Advanced Catalytic Hydrogenolysis for High-Purity Famciclovir Production and Commercial Scalability
Introduction to Advanced Famciclovir Manufacturing Technologies
The pharmaceutical industry continuously seeks robust synthetic routes for antiviral agents that balance high purity with economic viability. Patent CN1812990A presents a significant technological advancement in the preparation of famciclovir, a prodrug of penciclovir widely used for treating herpes zoster and genital herpes. This disclosure addresses critical challenges associated with impurity profiles in earlier synthetic methodologies, specifically targeting the reduction of hydrolytic by-products that complicate downstream purification. By shifting from traditional alcoholic solvent systems to optimized alkyl acetate-based media, the process achieves exceptional control over the reaction pathway. This innovation is particularly relevant for a reliable pharmaceutical intermediate supplier aiming to deliver materials that meet stringent regulatory standards without excessive processing costs. The structural integrity of the final active pharmaceutical ingredient is paramount, as illustrated by the core molecular architecture below.
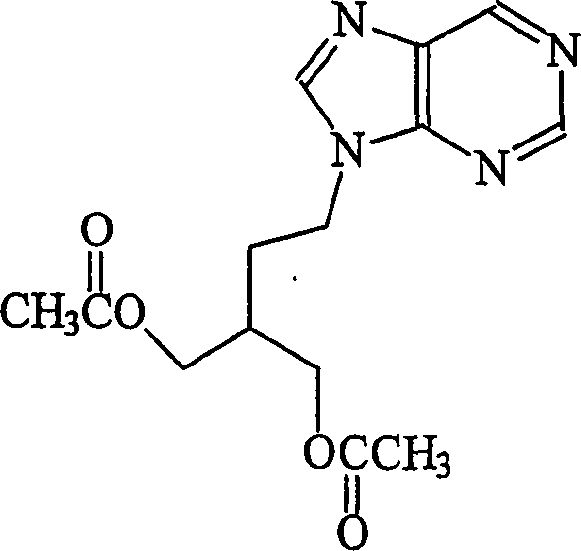
The disclosed methodology leverages transfer hydrogenation using ammonium formate and a heterogeneous palladium catalyst, eliminating the need for high-pressure hydrogen gas equipment in certain configurations. This approach not only enhances operational safety but also simplifies the engineering requirements for commercial scale-up of complex nucleoside analogs. For R&D directors evaluating process feasibility, the ability to consistently produce material with monohydroxy impurity levels below 0.1% represents a substantial quality improvement over prior art, directly impacting the cost of goods sold through reduced waste and higher yields.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of famciclovir involved the hydrogenolysis of chloro-precursors in methanol solvent systems, as described in earlier patents such as US 5,246,937. While effective for conversion, these conventional methods suffered from a significant drawback: the formation of unacceptable levels of hydrolytic impurities. Specifically, the use of pure methanol often led to the generation of monohydroxy-famciclovir and dihydroxy-famciclovir in quantities exceeding 3%. These impurities arise from the cleavage of the acetate protecting groups under the reaction conditions, a side reaction that is difficult to reverse once initiated. The presence of these polar impurities complicates the isolation process, often necessitating extensive chromatographic purification or multiple recrystallization steps to meet pharmacopeial standards. Furthermore, the variability in impurity profiles can lead to batch failures, creating uncertainty in supply chains and increasing the overall cost reduction in antiviral drug manufacturing efforts. The chemical structures of these problematic by-products are depicted below, highlighting the loss of acetyl groups that defines this degradation pathway.
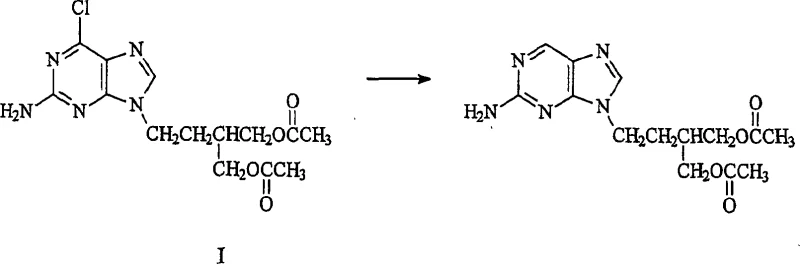
The Novel Approach
The process detailed in CN1812990A overcomes these limitations by fundamentally altering the solvent environment. By employing C1-C6 alkyl acetates, such as ethyl acetate or methyl acetate, either alone or in specific mixtures with lower alcohols, the reaction medium becomes less conducive to hydrolysis. Experimental data within the patent demonstrates that switching to an ethyl acetate system can reduce monohydroxy-famciclovir levels to less than 0.1%, and preferably below 0.03%. This drastic reduction in impurity load means that the crude product is of significantly higher quality immediately after filtration. For procurement managers, this translates to a more predictable supply of high-purity famciclovir that requires less intensive post-reaction processing. The novel approach maintains high conversion rates while preserving the delicate acetate esters, ensuring that the final API meets strict specifications with minimal additional effort, thereby streamlining the entire production workflow.
Mechanistic Insights into Pd/C Catalyzed Transfer Hydrogenolysis
The core of this synthetic strategy relies on a heterogeneous catalytic cycle involving palladium on charcoal (Pd/C) and ammonium formate as the hydrogen donor. In this mechanism, ammonium formate decomposes on the surface of the palladium catalyst to generate reactive hydrogen species in situ. These species then facilitate the reductive dechlorination of the imidazo[4,5-b]pyridine precursor (Cl-FMC). The choice of solvent plays a critical mechanistic role beyond merely dissolving the reactants; alkyl acetates act as a protective buffer against nucleophilic attack by methanol or water, which would otherwise lead to transesterification or hydrolysis. By maintaining a solvent environment that matches the ester functionality of the substrate, the thermodynamic drive towards deacetylation is minimized. This subtle yet powerful solvent effect allows the reduction of the carbon-chlorine bond to proceed selectively without compromising the integrity of the side-chain esters. Understanding this interplay is vital for process chemists aiming to replicate these results at scale, as even minor deviations in solvent composition can revert the system to the high-impurity regime observed in older methodologies.
Furthermore, the stability of the catalyst and the reaction kinetics are optimized within a temperature window of 40-70°C. At these moderate temperatures, the rate of hydrogen generation from ammonium formate is balanced with the rate of substrate reduction, preventing the accumulation of reactive intermediates that could lead to side reactions. The patent data indicates that both wet (containing approximately 50% water) and dry Pd/C catalysts are effective, suggesting that the surface chemistry of the palladium is robust enough to tolerate varying moisture levels without losing activity. This flexibility is advantageous for reducing lead time for high-purity API intermediates, as it removes the strict requirement for anhydrous catalyst handling, simplifying logistics and storage. The result is a clean reaction profile where the primary product dominates the HPLC trace, facilitating straightforward isolation via filtration and crystallization.
How to Synthesize Famciclovir Efficiently
Implementing this optimized route requires careful attention to solvent ratios and catalyst loading to maximize the benefits of impurity suppression. The general procedure involves charging a reactor with the chloro-precursor, the palladium catalyst, and ammonium formate in the selected alkyl acetate solvent system. The mixture is then heated under inert atmosphere to drive the reaction to completion. Detailed operational parameters, including specific molar equivalents and timing for reagent addition, are critical for reproducibility. The following guide outlines the standardized steps derived from the patent examples to ensure consistent high-quality output.
- Charge a reactor with the chloro-precursor (Cl-FMC), palladium on charcoal catalyst (5-10% w/w), and ammonium formate in a solvent system comprising C1-C6 alkyl acetates.
- Heat the reaction mixture to a controlled temperature range of 40-70°C under inert atmosphere to facilitate transfer hydrogenation.
- Filter off the catalyst after complete conversion, concentrate the filtrate, and crystallize the product from n-butanol to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the technical improvements in this patent translate directly into tangible commercial benefits. The primary advantage lies in the simplification of the purification train. By drastically lowering the initial burden of hydrolytic impurities, manufacturers can potentially eliminate expensive and time-consuming chromatographic separation steps. This reduction in unit operations not only lowers direct manufacturing costs but also increases the throughput capacity of existing facilities. Additionally, the use of common solvents like ethyl acetate and methyl acetate ensures that raw material sourcing remains stable and cost-effective, avoiding reliance on specialized or volatile reagents. This stability is crucial for maintaining a resilient supply chain in the face of global market fluctuations.
- Cost Reduction in Manufacturing: The elimination of complex purification stages significantly decreases operational expenditures. By achieving high purity directly from the reaction mixture, the process reduces solvent consumption, energy usage for distillation, and labor hours associated with extended workups. The ability to use wet catalysts further reduces preparation costs, as drying steps are rendered unnecessary. These cumulative efficiencies contribute to a more competitive cost structure for the final API, allowing for better margin management in a price-sensitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, particularly the tolerance for wet catalysts and moderate temperatures, reduces the risk of batch failures due to sensitive handling requirements. This reliability ensures consistent delivery schedules, which is a key performance indicator for supply chain heads. Moreover, the stability of the final product under various storage conditions (up to 55°C and 75% RH) minimizes the risk of degradation during transit and warehousing, ensuring that the material received by customers meets specification upon arrival without the need for re-testing or rejection.
- Scalability and Environmental Compliance: The process utilizes heterogeneous catalysis, which simplifies catalyst recovery and disposal compared to homogeneous systems. The solvents employed are standard industrial chemicals with well-established recycling protocols, supporting green chemistry initiatives. The scalability is demonstrated by the successful execution of reactions at multi-hundred gram scales in the patent examples, indicating a clear path to tonnage production. This ease of scale-up allows manufacturers to respond quickly to surges in demand for antiviral medications without requiring significant capital investment in new reactor types or safety systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this famciclovir synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps bridge the gap between laboratory research and industrial application, ensuring that all stakeholders have a clear picture of the process capabilities and limitations.
Q: How does the new solvent system improve famciclovir purity compared to traditional methanol methods?
A: Traditional methods using pure methanol often result in high levels of monohydroxy-famciclovir (>3%) due to hydrolysis. The patented process utilizes alkyl acetates (like ethyl acetate) or specific mixtures, which significantly suppress this hydrolysis, reducing impurity levels to below 0.1%.
Q: What catalyst loading is required for efficient conversion in this process?
A: The process typically employs palladium on charcoal (Pd/C) catalyst at a loading of approximately 5-10% (w/w) relative to the substrate. Both wet (50% water content) and dry forms of the catalyst are effective, offering flexibility in procurement and handling.
Q: Is the resulting famciclovir stable under long-term storage conditions?
A: Yes, stability studies indicate that the product remains stable for at least 6 months at relative humidity of 75% and temperatures up to 55°C, with no significant increase in monohydroxy impurity content, ensuring reliable shelf-life for inventory management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Famciclovir Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and process efficiency in the production of antiviral intermediates. Our technical team has extensively analyzed the advancements presented in CN1812990A and integrated similar high-efficiency catalytic strategies into our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full commercialization is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch of famciclovir we supply meets the highest international standards for pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your supply chain goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our specific route optimizations compare to your current sourcing strategies. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a partnership built on transparency, quality, and mutual growth.
