Advanced Catalytic Reductive Amination for High-Purity Pharmaceutical Intermediates
Advanced Catalytic Reductive Amination for High-Purity Pharmaceutical Intermediates
The landscape of fine chemical synthesis is continuously evolving towards safer, more efficient, and environmentally benign methodologies. Patent CN1538950A introduces a groundbreaking approach for the preparation of amine compounds through the reductive amination of carbonyl compounds under transfer hydrogenation conditions. This technology represents a significant paradigm shift from traditional high-pressure hydrogenation, utilizing homogeneous transition metal catalysts based on Ruthenium, Rhodium, or Iridium in conjunction with mild hydrogen donors. For R&D directors and process chemists, this patent offers a robust pathway to synthesize complex amine intermediates, including chiral amines, without the need for hazardous high-pressure infrastructure. The ability to operate under mild temperatures and atmospheric pressure not only enhances laboratory safety but also simplifies the scale-up process for commercial manufacturing, making it an attractive option for the production of high-value pharmaceutical intermediates and agrochemical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of amines via reductive amination has relied heavily on catalytic hydrogenation using molecular hydrogen gas. While effective, this conventional approach imposes severe constraints on manufacturing facilities, requiring expensive high-pressure reactors and rigorous safety protocols to manage the explosive nature of hydrogen gas. Furthermore, alternative methods utilizing stoichiometric metal hydrides, such as sodium borohydride or sodium cyanoborohydride, generate substantial amounts of toxic waste and pose significant disposal challenges, conflicting with modern green chemistry principles. Other techniques like the Leuckart-Wallach reaction often necessitate harsh thermal conditions exceeding 150°C, leading to poor selectivity and the formation of undesirable by-products that complicate downstream purification. These legacy methods collectively contribute to elevated operational expenditures and extended lead times, creating bottlenecks in the supply chain for critical chemical intermediates.
The Novel Approach
In stark contrast, the novel methodology described in the patent leverages transfer hydrogenation to overcome these inherent limitations. By employing catalytically active transition metal complexes, specifically those containing metals from the eighth subgroup like Ruthenium or Rhodium, the reaction proceeds efficiently under remarkably mild conditions. The use of organic hydrogen donors, such as ammonium formate, isopropanol, or formic acid-triethylamine mixtures, eliminates the need for gaseous hydrogen, thereby drastically reducing capital investment in pressure-rated equipment. This approach not only accelerates reaction kinetics but also allows for precise control over stereoselectivity when chiral ligands are introduced. The result is a streamlined process that delivers high yields of target amines with minimal by-product formation, offering a compelling solution for cost reduction in API manufacturing and enhancing the overall sustainability of the production lifecycle.
Mechanistic Insights into Transition Metal-Catalyzed Transfer Hydrogenation
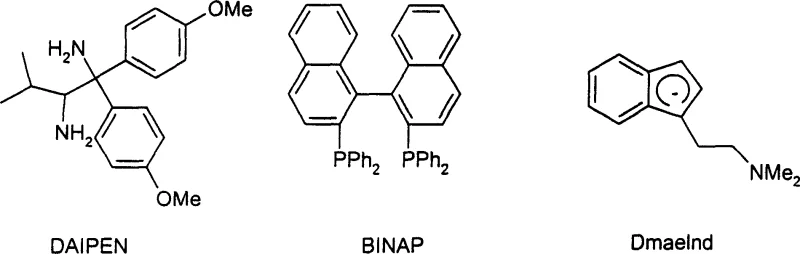
The core of this innovative synthesis lies in the sophisticated interplay between the transition metal center and the coordinating ligands. The catalytic cycle typically involves the formation of a metal-hydride species through the activation of the hydrogen donor, followed by the insertion of the carbonyl substrate and subsequent reaction with the amine source. The choice of ligand is paramount in dictating both the activity and the stereochemical outcome of the transformation. For instance, bidentate phosphine ligands like BINAP or chiral diamines like DAIPEN create a specific chiral environment around the metal center, facilitating the asymmetric induction required for producing optically pure amines. This level of control is essential for pharmaceutical applications where the biological activity is often dependent on a single enantiomer. The versatility of the catalyst system allows it to accommodate a wide range of electronic and steric environments presented by different substrates, ensuring consistent performance across diverse chemical scaffolds.
Furthermore, the mechanism inherently favors the formation of the desired amine over the competing alcohol by-product, a common issue in reduction reactions. The presence of the amine source drives the equilibrium towards imine formation, which is then rapidly reduced by the metal-hydride species. This kinetic preference ensures a high amine-to-alcohol ratio in the final product mixture, simplifying the purification process and improving overall material throughput. Impurity control is further enhanced by the mild reaction conditions, which prevent the degradation of sensitive functional groups that might otherwise decompose under the harsh conditions of traditional methods. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as catalyst loading and temperature, to maximize efficiency and minimize waste generation, aligning with the stringent quality standards required for regulatory compliance in the pharmaceutical industry.
How to Synthesize Chiral Amines Efficiently
The practical implementation of this transfer hydrogenation protocol involves a straightforward sequence of operations that can be easily adapted for both laboratory and pilot-scale production. Typically, the catalyst is generated either in situ by mixing the metal precursor with the ligand in a suitable solvent or pre-formed and added as a purified complex. The substrate, amine source, and hydrogen donor are then introduced, and the mixture is heated to the optimal temperature range specified for the specific catalyst system. Detailed standard operating procedures regarding specific molar ratios, solvent choices, and work-up protocols are critical for reproducibility and yield optimization. For a comprehensive guide on executing this synthesis with precision, please refer to the standardized steps outlined below.
- Prepare the catalytic system by reacting a transition metal precursor (such as Ru, Rh, or Ir salts) with specific chiral or achiral ligands like BINAP or DAIPEN in an inert solvent.
- Combine the carbonyl substrate (ketone or aldehyde) with the amine source (ammonia, primary, or secondary amine) and a hydrogen donor such as ammonium formate or isopropanol.
- Heat the reaction mixture to mild temperatures ranging from 0°C to 110°C and stir for several hours to achieve high conversion and optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this transfer hydrogenation technology offers profound benefits for procurement managers and supply chain leaders seeking to optimize their sourcing strategies. The elimination of high-pressure hydrogenation equipment translates directly into significant capital expenditure savings, as facilities do not require specialized autoclaves or extensive safety infrastructure. Moreover, the use of stable, non-gaseous hydrogen donors simplifies logistics and storage, reducing the risks associated with handling hazardous materials. This operational simplicity enhances supply chain reliability by minimizing downtime related to equipment maintenance or safety inspections, ensuring a more consistent flow of materials to downstream processes. The ability to produce high-purity intermediates with fewer purification steps also contributes to substantial cost savings in terms of solvent usage and waste disposal fees.
- Cost Reduction in Manufacturing: The transition from high-pressure hydrogenation to transfer hydrogenation removes the dependency on expensive pressure-rated reactors and complex gas handling systems, leading to a drastic simplification of the manufacturing setup. By utilizing inexpensive and readily available hydrogen donors like ammonium formate, the process reduces raw material costs while simultaneously lowering energy consumption due to milder reaction temperatures. This cumulative effect results in a highly competitive cost structure for the final amine products, allowing manufacturers to offer better pricing to their clients without compromising on quality or margin.
- Enhanced Supply Chain Reliability: The reliance on stable liquid reagents rather than compressed gases mitigates supply chain vulnerabilities associated with the transportation and storage of hazardous materials. Since the catalysts and ligands used are often commercially available or easily synthesized, the risk of raw material shortages is significantly minimized. This stability ensures that production schedules can be maintained with greater predictability, reducing lead times for high-purity pharmaceutical intermediates and enabling manufacturers to respond more agilely to fluctuating market demands. The robustness of the process also means fewer batch failures, further securing the continuity of supply for critical customers.
- Scalability and Environmental Compliance: The homogeneous nature of the catalytic system facilitates seamless scale-up from gram-scale laboratory experiments to multi-ton commercial production without the need for fundamental process redesign. The mild conditions and reduced waste generation align perfectly with increasingly stringent environmental regulations, lowering the burden of effluent treatment and disposal. This environmental compatibility not only reduces compliance costs but also enhances the corporate sustainability profile, making the supply chain more resilient to future regulatory changes. The process efficiency ensures that complex amine structures can be manufactured at scale with consistent quality, supporting the long-term growth of the business.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this reductive amination technology. These insights are derived directly from the patent specifications and are intended to clarify the operational capabilities and advantages of the method for potential partners and stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What are the primary safety advantages of this transfer hydrogenation method compared to traditional hydrogenation?
A: Unlike conventional catalytic hydrogenation which requires high-pressure hydrogen gas and specialized explosion-proof equipment, this method utilizes safe, liquid hydrogen donors like ammonium formate or isopropanol under atmospheric or low pressure, significantly reducing operational risks and infrastructure costs.
Q: Can this process achieve high enantioselectivity for chiral amine production?
A: Yes, by employing chiral transition metal complexes containing ligands such as BINAP, DAIPEN, or Norphos, the process enables the stereoselective synthesis of optically pure amines with high ee values, which is critical for pharmaceutical applications.
Q: What types of substrates are compatible with this reductive amination protocol?
A: The method demonstrates broad substrate scope, successfully converting various aromatic and aliphatic ketones and aldehydes, including functionalized substrates like naphthyl ketones and ferrocene derivatives, into their corresponding amine products with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN1538950A for the production of high-value chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from concept to reality. Our state-of-the-art facilities are equipped to handle complex homogeneous catalysis reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards. We are committed to delivering reliable solutions that drive innovation in the pharmaceutical and fine chemical sectors.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, demonstrating how this technology can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and efficient supply chain for your critical amine intermediates.
