Revolutionizing Antiviral Intermediate Production: A Safe and Scalable Synthesis of 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane
Introduction to Advanced Antiviral Intermediate Synthesis
The pharmaceutical industry continuously seeks robust and scalable pathways for critical antiviral intermediates, particularly those serving as the backbone for hepatitis C protease inhibitors and emerging therapeutic agents. Patent CN113999160B introduces a groundbreaking preparation method for 6,6-dimethyl-3-azabicyclo[3.1.0]hexane, a pivotal structural motif found in drugs like Boceprevir and the oral neo-crown therapeutic drug PF-07321332. This specific chemical architecture, characterized by its fused cyclopropane-pyrrolidine ring system, presents significant synthetic challenges due to ring strain and stereochemical requirements. The disclosed technology addresses these challenges by shifting away from hazardous traditional reagents towards a more benign, industrially viable protocol. By leveraging dichlorochrysanthemic acid as a cost-effective starting material, the invention not only mitigates safety risks associated with explosive reducing agents but also streamlines the supply chain for high-purity pharmaceutical intermediates. This report analyzes the technical merits and commercial implications of this novel synthetic strategy.
![Chemical structure of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane (Formula I)](/insights/img/6-6-dimethyl-3-azabicyclo-hexane-synthesis-pharma-supplier-20260305063512-01.png)
The strategic importance of this intermediate cannot be overstated, as it serves as a core scaffold for next-generation antiviral therapies. As global demand for effective treatments against RNA viruses escalates, the ability to manufacture key building blocks like Formula I with consistent quality and safety becomes a critical competitive advantage. The patent details a multi-step sequence that transforms a readily available agricultural chemical precursor into a high-value pharmaceutical ingredient through a series of controlled transformations including reduction, hydrolysis, amidation, and intramolecular cyclization. This approach represents a paradigm shift from batch processes reliant on extreme conditions to a more continuous and manageable workflow. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for evaluating potential suppliers and optimizing the cost of goods sold (COGS) for final drug products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane has been plagued by significant safety hazards and economic inefficiencies. Prior art, such as the methods described in WO2008082508 and US7723531, predominantly relies on lithium aluminum hydride (LAH) for the reduction of lactams or anhydrides. LAH is notoriously pyrophoric, requiring strictly anhydrous conditions and specialized handling equipment to prevent catastrophic fires. Furthermore, these conventional routes often necessitate a subsequent hydrogenation step to remove benzyl protecting groups, which involves high-pressure reactors and flammable palladium-carbon catalysts. The combination of high-pressure hydrogen gas and pyrophoric catalysts creates a volatile environment that demands extensive safety infrastructure and insurance overheads. Additionally, the use of caronic anhydride as a starting material in some legacy routes drives up raw material costs due to its limited availability and complex synthesis. These factors collectively result in a manufacturing process that is difficult to scale, expensive to operate, and fraught with regulatory scrutiny regarding process safety management.
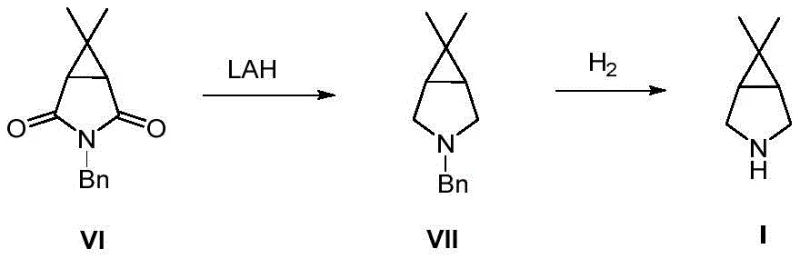
The Novel Approach
In stark contrast, the methodology outlined in CN113999160B offers a transformative solution by completely eliminating the need for LAH and high-pressure hydrogenation. The new route initiates with dichlorochrysanthemic acid, a commodity chemical that is significantly cheaper and more accessible than caronic anhydride. The process employs alkali metal borohydrides, such as sodium borohydride, in conjunction with Lewis acids for the initial reduction step. This reagent combination provides sufficient reducing power under much milder conditions, typically between 0°C and 30°C, thereby removing the thermal runaway risks associated with LAH. The subsequent steps involve standard hydrolysis and a Hofmann rearrangement using hypochlorite, which are well-understood unit operations in fine chemical manufacturing. The final cyclization is achieved using organic bases at atmospheric pressure, avoiding the need for autoclaves. This holistic redesign of the synthetic tree not only enhances operator safety but also simplifies the equipment requirements, allowing for production in standard glass-lined or stainless steel reactors without specialized high-pressure ratings.
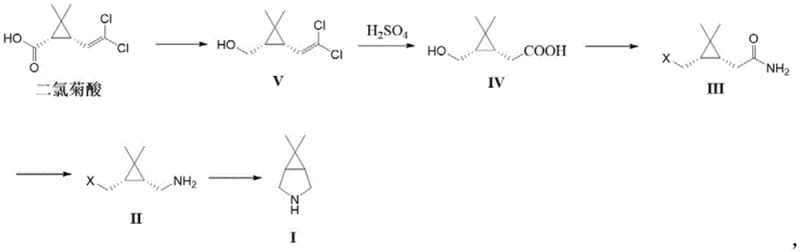
Mechanistic Insights into the Borohydride-Lewis Acid Reduction and Hofmann Rearrangement
The core innovation of this process lies in the clever application of a borohydride-Lewis acid system to reduce the carboxylic acid derivative directly to the corresponding alcohol (Compound V). Mechanistically, the Lewis acid, such as boron trifluoride diethyl etherate or aluminum chloride, activates the carbonyl oxygen of the acid, making it more susceptible to nucleophilic attack by the hydride species generated in situ from the borohydride. This activation allows the reduction to proceed efficiently at near-ambient temperatures, a condition under which sodium borohydride alone would be inert towards carboxylic acids. This step is crucial because it bypasses the need to convert the acid to a more reactive ester or acid chloride prior to reduction, saving both time and reagents. The reaction is carefully quenched with methanol to destroy excess borane species, ensuring that the workup is safe and manageable. The resulting alcohol V is then hydrolyzed under acidic conditions to yield the hydroxy-acid IV, setting the stage for the nitrogen insertion.
The transformation of the amide (Compound III) to the amine (Compound II) utilizes the classic Hofmann rearrangement, mediated by hypochlorite in an alkaline medium. This reaction involves the formation of an N-chloroamide intermediate, which undergoes deprotonation and rearrangement to an isocyanate. The isocyanate is subsequently hydrolyzed to the primary amine with loss of carbon dioxide. In the context of this specific substrate, the stereochemical integrity of the cyclopropane ring is maintained throughout this rigorous oxidative process, which is a testament to the robustness of the reaction conditions. The use of aqueous hypochlorite is particularly advantageous from a green chemistry perspective, as it avoids the use of heavy metal oxidants. Finally, the intramolecular nucleophilic substitution where the amine attacks the chloromethyl group to close the pyrrolidine ring is driven by the entropic favorability of forming a five-membered ring and the relief of steric strain. The use of bulky organic bases like potassium tert-butoxide facilitates this cyclization by generating the free amine nucleophile without promoting elimination side reactions.
How to Synthesize 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Efficiently
The execution of this synthesis requires precise control over reaction parameters to maximize yield and purity while maintaining safety. The process begins with the dissolution of dichlorochrysanthemic acid in an anhydrous solvent like tetrahydrofuran, followed by the controlled addition of sodium borohydride and a Lewis acid at low temperatures to manage the exotherm. Following the reduction, the crude alcohol is subjected to acid-catalyzed hydrolysis to generate the key hydroxy-acid intermediate. This intermediate is then activated via halogenation, typically using thionyl chloride, and immediately treated with ammonia to form the amide. The subsequent Hofmann rearrangement is performed in a biphasic or aqueous system, requiring careful temperature modulation to ensure complete conversion of the isocyanate intermediate. The final cyclization step is a reflux operation in a non-polar solvent like toluene, which aids in the removal of water and drives the equilibrium towards the bicyclic product. Detailed standard operating procedures for each of these critical stages are provided below to ensure reproducibility and compliance with GMP standards.
- Reduce dichlorochrysanthemic acid with alkali metal borohydride and Lewis acid to form compound V.
- Hydrolyze compound V in sulfuric acid to obtain carboxylic acid derivative IV.
- Convert IV to amide III via halogenation and ammonolysis, then perform Hofmann rearrangement to yield amine II.
- Execute self-cyclization of compound II using an organic base to finalize the bicyclic structure I.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers substantial strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By switching from specialized, high-cost precursors like caronic anhydride to dichlorochrysanthemic acid, manufacturers can leverage a broader supplier base and negotiate more favorable pricing terms. Dichlorochrysanthemic acid is a bulk chemical with established production capacity, ensuring supply continuity even during market fluctuations. Furthermore, the elimination of lithium aluminum hydride removes a significant logistical burden, as LAH is classified as a dangerous good requiring special shipping and storage protocols. Replacing it with sodium borohydride, a stable and widely available solid, simplifies warehouse management and reduces hazmat handling costs. This shift also mitigates the risk of production stoppages due to the unavailability of niche reagents.
From a capital expenditure (CAPEX) and operational expenditure (OPEX) perspective, the new process is equally compelling. The removal of high-pressure hydrogenation steps means that production facilities do not require expensive autoclaves or the associated safety systems for handling hydrogen gas. This allows the synthesis to be performed in standard multipurpose reactors, increasing asset utilization and flexibility. The absence of noble metal catalysts like palladium-on-carbon eliminates the need for complex metal scavenging and recovery units, which are often required to meet strict residual metal limits in pharmaceutical ingredients. Consequently, the overall cost of manufacturing is significantly reduced through lower utility consumption, reduced waste treatment costs, and decreased equipment maintenance. The milder reaction conditions also translate to longer equipment lifespan and reduced downtime, further enhancing the economic viability of large-scale production.
Environmental compliance and sustainability are increasingly critical factors in vendor selection, and this process excels in these areas. By avoiding toxic heavy metals and explosive reagents, the waste stream generated is less hazardous and easier to treat. The use of aqueous workups and common organic solvents facilitates solvent recovery and recycling, aligning with green chemistry principles. This reduced environmental footprint not only lowers disposal costs but also enhances the corporate social responsibility profile of the supply chain. For pharmaceutical companies aiming to reduce the carbon footprint of their drug portfolios, sourcing intermediates produced via this cleaner technology provides a tangible metric for sustainability reporting. The combination of cost efficiency, supply security, and environmental stewardship makes this manufacturing route a superior choice for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane using the patented method. These insights are derived directly from the experimental data and process descriptions within CN113999160B, providing clarity on scalability, purity, and regulatory considerations. Understanding these aspects is vital for technical teams evaluating the feasibility of integrating this intermediate into their existing manufacturing workflows. The answers reflect a commitment to transparency and technical excellence, ensuring that partners have a comprehensive understanding of the process capabilities and limitations.
Q: Why is the new synthesis route for 6,6-dimethyl-3-azabicyclo[3.1.0]hexane considered safer than prior art?
A: The novel method eliminates the use of lithium aluminum hydride (LAH) and high-pressure hydrogenation, replacing them with milder reducing agents like sodium borohydride and ambient pressure cyclization, drastically reducing explosion and fire risks.
Q: What are the primary cost drivers reduced in this manufacturing process?
A: By utilizing dichlorochrysanthemic acid as a starting material instead of expensive caronic anhydride, and avoiding noble metal catalysts like palladium-carbon, the process significantly lowers raw material and catalyst recovery costs.
Q: How does this method impact the purity profile of the final antiviral intermediate?
A: The stepwise purification and mild reaction conditions minimize side reactions and degradation, allowing for the production of high-purity intermediates suitable for stringent pharmaceutical applications without complex chromatographic separations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,6-Dimethyl-3-Azabicyclo[3.1.0]Hexane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of life-saving antiviral therapies. Our technical team has extensively analyzed the pathway described in CN113999160B and possesses the expertise to implement this safer, more efficient synthesis at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped to handle the specific unit operations required for this process, including controlled low-temperature reductions and safe hypochlorite oxidations, all while adhering to stringent purity specifications. With our rigorous QC labs and commitment to process safety, we are positioned to be your trusted partner in securing the supply chain for next-generation pharmaceuticals.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your project economics. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to request specific COA data and route feasibility assessments to verify our capabilities firsthand. Let us help you navigate the complexities of antiviral intermediate sourcing with a solution that balances performance, safety, and cost-effectiveness. Contact us today to initiate a dialogue about securing a sustainable and competitive supply of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane for your critical drug development programs.
