sensitive beta-lactam ring. The presence of an organic base, such as triethylamine, or a mineral alkali like sodium bicarbonate, is crucial for scavenging the hydrogen bromide byproduct, thereby driving the equilibrium towards the formation of the 7-acetobromo intermediate. 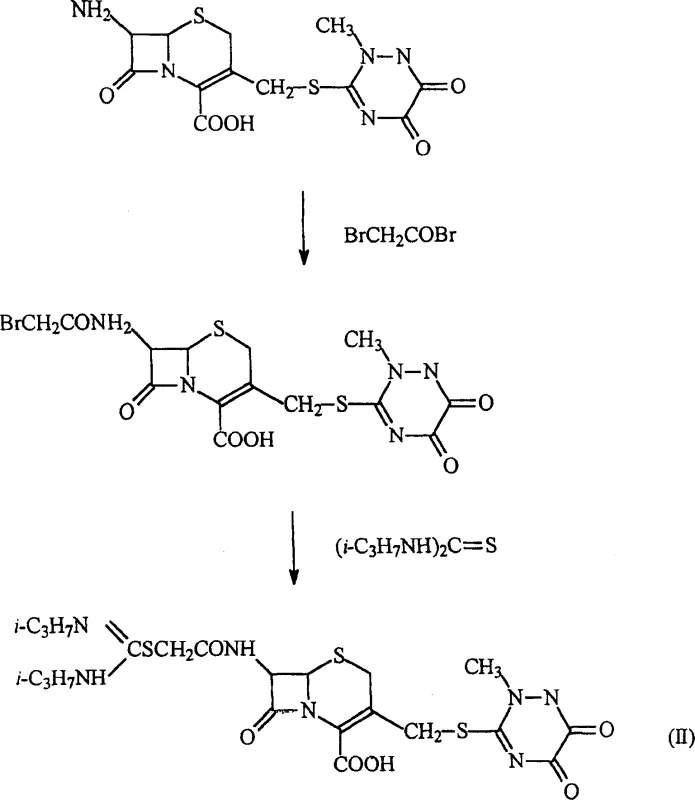 Following the formation of this reactive intermediate, the second stage entails a nucleophilic displacement of the bromide ion by the sulfur atom of the N,N-disubstituted thiourea. This substitution reaction proceeds efficiently due to the high nucleophilicity of the thiourea sulfur and the excellent leaving group ability of the bromide. The reaction conditions are carefully optimized, often involving reflux in solvents like dichloromethane or acetone, to ensure complete conversion while minimizing epimerization at the chiral centers of the cephem ring. The final product precipitates out of the solution upon cooling or acidification, allowing for easy isolation via filtration. This mechanistic pathway ensures that the stereochemical integrity of the antibiotic core is preserved, which is essential for maintaining its biological activity against penicillin-binding proteins in bacterial cell walls.
Following the formation of this reactive intermediate, the second stage entails a nucleophilic displacement of the bromide ion by the sulfur atom of the N,N-disubstituted thiourea. This substitution reaction proceeds efficiently due to the high nucleophilicity of the thiourea sulfur and the excellent leaving group ability of the bromide. The reaction conditions are carefully optimized, often involving reflux in solvents like dichloromethane or acetone, to ensure complete conversion while minimizing epimerization at the chiral centers of the cephem ring. The final product precipitates out of the solution upon cooling or acidification, allowing for easy isolation via filtration. This mechanistic pathway ensures that the stereochemical integrity of the antibiotic core is preserved, which is essential for maintaining its biological activity against penicillin-binding proteins in bacterial cell walls.
Impurity control is a critical aspect of this mechanism, particularly regarding the potential for hydrolysis of the beta-lactam ring or the formation of polymeric byproducts. The patent specifies strict pH control during the acidification step, typically adjusting to pH 3.0 using hydrochloric or sulfuric acid, which promotes the crystallization of the desired zwitterionic species while keeping acidic or basic impurities in the aqueous phase. Additionally, the choice of solvent plays a pivotal role in purity; for instance, the use of methyl isobutyl ketone (MIBK) or acetonitrile can enhance the solubility of organic impurities, allowing them to be washed away during the filtration of the solid product. The process is designed to achieve product purity levels exceeding 90%, a benchmark that is vital for meeting the stringent regulatory requirements of pharmaceutical grade intermediates. By understanding these mechanistic details, R&D directors can better appreciate the robustness of the process and its suitability for scale-up in a GMP environment.
How to Synthesize Amidinothioacetamide Cephalosporins Efficiently
The synthesis of these high-value antibiotic intermediates requires a disciplined approach to reaction parameters and workup procedures to ensure consistent quality and yield. The process begins with the dissolution of the starting cephalosporanic acid derivative in a suitable solvent system, followed by pH adjustment to ensure the amine is in its free base form for optimal reactivity. Subsequent addition of bromoacetyl bromide must be controlled to manage the exotherm and prevent side reactions. After the formation of the acetobromo intermediate, the reaction mixture is acidified to isolate the solid, which is then redissolved for the final coupling with the thiourea derivative. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and crystallization techniques, are provided in the guide below to assist technical teams in replicating this advanced chemistry.
- Dissolve the 7-amino C-3 methylene nitrogen heterocycle substituted cephalosporanic acid in a solvent system and adjust pH using organic bases or mineral alkalis.
- React the clarified solution with bromoacetyl bromide at controlled temperatures (0-50°C) to form the acetobromo intermediate.
- Acidify the reaction mixture to separate the intermediate, then react with N,N-disubstituted thiourea to finalize the amidinothioacetamide structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. The streamlined nature of the process, which avoids complex multi-step protections and utilizes commodity chemicals, translates into a significantly simplified manufacturing workflow. This simplicity reduces the operational burden on production facilities, allowing for faster batch turnover and more efficient utilization of reactor capacity. Furthermore, the reliance on stable, commercially available starting materials like 7-ACT and common reagents such as bromoacetyl bromide mitigates the risk of supply disruptions associated with exotic or custom-synthesized precursors. The robustness of the crystallization steps ensures high recovery rates, minimizing raw material waste and maximizing the value extracted from every kilogram of input. These factors collectively contribute to a more resilient and cost-effective supply chain for critical antibiotic intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in the number of unit operations lead to substantial cost savings in the overall production process. By utilizing straightforward acylation and substitution reactions that proceed with high conversion rates, the need for extensive chromatographic purification is removed, replaced by simple filtration and washing steps. This drastic simplification of the downstream processing significantly lowers the cost of goods sold (COGS), making the final API more competitive in the global market. Additionally, the use of recyclable solvent systems further enhances the economic viability of the process by reducing solvent procurement and disposal costs.
- Enhanced Supply Chain Reliability: The reliance on widely sourced chemical building blocks ensures a stable and continuous supply of raw materials, shielding the production schedule from the volatility often seen with specialized reagents. The robustness of the reaction conditions, which tolerate a reasonable range of temperatures and pH levels, reduces the likelihood of batch failures due to minor process deviations. This reliability is crucial for maintaining consistent inventory levels and meeting the demanding delivery schedules of pharmaceutical customers. The ability to produce high-purity intermediates consistently also reduces the risk of quality-related delays, ensuring a smooth flow of materials through the value chain.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory to pilot scales without significant loss of efficiency or selectivity. The use of aqueous workups and the ability to recover solvents align with increasingly stringent environmental regulations, reducing the ecological footprint of the manufacturing operation. The high purity of the final product minimizes the generation of hazardous waste streams associated with purification, supporting sustainable manufacturing practices. This scalability and environmental compatibility make the technology an attractive option for long-term commercial production, ensuring that supply can be ramped up to meet global demand without compromising on safety or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these novel cephalosporin derivatives. The answers are derived directly from the patented technology and provide clarity on the practical implementation of this chemistry. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their existing product portfolios or development pipelines.
Q: What is the primary therapeutic advantage of these new cephalosporin derivatives?
A: The novel C3-methylene nitrogen heterocycle substitution combined with the C-7 amidinothioacetamide group significantly enhances antimicrobial activity against Gram-positive bacteria, including resistant strains like Staphylococcus aureus, while maintaining stability against beta-lactamases.
Q: What are the key reagents used in this synthesis pathway?
A: The synthesis primarily utilizes 7-amino-3-((nitrogen heterocycle) methyl)-cephalosporanic acid (such as 7-ACT), bromoacetyl bromide for acylation, and N,N-diisopropyl thiourea for the final substitution step, often employing triethylamine or sodium bicarbonate as base catalysts.
Q: How does this process ensure high product purity?
A: The process incorporates specific acidification and crystallization steps, such as adjusting pH to 3.0 and utilizing solvent mixtures like acetone-water or dichloromethane-alcohol, which allow for the effective precipitation of the target compound while leaving impurities in the mother liquor, achieving purity levels exceeding 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amidinothioacetamide Cephalosporin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving antibiotics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global pharmaceutical industry. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our expertise in semi-synthetic beta-lactam chemistry positions us as a strategic partner capable of navigating the complexities of cephalosporin synthesis with precision and reliability.
We invite you to collaborate with us to explore the potential of these advanced amidinothioacetamide cephalosporins for your next-generation antibiotic projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and process constraints. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can drive value and innovation in your supply chain.
