Scalable Metal-Free Acetoxylation of Pyrrolo[2,3-d]pyrimidines for Commercial API Production
Scalable Metal-Free Acetoxylation of Pyrrolo[2,3-d]pyrimidines for Commercial API Production
The pharmaceutical industry is constantly seeking robust synthetic routes that eliminate toxic impurities while maintaining high efficiency, and the technology disclosed in patent CN112979662A represents a significant leap forward in this domain. This patent introduces a novel, transition-metal-free method for the carbon-hydrogen activated acetoxylation of pyrrolo[2,3-d]pyrimidine derivatives, a structural motif critical for kinase inhibitors and anticancer agents. By leveraging hypervalent iodine chemistry rather than traditional palladium or rhodium catalysis, this process addresses the stringent purity requirements demanded by modern regulatory bodies. For R&D directors and procurement specialists alike, this methodology offers a pathway to high-purity intermediates without the burden of heavy metal scavenging steps. The ability to achieve yields up to 93% under mild conditions underscores the commercial viability of this approach for scaling complex heterocyclic scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of electron-rich heteroaromatic systems like pyrrolo[2,3-d]pyrimidines has relied heavily on noble metal catalysts such as Palladium (Pd), Rhodium (Rh), and Ruthenium (Ru). While effective, these conventional methods suffer from severe drawbacks that impact both cost and supply chain reliability. The primary concern is the residual toxicity of these heavy metals, which necessitates complex and expensive purification protocols to meet International Council for Harmonisation (ICH) guidelines for drug substances. Furthermore, noble metals are subject to volatile market pricing and geopolitical supply constraints, introducing significant risk to long-term manufacturing contracts. The use of these catalysts often requires specialized ligands and harsh reaction conditions, which can lead to lower atom economy and increased waste generation, making the process less sustainable and more costly for large-scale production.
The Novel Approach
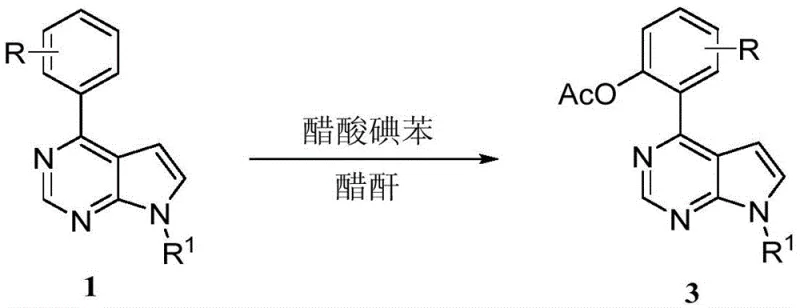
In stark contrast, the methodology described in CN112979662A utilizes a metal-free system driven by iodobenzene diacetate (PIDA) and sodium iodide in acetic anhydride. This innovative route bypasses the need for transition metals entirely, thereby eliminating the risk of metal contamination at the source. The reaction operates under relatively mild thermal conditions, ranging from 50°C to 110°C, and utilizes acetic anhydride which serves as both solvent and acetylating agent. This dual functionality simplifies the reaction matrix and reduces the volume of auxiliary solvents required. The strategic use of sodium iodide allows for precise regioselective control, enabling manufacturers to target specific positions on the molecule simply by adjusting the additive profile and temperature. This flexibility transforms a potentially multi-step synthesis into a streamlined one-pot operation, drastically reducing processing time and operational complexity.
![Reaction scheme showing NaI-controlled C5 acetoxylation of pyrrolo[2,3-d]pyrimidine](/insights/img/pyrrolo-pyrimidine-synthesis-metal-free-pharma-supplier-20260307093801-01.webp)
The visual representation of the reaction pathways highlights the elegance of this chemical transformation. As depicted in the reaction schemes, the presence of sodium iodide acts as a crucial switch for regioselectivity. When sodium iodide is introduced into the reaction mixture containing the substrate and iodobenzene diacetate, the acetoxylation occurs selectively at the C5 position of the pyrrolo[2,3-d]pyrimidine core. This specific functionalization is achieved at temperatures between 50°C and 80°C over a period of 4 to 7 hours. Conversely, when sodium iodide is omitted and the temperature is elevated to the 90°C to 110°C range, the reaction trajectory shifts entirely, resulting in acetoxylation at the ortho-position of the phenyl ring attached to the heterocycle. This level of control without changing the core reagents is a testament to the sophistication of the hypervalent iodine mechanism employed here.
Mechanistic Insights into Iodine-Mediated C-H Activation
The underlying mechanism of this transformation relies on the unique oxidative properties of hypervalent iodine species generated in situ. In the presence of acetic anhydride, iodobenzene diacetate acts as a potent oxidant capable of activating inert C-H bonds. The addition of sodium iodide likely facilitates the formation of a more reactive iodine(III) species or an iodonium intermediate that coordinates preferentially with the electron-rich C5 position of the pyrrole ring. This coordination lowers the activation energy for the C-H bond cleavage at this specific site, allowing the acetoxy group to be installed with high fidelity. The absence of transition metals means that the catalytic cycle does not involve oxidative addition or reductive elimination steps typical of Pd-catalysis, but rather proceeds through ligand exchange and reductive elimination from the hypervalent iodine center. This distinct mechanistic pathway avoids the formation of metal-carbon bonds that are often prone to side reactions like homocoupling or beta-hydride elimination.
From an impurity control perspective, this mechanism offers substantial advantages for process chemistry teams. Since the oxidant is organic and the byproduct is iodobenzene, the impurity profile is significantly cleaner compared to metal-catalyzed reactions which often generate complex organometallic byproducts. The high selectivity observed, with yields reaching up to 93% for certain derivatives like the 4-chlorophenyl analog, indicates that competing side reactions such as over-oxidation or polymerization are effectively suppressed. The ability to tune the reaction outcome by simply adding or removing a salt like sodium iodide suggests a robust process window where minor fluctuations in conditions do not lead to catastrophic failure. This reliability is paramount when transferring a process from the laboratory bench to pilot plant reactors, ensuring consistent quality across batches.
How to Synthesize Acetoxylated Pyrrolo[2,3-d]pyrimidine Efficiently
To implement this synthesis effectively, operators must adhere to the specific stoichiometric ratios and thermal profiles outlined in the patent data. The standard protocol involves dissolving the starting pyrrolo[2,3-d]pyrimidine compound, iodobenzene diacetate, and sodium iodide in acetic anhydride at a mass-to-volume ratio that ensures complete solubility and efficient heat transfer. For the C5-acetoxylation pathway, the mixture is heated to a preferred temperature of 70°C and maintained for approximately 6 hours. Precise control of the sodium iodide loading is critical, with a molar ratio of substrate to oxidant to additive of 1:3:2 proving optimal for maximizing yield. Following the reaction, the workup procedure is straightforward, involving extraction with ethyl acetate and washing with saturated brine to remove inorganic salts, followed by drying and concentration. Detailed standardized synthesis steps are provided in the guide below.
- Dissolve the pyrrolo[2,3-d]pyrimidine substrate, iodobenzene diacetate, and sodium iodide in acetic anhydride solvent.
- Heat the reaction mixture to 50-80°C for C5-acetoxylation or 90-110°C for ortho-acetoxylation, stirring for 4-14 hours depending on the target isomer.
- Perform post-treatment by extracting with ethyl acetate, washing with saturated brine, drying over sodium sulfate, and purifying via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free technology translates directly into tangible operational efficiencies and risk mitigation. The most immediate benefit is the drastic simplification of the supply chain for raw materials. By eliminating the dependency on scarce and expensive noble metals like palladium or rhodium, manufacturers can insulate themselves from the volatility of the precious metals market. Furthermore, the reagents used in this process, such as iodobenzene diacetate and acetic anhydride, are commodity chemicals produced at a massive global scale, ensuring consistent availability and competitive pricing. This stability allows for more accurate long-term cost forecasting and reduces the likelihood of production delays caused by material shortages. The simplified reagent profile also means fewer vendors to qualify and manage, streamlining the procurement workflow.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the need for expensive metal scavengers and specialized filtration equipment typically required to reduce metal residues to ppm levels. This reduction in downstream processing steps leads to significant savings in both material costs and labor hours. Additionally, the high atom economy and excellent yields reported in the patent, reaching up to 93%, mean that less starting material is wasted, further driving down the cost per kilogram of the final active pharmaceutical ingredient. The use of acetic anhydride as both solvent and reagent reduces the overall volume of chemicals required, lowering waste disposal costs and improving the environmental footprint of the manufacturing process.
- Enhanced Supply Chain Reliability: Relying on abundant organic reagents rather than geopolitically sensitive precious metals enhances the resilience of the supply chain. The robustness of the reaction conditions, which tolerate a range of temperatures and times without significant loss in yield, ensures that production schedules can be met consistently even if minor operational variances occur. This reliability is crucial for maintaining continuous supply to downstream drug formulation partners. Moreover, the one-pot nature of the reaction reduces the number of intermediate isolation steps, shortening the overall manufacturing lead time and allowing for faster response to market demand fluctuations.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simple setup and lack of hazardous metal catalysts. Scaling from grams to tons does not require complex engineering solutions for metal containment or recovery. From an environmental standpoint, the absence of heavy metals simplifies wastewater treatment and waste disposal, helping facilities meet increasingly stringent environmental regulations. The generation of benign byproducts like iodobenzene, which can potentially be recycled or easily disposed of, aligns with green chemistry principles and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this acetoxylation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of this method. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines. The focus is on reproducibility, safety, and the specific advantages over legacy methods.
Q: Does this synthesis method leave heavy metal residues in the final API intermediate?
A: No. The patented process (CN112979662A) explicitly eliminates the need for transition metal catalysts such as Palladium, Rhodium, or Ruthenium. Instead, it utilizes organic hypervalent iodine reagents and sodium iodide, ensuring the final product is free from toxic heavy metal contamination, which significantly simplifies downstream purification for pharmaceutical applications.
Q: How is regioselectivity controlled between the C5 position and the phenyl ring?
A: Regioselectivity is precisely controlled by the presence or absence of sodium iodide. Adding sodium iodide directs the acetoxylation specifically to the C5 position of the pyrrolo[2,3-d]pyrimidine core at lower temperatures (50-80°C). Conversely, omitting sodium iodide and increasing the temperature to 90-110°C shifts the reaction to occur at the ortho-position of the pendant phenyl ring.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable. It operates as a one-pot reaction using cheap and readily available solvents like acetic anhydride. The absence of expensive noble metal catalysts and the high yields (up to 93%) make it economically viable for producing complex heteroaromatic intermediates on a multi-kilogram to ton scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolo[2,3-d]pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in the production of high-value pharmaceutical intermediates. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the highest standards for metal content and chemical identity. Our facility is equipped to handle the specific requirements of hypervalent iodine chemistry, ensuring safe and efficient processing of these energetic reagents.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this metal-free acetoxylation process can optimize your supply chain and reduce your overall cost of goods sold. Let us help you secure a reliable supply of these critical kinase inhibitor intermediates.
