Optimizing 6-Aminocapronitrile Production via Advanced Liquid-Phase Catalysis for Global Supply Chains
Optimizing 6-Aminocapronitrile Production via Advanced Liquid-Phase Catalysis for Global Supply Chains
The chemical industry is constantly seeking more efficient pathways to produce critical intermediates like 6-aminocapronitrile, a vital precursor for 1,6-hexamethylene diamine and subsequently Nylon 66 and Nylon 610. Recent advancements documented in patent CN111233704A introduce a transformative liquid-phase synthesis method that utilizes caprolactam as a primary feedstock. This innovation addresses long-standing challenges associated with traditional gas-phase hydrogenation and olefinic nitrile routes, which often suffer from harsh operating conditions and environmental concerns. By shifting to a mild, acid-catalyzed liquid-phase protocol, manufacturers can achieve superior reaction conversion rates and product purity levels exceeding 98%. This report analyzes the technical merits of this novel approach, providing R&D directors and procurement leaders with a comprehensive understanding of its potential to optimize supply chain resilience and reduce manufacturing costs in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 6-aminocapronitrile has relied heavily on the partial hydrogenation of 1,6-adiponitrile or the hydrogenation of olefinic nitriles, processes that are technically demanding and capital intensive. Existing patents, such as CN1238334C and CN101309897B, describe methods that often require precise control over hydrogen pressure and specialized catalysts, which can be prone to deactivation and require complex regeneration cycles. Furthermore, alternative approaches involving caprolactam gas-phase methods, as seen in CN107602416A, necessitate reaction temperatures exceeding 300°C and elevated pressures, creating significant safety hazards and energy burdens. These harsh conditions not only accelerate equipment degradation but also limit the feasibility of large-scale industrialization due to the high operational expenditure required to maintain such extreme environments. Additionally, some liquid-phase preparations have struggled with low reaction conversion rates and the extensive use of nitrile solvents, leading to substantial environmental pollution and waste treatment costs that undermine overall process sustainability.
The Novel Approach
In contrast, the novel method outlined in CN111233704A presents a robust alternative by leveraging a multi-step liquid-phase reaction sequence that operates under significantly milder conditions. This process initiates with the hydrolysis of caprolactam in an acidic aqueous solution, effectively opening the lactam ring to form a stable 6-aminocaproic acid salt without the need for extreme thermal energy. The subsequent steps involve a strategic recrystallization to purify the intermediate, followed by a dehydration and ammoniation reaction using readily available reagents like diols and ammonium salts. This pathway eliminates the need for high-pressure hydrogenation equipment and avoids the use of toxic nitrile solvents, thereby simplifying the reactor design and reducing the environmental footprint. The integration of a vacuum rectification step ensures the final isolation of 6-aminocapronitrile with high purity, demonstrating a clear technological leap forward in terms of operational safety, energy efficiency, and product quality for commercial chemical manufacturing.
Mechanistic Insights into Caprolactam Liquid-Phase Conversion
The core of this innovative synthesis lies in the controlled acid-catalyzed ring-opening of caprolactam, which serves as the foundational step for generating the linear aminocaproic structure. In this mechanism, caprolactam is uniformly mixed with an organic acid solution, such as formic acid, acetic acid, or methanesulfonic acid, and heated to initiate hydrolysis. The presence of the acidic medium is critical, as it protonates the carbonyl oxygen of the lactam ring, making it more susceptible to nucleophilic attack by water molecules, thereby facilitating ring opening to form the 6-aminocaproic acid salt. Without the addition of acid, direct hydrolysis would yield unstable 6-aminocaproic acid, which is prone to re-cyclization, halting the process; however, the formation of the salt stabilizes the linear structure and ensures the reaction proceeds in a homogeneous phase. This homogeneity is vital for maximizing contact between reactants, leading to improved reaction kinetics and higher conversion rates compared to heterogeneous gas-phase systems.
Following the initial hydrolysis, the process employs a sophisticated purification and dehydration strategy to drive the equilibrium towards the desired nitrile product. The intermediate 6-aminocaproate undergoes recrystallization using a solvent system comprising a good solvent like methanol and a poor solvent like toluene, which effectively removes impurities and provides a high-purity substrate for the next stage. In the subsequent reaction, the purified salt reacts with a dihydric alcohol, an ammoniating agent, and a dehydrating agent such as phosphorus oxychloride or trifluoroacetic anhydride. The diol enhances reactivity and reduces alcohol consumption, while the dehydrating agent facilitates the removal of water generated during the amidation, pushing the reaction forward to form the nitrile group. This careful orchestration of reagents ensures that the terminal amino groups participate effectively in the ammoniation reaction, minimizing side reactions and maximizing the yield of the final 6-aminocapronitrile product.
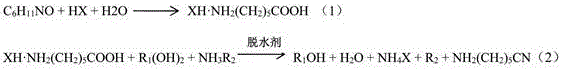
How to Synthesize 6-Aminocapronitrile Efficiently
Implementing this synthesis route requires precise control over reaction parameters to ensure optimal yield and purity, making it essential for process engineers to follow a standardized operational protocol. The procedure begins with the careful preparation of the acidic hydrolysis mixture, where the mass ratio of caprolactam to the acidic solution must be maintained within specific ranges to ensure complete ring opening without excessive acid waste. Following the reaction, the removal of low-boiling-point substances under reduced pressure is critical before initiating the recrystallization step, which acts as a key purification barrier to prevent the carryover of impurities into the final dehydration stage. The detailed standardized synthesis steps见下方的指南 outline the specific temperatures, stirring speeds, and reagent ratios required to replicate the high success rates observed in the patent examples, providing a clear roadmap for technical teams aiming to adopt this technology.
- Mix caprolactam with an organic acid solution and heat to initiate ring-opening hydrolysis.
- Remove low-boiling substances and recrystallize the resulting 6-aminocaproic acid salt for purification.
- React the purified salt with alcohol, ammoniating agent, and dehydrating agent, followed by vacuum rectification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this manufacturing process offers substantial benefits that directly address the pain points of procurement managers and supply chain heads regarding cost stability and operational reliability. By eliminating the need for expensive transition metal catalysts and high-pressure hydrogenation infrastructure, the process significantly reduces capital expenditure and ongoing maintenance costs associated with complex reactor systems. The use of common industrial chemicals like caprolactam, organic acids, and diols ensures a stable and diverse supply base, mitigating the risks associated with sourcing specialized or scarce reagents that often plague traditional synthetic routes. Furthermore, the mild reaction conditions translate to lower energy consumption per unit of product, contributing to a more sustainable and cost-effective manufacturing profile that aligns with modern corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts is a primary driver for cost optimization, as it removes the necessity for expensive and time-consuming heavy metal removal steps typically required in pharmaceutical and fine chemical production. This simplification of the downstream processing workflow reduces the consumption of purification media and lowers waste disposal costs, leading to substantial overall savings in the cost of goods sold. Additionally, the high reaction conversion rate minimizes the loss of raw materials, ensuring that a greater proportion of the input caprolactam is converted into valuable saleable product, thereby improving the overall material efficiency of the plant.
- Enhanced Supply Chain Reliability: Utilizing caprolactam as a starting material leverages an existing, mature global supply chain, ensuring consistent availability and price stability compared to more niche precursors used in alternative methods. The process flexibility allows for the use of various organic acids and ammoniating agents, giving procurement teams the agility to switch suppliers based on market conditions without compromising the integrity of the synthesis. This adaptability reduces the risk of supply disruptions and enables manufacturers to maintain continuous production schedules even during periods of raw material volatility.
- Scalability and Environmental Compliance: The reliance on standard unit operations such as reflux, crystallization, and vacuum distillation makes this process highly scalable from pilot plant to full commercial production without requiring bespoke engineering solutions. The avoidance of toxic nitrile solvents and the reduction of hazardous waste streams simplify environmental compliance and permit acquisition, accelerating the timeline for new facility commissioning. This streamlined regulatory pathway allows companies to bring products to market faster while maintaining a strong commitment to environmental stewardship and operational safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this caprolactam-based synthesis route, derived directly from the patent's background and beneficial effect sections. These insights are intended to clarify the operational advantages and technical feasibility for stakeholders evaluating this technology for potential adoption in their manufacturing portfolios. Understanding these details is crucial for making informed decisions about process integration and supply chain optimization strategies.
Q: What are the primary advantages of the caprolactam liquid-phase method over gas-phase hydrogenation?
A: The liquid-phase method operates under significantly milder conditions, avoiding the extreme temperatures above 300°C and high pressures required by gas-phase processes, thereby reducing energy consumption and equipment stress.
Q: How does the recrystallization step impact the final purity of 6-aminocapronitrile?
A: Recrystallization of the intermediate 6-aminocaproic acid salt effectively removes impurities and unreacted starting materials, ensuring a high-purity feed for the subsequent amidation reaction and achieving final product purity exceeding 98%.
Q: Is this process scalable for industrial production of nylon precursors?
A: Yes, the process utilizes standard unit operations such as reflux, crystallization, and vacuum distillation, which are well-established in industrial chemical engineering, facilitating straightforward scale-up from laboratory to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Aminocapronitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes like the one described in CN111233704A for securing the supply of high-value polymer intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries are translated into reliable industrial processes. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 6-aminocapronitrile meets the exacting standards required for downstream nylon and polyamide applications. We are committed to delivering consistent quality and supply continuity, positioning ourselves as a strategic partner for global chemical enterprises.
We invite you to collaborate with us to explore how this advanced synthesis method can enhance your production capabilities and reduce overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can support your supply chain optimization goals and drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
