Advanced Biocatalytic Synthesis of Optically Active Beta-Amino Alcohols for Pharmaceutical Scale-Up
The pharmaceutical industry continuously seeks robust methodologies for the production of chiral intermediates, particularly optically active beta-amino alcohols which serve as critical scaffolds for sympathomimetic drugs and other therapeutic agents. Patent CN1282746C discloses a groundbreaking biocatalytic approach that addresses the longstanding challenges associated with the stereoselective synthesis of these compounds. Unlike traditional chemical routes that often struggle with poor diastereoselectivity and require cumbersome resolution steps, this invention leverages the exquisite specificity of microbial enzymes to transform alpha-aminoketone precursors directly into the desired optical isomers. By utilizing a diverse library of microorganisms ranging from Morganella and Microbacterium to Rhodococcus and Amycolatopsis, the process achieves high yields and exceptional optical purity under mild, environmentally benign conditions. This technical insight report analyzes the mechanistic advantages and commercial implications of this biotechnology, providing a strategic overview for R&D directors and procurement leaders seeking to optimize their supply chains for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of optically active beta-amino alcohols, such as ephedrine and pseudoephedrine, has relied heavily on chemical synthesis pathways that are inherently inefficient regarding stereochemical control. Conventional methods typically involve the reduction of alpha-aminoketones using chemical reducing agents, which frequently results in the formation of racemic mixtures or complex blends of diastereomers. Because the target molecules possess two asymmetric carbon atoms, obtaining a single specific optical isomer necessitates intricate optical resolution processes, often involving the formation of diastereomeric salts followed by repeated recrystallizations. These traditional procedures are not only labor-intensive and time-consuming but also suffer from significant material loss, as the unwanted isomers are often discarded or require energy-intensive recycling loops. Furthermore, the use of harsh chemical reagents and heavy metal catalysts in these legacy processes introduces substantial environmental liabilities and complicates the removal of toxic impurities, posing risks to product safety and regulatory compliance in the highly scrutinized pharmaceutical sector.
The Novel Approach
In stark contrast to these archaic techniques, the methodology described in patent CN1282746C introduces a paradigm shift by employing specific microorganisms to effectuate a highly stereoselective bioreduction. This novel approach capitalizes on the natural enzymatic machinery of bacteria and fungi to selectively reduce only one enantiomer of the racemic alpha-aminoketone substrate, or to dynamically resolve the mixture in situ, thereby generating the target beta-amino alcohol with superior optical purity. The process bypasses the need for external chiral auxiliaries or complex resolution steps, streamlining the synthesis into a single, efficient biotransformation stage. By selecting from a vast array of microbial strains, manufacturers can tailor the reaction to produce specific isomers, such as the (1S,2S) or (1R,2R) configurations, simply by choosing the appropriate biological catalyst. This biological precision ensures that the formation of unwanted diastereomeric by-products is sufficiently prevented, leading to a cleaner reaction profile and a drastically simplified downstream processing workflow.
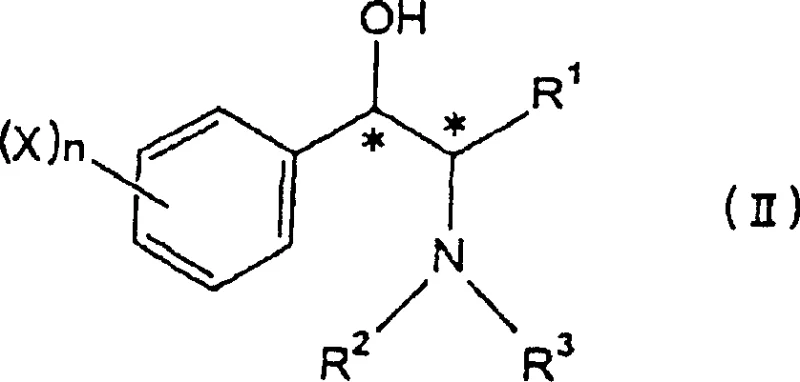
Mechanistic Insights into Microbial Stereoselective Reduction
The core mechanism driving this innovation lies in the substrate specificity and stereoselectivity of the oxidoreductase enzymes present within the selected microbial cells. When the racemic alpha-aminoketone compound, represented by general formula (I), interacts with the microbial biomass, specific dehydrogenases recognize and bind to only one enantiomeric form of the substrate due to the chiral nature of the enzyme's active site. This enzymatic recognition facilitates the transfer of hydride equivalents, typically derived from cofactors like NADH or NADPH, to the carbonyl group of the ketone, reducing it to a hydroxyl group with strict stereochemical control. The patent highlights that different genera of microorganisms exhibit distinct selectivities; for instance, strains belonging to Morganella and Microbacterium tend to produce (1S,2S)-beta-amino alcohols, whereas species from Amycolatopsis and Rhodotorula favor the formation of (1R,2R) isomers. This tunability allows for precise control over the absolute configuration of the final product, enabling the synthesis of either ephedrine or pseudoephedrine derivatives from the same starting material simply by switching the biocatalyst.
Furthermore, the process incorporates sophisticated impurity control mechanisms inherent to the biological system. The high specificity of the microbial enzymes ensures that side reactions, such as the reduction of other functional groups or the formation of regioisomers, are minimized. The patent data indicates that by optimizing culture conditions, such as pH levels between 5 and 9 and temperatures ranging from 20°C to 40°C, the metabolic activity of the cells can be directed almost exclusively towards the desired transformation. Additionally, the inclusion of activity inducers, such as 1-amino-2-propanol or specific salts like phosphates, can promote the racemization of the unreacted substrate enantiomer, effectively allowing for a dynamic kinetic resolution where the theoretical yield can exceed the standard 50% limit of classical kinetic resolutions. This mechanistic elegance results in a product stream with high optical purity, often exceeding 95% ee, significantly reducing the burden on analytical quality control and purification units.
How to Synthesize Optically Active Beta-Amino Alcohols Efficiently
Implementing this biocatalytic route requires a systematic approach to microbial cultivation and biotransformation to ensure consistent quality and yield. The process begins with the selection of a high-performance strain, such as Rhodococcus erythropolis or Microbacterium arborescens, followed by optimization of the growth medium to maximize cell density and enzymatic activity. Once the biomass is harvested, it is suspended in a buffered aqueous solution where the substrate is introduced under controlled conditions to initiate the reduction. The detailed standardized synthesis steps, including specific media compositions, induction protocols, and workup procedures derived from the patent examples, are outlined below to guide process development teams in replicating this high-efficiency route.
- Cultivate selected microorganisms (e.g., Rhodococcus erythropolis or Microbacterium arborescens) in a nutrient medium containing carbon and nitrogen sources at 20-40°C.
- Harvest the microbial cells and suspend them in a buffer solution, optionally adding activity inducers like 1-amino-2-propanol to enhance enzymatic efficiency.
- React the suspended cells with the racemic alpha-aminoketone substrate at pH 5-9 and 20-40°C, followed by separation and purification to isolate the desired optical isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic technology offers transformative benefits that extend far beyond mere technical feasibility. The shift from multi-step chemical synthesis to a streamlined microbial process fundamentally alters the cost structure and risk profile of manufacturing these critical intermediates. By eliminating the need for expensive chiral resolving agents, heavy metal catalysts, and energy-intensive purification stages, the overall cost of goods sold is significantly reduced. This economic efficiency is compounded by the use of readily available, renewable raw materials and aqueous reaction media, which lowers the dependency on volatile petrochemical feedstocks and mitigates exposure to fluctuating commodity prices. Consequently, this technology enables a more stable and predictable pricing model for long-term supply agreements, providing a strategic advantage in a competitive market.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic simplification of the production workflow. Traditional chemical routes often incur high costs due to low overall yields resulting from multiple resolution steps and the disposal of unwanted isomers. In contrast, the microbial method described in the patent achieves high selectivity in a single reaction vessel, effectively consolidating multiple unit operations into one. This consolidation reduces capital expenditure on equipment, lowers utility consumption for heating and cooling, and minimizes waste disposal costs associated with organic solvents and heavy metals. Furthermore, the ability to recycle unreacted substrates through in-situ racemization enhances atom economy, ensuring that a greater proportion of raw material input is converted into valuable saleable product, thereby maximizing return on investment for manufacturing facilities.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness and scalability of the fermentation-based process. Unlike chemical synthesis which may rely on specialized reagents with limited global availability, the microorganisms used in this invention can be cultivated using standard industrial fermentation infrastructure that is widely accessible. The mild reaction conditions reduce the risk of safety incidents and equipment corrosion, leading to higher plant uptime and fewer unplanned shutdowns. Additionally, the stability of the microbial strains allows for the creation of master cell banks, ensuring batch-to-batch consistency and long-term supply continuity. This reliability is crucial for pharmaceutical customers who require guaranteed delivery schedules to maintain their own production timelines for finished dosage forms, making this biocatalytic route a preferred choice for securing critical API intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this green chemistry approach aligns perfectly with modern sustainability goals and stringent regulatory standards. The process operates in water-based systems, drastically reducing the volume of volatile organic compounds (VOCs) emitted into the atmosphere and minimizing the generation of hazardous liquid waste. The absence of toxic heavy metals simplifies the purification of the final product, ensuring that residual impurity levels easily meet the rigorous specifications required for pharmaceutical ingredients. This inherent cleanliness facilitates faster regulatory approvals and reduces the environmental compliance burden on manufacturing sites. Moreover, the process is inherently scalable; the linear relationship between fermentation volume and product output allows for seamless expansion from pilot plant trials to multi-ton commercial production without the need for complex process re-engineering, supporting rapid market entry and capacity scaling.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microbial synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN1282746C, providing clarity on the operational parameters and expected outcomes of the process. Understanding these details is essential for technical teams evaluating the feasibility of integrating this biocatalytic route into their existing manufacturing portfolios.
Q: How does this biocatalytic method improve upon traditional chemical synthesis for beta-amino alcohols?
A: Traditional chemical methods often produce racemic mixtures or diastereomers requiring complex and costly resolution steps. This patented microbial process utilizes specific enzyme systems to selectively reduce only one enantiomer of the alpha-aminoketone precursor, directly yielding the desired optically active beta-amino alcohol with high stereochemical purity, thereby eliminating the need for difficult downstream separation.
Q: What represents the primary cost driver reduction in this manufacturing process?
A: The primary cost reduction stems from the elimination of expensive chiral resolving agents and the simplification of the purification workflow. By achieving high optical purity directly through fermentation or biotransformation, manufacturers avoid the yield losses typically associated with recrystallization and chromatographic separation of diastereomeric salts found in conventional synthetic routes.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable as it operates under mild aqueous conditions (pH 5-9, 20-40°C) using standard fermentation equipment. The use of robust microbial strains such as Rhodococcus and Microbacterium allows for high cell density cultivation, facilitating the transition from laboratory bench-scale reactions to multi-ton commercial production without the safety hazards associated with high-pressure chemical hydrogenation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Alcohols Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic processes detailed in patent CN1282746C for the production of high-value chiral intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory findings into robust, commercial-scale manufacturing solutions. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench to plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify optical purity and impurity profiles against the highest international standards. Our state-of-the-art fermentation facilities are equipped to handle the specific microbial strains and culture conditions required for this technology, guaranteeing a consistent and reliable supply of optically active beta-amino alcohols for your pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this cost-effective and sustainable manufacturing route into your supply chain. Let NINGBO INNO PHARMCHEM be your partner in driving innovation and efficiency in the production of complex pharmaceutical intermediates.
