Advanced Asymmetric Hydrogenation Technology for High-Purity Beta-Amino Alcohol Intermediates
Advanced Asymmetric Hydrogenation Technology for High-Purity Beta-Amino Alcohol Intermediates
The pharmaceutical industry continuously seeks more efficient pathways to access chiral building blocks, particularly for central nervous system (CNS) active compounds. Patent CN1842523A introduces a groundbreaking methodology for the preparation of enantiomerically enriched or pure (S)- or (R)-N-monosubstituted beta-amino alcohols of formula (I). This technology represents a paradigm shift from conventional racemic synthesis and resolution strategies, offering a direct route to high-value intermediates such as those used in the production of Duloxetine. By leveraging advanced transition metal catalysis, this process addresses critical pain points in modern drug substance manufacturing, including step count reduction and waste minimization. For R&D directors and procurement specialists, understanding the mechanistic elegance and commercial viability of this asymmetric hydrogenation is essential for optimizing supply chains and reducing the cost of goods sold (COGS) for complex APIs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-amino alcohols has relied heavily on the preparation of racemic mixtures followed by classical resolution techniques, which inherently cap the maximum theoretical yield at 50%. Furthermore, established synthetic routes often necessitate the use of amino-protecting groups, such as benzyl or methyl groups, to prevent catalyst poisoning or side reactions during the reduction phase. These protecting groups must be installed in early stages and subsequently removed in late-stage processing, adding at least two additional synthetic steps to the overall sequence. This not only increases the consumption of reagents and solvents but also generates significant chemical waste, complicating environmental compliance and driving up production costs. Additionally, the harsh conditions often required for deprotection can lead to product degradation or racemization, compromising the optical purity required for stringent regulatory standards in pharmaceutical applications.
The Novel Approach
In stark contrast, the methodology disclosed in CN1842523A enables the direct asymmetric hydrogenation of N-monosubstituted beta-amino ketones without the need for any amino-protecting groups. This innovative approach streamlines the synthetic pathway by converting the ketone precursor directly into the desired chiral alcohol in a single catalytic step. The elimination of protection and deprotection sequences drastically reduces the number of unit operations, leading to a substantial improvement in overall process efficiency and throughput. By utilizing specific chiral bidentate phosphine ligands in conjunction with rhodium or ruthenium catalysts, the process achieves exceptional stereocontrol, routinely delivering products with enantiomeric excess (ee) values exceeding 99%. This direct route not only enhances the economic profile of the manufacturing process but also aligns perfectly with green chemistry principles by minimizing waste generation and energy consumption.

Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the sophisticated design of the catalyst system, which employs cationic transition metal complexes coordinated with chiral bidentate phosphine ligands. The patent highlights the efficacy of ligands belonging to the DuPhos and KetalPhos families, such as (S,S)-Me-DuPhos and (S,S,S,S)-Me-KetalPhos, which create a highly defined chiral environment around the metal center. When the N-monosubstituted beta-amino ketone substrate coordinates to the rhodium or ruthenium center, the steric bulk and electronic properties of these ligands dictate the facial selectivity of the hydride transfer. This precise spatial arrangement ensures that hydrogen addition occurs exclusively from one face of the carbonyl group, thereby locking in the desired stereochemistry with high fidelity. The presence of the free secondary amine in the substrate, which typically acts as a catalyst poison in other systems, is effectively managed through the specific electronic tuning of the catalyst and the use of appropriate bases.
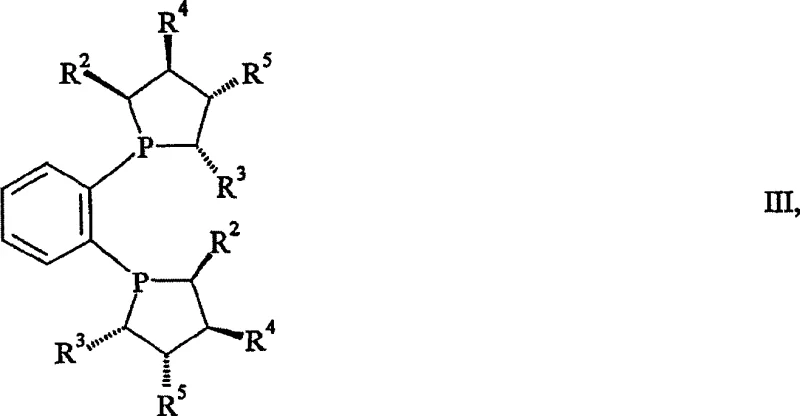
Furthermore, the mechanism involves the formation of a stable substrate-catalyst complex that facilitates rapid turnover while maintaining rigorous stereochemical control throughout the reaction. The addition of alkali bases, such as sodium methoxide or potassium hydroxide, plays a dual role by neutralizing acid salts present in the starting material and promoting the formation of the active catalytic species. Experimental data from the patent indicates that operating within a temperature range of 30°C to 50°C and hydrogen pressures of 10 to 30 bar provides the optimal balance between reaction rate and enantioselectivity. This robustness against varying reaction parameters suggests a wide operating window, which is a critical factor for ensuring consistent product quality during large-scale commercial production. The ability to achieve >99% ee without recrystallization underscores the potency of this catalytic system in controlling impurity profiles at the molecular level.
How to Synthesize (S)-3-N-methylamino-1-(2-thienyl)-1-propyl alcohol Efficiently
The synthesis of this critical duloxetine intermediate begins with the preparation of the beta-amino ketone precursor via a Mannich-type condensation, followed by the pivotal asymmetric hydrogenation step. The process is designed to be operationally simple, utilizing common polar solvents like methanol or ethanol and standard hydrogenation equipment found in most pilot and production plants. Detailed below is the strategic workflow that leverages the patented catalyst system to achieve high yields and optical purity. For laboratory and pilot-scale execution, precise control over the catalyst-to-substrate ratio and the addition of base is essential to maximize the efficiency of the chiral induction.
- Preparation of the N-monosubstituted beta-amino ketone substrate via Mannich-type condensation of methyl ketones, formaldehyde, and primary amines.
- In-situ formation of the catalyst complex by mixing a transition metal precursor (Rh or Ru) with a chiral bidentate phosphine ligand such as Me-DuPhos or Me-KetalPhos in a polar solvent.
- Execution of asymmetric hydrogenation under controlled pressure (10-30 bar) and temperature (30-50°C) in the presence of a base to yield the target chiral alcohol with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric hydrogenation technology translates into tangible strategic benefits that extend beyond mere technical superiority. The most significant advantage is the drastic simplification of the supply chain, as the elimination of protecting group chemistry removes the need for sourcing and handling hazardous reagents associated with protection and deprotection steps. This reduction in raw material complexity directly correlates to a lower risk of supply disruption and a simplified inventory management process. Moreover, the high atom economy of the direct hydrogenation route means that less raw material is required to produce the same amount of final product, leading to significant cost reduction in pharmaceutical intermediate manufacturing. The process operates under relatively mild conditions, which reduces energy consumption and extends the lifespan of production equipment, further contributing to long-term operational expenditure savings.
- Cost Reduction in Manufacturing: The removal of amino-protection and deprotection steps fundamentally alters the cost structure of producing chiral beta-amino alcohols. By bypassing these resource-intensive stages, manufacturers can eliminate the costs associated with additional reagents, solvents, and purification media required for intermediate isolation. This streamlined approach results in a higher overall yield per batch, effectively lowering the cost per kilogram of the active pharmaceutical ingredient. Additionally, the high enantiomeric excess achieved directly from the reactor minimizes the need for costly downstream purification processes such as chiral chromatography or repeated recrystallizations, providing a clear path to margin improvement.
- Enhanced Supply Chain Reliability: The reliance on robust, commercially available catalyst precursors and ligands ensures a stable supply of critical processing aids. Unlike bespoke enzymatic processes that may suffer from batch-to-batch variability or limited shelf-life, this chemical catalysis approach offers consistent performance and scalability. The use of standard hydrogenation infrastructure means that production can be easily shifted between different manufacturing sites without the need for specialized retrofitting. This flexibility enhances supply continuity, allowing for rapid response to market demand fluctuations and ensuring reliable delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in synthetic steps leads to a significantly lower E-factor (mass of waste per mass of product), facilitating easier compliance with increasingly stringent environmental regulations. The process generates less solvent waste and avoids the use of toxic deprotecting agents, simplifying waste treatment protocols and reducing disposal costs. The scalability of the hydrogenation reaction is well-documented, with the potential to transition seamlessly from gram-scale screening to multi-ton commercial production. This scalability ensures that the technology can support the growing global demand for CNS therapeutics without compromising on quality or sustainability metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the key advantages of this asymmetric hydrogenation method over traditional resolution?
A: Unlike traditional methods that require the synthesis of racemic mixtures followed by inefficient chiral resolution (max 50% yield), this patented process achieves direct asymmetric synthesis. It eliminates the need for amino-protecting groups and subsequent deprotection steps, significantly improving atom economy and overall yield while delivering enantiomeric excess greater than 99%.
Q: Which catalyst systems are most effective for this transformation?
A: The patent identifies cationic rhodium complexes coordinated with chiral bidentate phosphine ligands as highly effective. Specifically, ligands from the DuPhos family (e.g., (S,S)-Me-DuPhos, (S,S)-Et-DuPhos) and KetalPhos derivatives demonstrate superior stereocontrol, achieving >99% ee under mild hydrogenation conditions.
Q: Is this process scalable for industrial production of duloxetine intermediates?
A: Yes, the process utilizes standard hydrogenation equipment and commercially available solvents like methanol or ethanol. The removal of complex protection-deprotection sequences simplifies the workflow, making it highly suitable for commercial scale-up from kilogram to multi-ton production levels required for API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Amino Alcohols Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-purity chiral intermediates for the development of next-generation pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of beta-amino alcohol intermediates meets the exacting standards required by global regulatory bodies. Our expertise in asymmetric catalysis allows us to optimize these patented routes for maximum efficiency, delivering cost-effective solutions without compromising on quality or safety.
We invite you to collaborate with us to explore how this advanced hydrogenation technology can enhance your product portfolio and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and supply chain planning initiatives, ensuring a successful partnership in the competitive landscape of fine chemical manufacturing.
