Advanced Catalytic Routes for High-Purity Heterocyclic Hydroxyamine Intermediates
The pharmaceutical industry continuously seeks robust methodologies for synthesizing chiral intermediates, particularly for antidepressants like duloxetine. Patent CN1694878A introduces a groundbreaking process for the preparation of heterocyclic hydroxyamine compounds, specifically targeting the efficient synthesis of key pharmaceutical intermediates. This technology leverages advanced asymmetric transfer hydrogenation to achieve high optical purity without the limitations of traditional resolution techniques. By utilizing specialized transition metal catalysts, manufacturers can access complex chiral structures with superior stereocontrol. The following analysis explores the technical depth and commercial viability of this patented route for producing high-purity pharmaceutical intermediates.
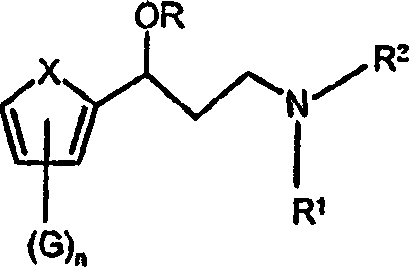
The limitations of conventional methods often revolve around the inefficiency of resolving racemic mixtures, which inherently caps the maximum yield at 50% and generates significant waste. Traditional approaches frequently rely on harsh reducing agents or multi-step protection-deprotection sequences that increase operational complexity and cost. In contrast, the novel approach described in the patent utilizes a streamlined two-step sequence involving amidation followed by reduction. This method bypasses the need for chiral resolution by installing chirality early in the synthesis via catalytic asymmetric reduction. The use of transfer hydrogenation eliminates the safety hazards associated with high-pressure hydrogen gas, allowing for safer operation in standard reactor vessels while maintaining exceptional conversion rates.
Mechanistic insights into the Rhodium-Catalyzed Asymmetric Transfer Hydrogenation reveal the critical role of the chiral ligand environment in dictating stereoselectivity. The catalyst system typically comprises a rhodium center coordinated with a pentamethylcyclopentadienyl ligand and a chiral diamine, often modified with a bulky chiral sulfonyl group such as camphorsulfonyl. This specific architecture creates a rigid chiral pocket that directs the hydride transfer from the donor (e.g., formic acid/triethylamine azeotrope) to the prochiral ketone face with high fidelity. The interaction between the substrate and the catalyst ensures that the reduction proceeds with >95% e.e., effectively suppressing the formation of the unwanted enantiomer. This level of control is essential for meeting the stringent impurity profiles required by global regulatory agencies for active pharmaceutical ingredients.
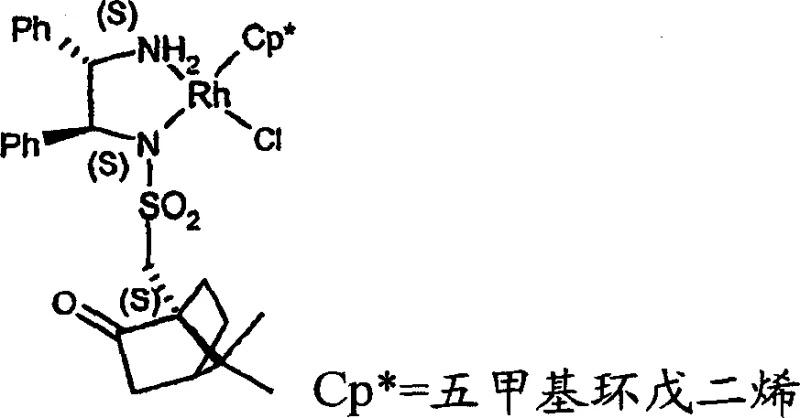
Understanding the impurity control mechanism is vital for R&D teams aiming to replicate this success. The choice of hydrogen donor and the specific ratio of formic acid to triethylamine (optimally 5:2) plays a pivotal role in minimizing side reactions such as over-reduction or ester hydrolysis. Furthermore, the use of non-nucleophilic bases during the initial acylation step prevents unwanted polymerization or degradation of the sensitive thiophene ring. By strictly controlling the temperature profile, typically keeping the reduction between 0°C and 50°C, the process avoids thermal degradation pathways. This precise control over reaction parameters ensures that the final product meets the rigorous quality standards necessary for downstream drug substance manufacturing.
How to Synthesize Heterocyclic Hydroxyamines Efficiently
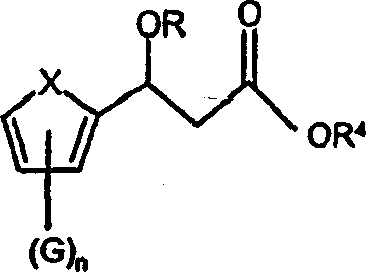
The synthesis of these valuable intermediates begins with the preparation of a beta-keto ester, followed by the crucial asymmetric reduction step. The process is designed to be operationally simple yet chemically sophisticated, allowing for the direct generation of chiral alcohols from achiral ketones. Detailed standardized synthesis steps are provided in the guide below, outlining the specific reagents, stoichiometry, and workup procedures required to achieve the reported yields and purity levels. This section serves as a technical roadmap for process chemists looking to implement this technology in their own laboratories.
- Prepare the beta-keto ester intermediate by acylating 2-acetylthiophene with dialkyl carbonate in the presence of a strong base like sodium hydride.
- Perform asymmetric transfer hydrogenation using a chiral rhodium catalyst and a hydrogen donor mixture such as formic acid and triethylamine to establish stereochemistry.
- Convert the resulting chiral ester to the amide using methylamine, followed by final reduction with lithium aluminum hydride to yield the target hydroxyamine.
Commercial Advantages for Procurement and Supply Chain Teams
This patented technology offers substantial benefits for procurement and supply chain management by fundamentally altering the cost structure of chiral intermediate production. The shift from resolution-based methods to asymmetric catalysis removes the inherent 50% yield penalty, effectively doubling the output from the same amount of raw materials. This improvement in atom economy translates directly into lower raw material costs and reduced waste disposal expenses. Additionally, the avoidance of high-pressure hydrogenation equipment reduces capital expenditure requirements for manufacturing facilities, making the technology accessible to a wider range of contract manufacturing organizations.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps significantly lowers processing costs by removing the need for multiple crystallization cycles and the disposal of the unwanted enantiomer. The use of transfer hydrogenation with formic acid is also more cost-effective than purchasing high-purity hydrogen gas and maintaining specialized high-pressure reactors. These factors combine to create a leaner manufacturing process with a significantly reduced cost of goods sold, providing a competitive edge in pricing negotiations for bulk pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reagents required for this process, such as dialkyl carbonates and common amine sources, are commodity chemicals with stable global supply chains. Unlike specialized chiral pool starting materials which can suffer from supply volatility, the catalysts described can be synthesized from readily available precursors. This reliance on abundant raw materials ensures consistent production schedules and minimizes the risk of supply disruptions, allowing procurement managers to secure long-term contracts with greater confidence in delivery timelines.
- Scalability and Environmental Compliance: The process is inherently scalable due to the mild reaction conditions and the absence of hazardous high-pressure gases. Transfer hydrogenation generates benign byproducts like carbon dioxide and water, simplifying effluent treatment and reducing the environmental footprint of the manufacturing site. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the supply chain more resilient against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and scope of the technology. Understanding these details is crucial for stakeholders evaluating the potential integration of this method into their existing production portfolios.
Q: What is the primary advantage of the catalytic method in CN1694878A over traditional resolution?
A: The primary advantage is the ability to directly synthesize the desired enantiomer with high optical purity (up to 95% e.e. or higher) without the 50% theoretical yield loss associated with racemic resolution, significantly improving atom economy.
Q: Which catalysts are preferred for this asymmetric transfer hydrogenation process?
A: The patent specifies Rhodium, Ruthenium, or Iridium complexes containing chiral diamine ligands, particularly those substituted with chiral sulfonyl groups like camphorsulfonyl, which provide excellent stereocontrol.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes transfer hydrogenation which avoids high-pressure hydrogen gas equipment, making it safer and easier to scale in standard chemical reactors while maintaining high conversion rates above 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclic Hydroxyamine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patent technologies into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of heterocyclic hydroxyamine intermediate meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of asymmetric catalysis with precision and reliability.
We invite you to collaborate with us to optimize your supply chain for these critical pharmaceutical building blocks. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's success and reduce your overall time to market.
