Advanced Chiral Resolution Technology for Scalable Fexofenadine Hydrochloride Production
Advanced Chiral Resolution Technology for Scalable Fexofenadine Hydrochloride Production
The pharmaceutical industry continuously seeks robust methodologies for producing optically pure antihistamine intermediates, and the technical disclosure within patent CN1442407A offers a compelling solution for the synthesis of fexofenadine enantiomers. This specific intellectual property details a chemical resolution preparation method that utilizes tartaric acid derivatives to separate the left-handed (-) and right-handed (+) enantiomers of 4-[1-hydroxyl-4-(4-diphenylhydroxymethyl-1-piperidinyl)butyl]-α,α-dimethylphenylacetic acid esters. By leveraging the differential solubility of diastereomeric salts formed between the racemic intermediate and chiral resolving agents, this process bypasses the need for expensive asymmetric catalysis while delivering exceptional stereochemical purity. For R&D directors and procurement specialists, understanding the nuances of this resolution technique is critical for evaluating supply chain resilience and cost structures in the production of third-generation antihistamines. The method described provides a foundational pathway for manufacturing high-purity pharmaceutical intermediates that align with global regulatory expectations for chiral drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active fexofenadine has relied heavily on asymmetric reduction techniques, such as the borane reduction methods cited in prior art like US6194431 and Tetrahedron Letters 39 (1998). These conventional approaches often necessitate the use of hazardous reducing agents and transition metal catalysts, which introduce significant safety risks and environmental compliance burdens during large-scale manufacturing. Furthermore, the removal of trace metal impurities from the final active pharmaceutical ingredient requires additional purification steps, such as specialized filtration or scavenging resins, which drastically increase operational expenditures and extend production lead times. The complexity of managing exothermic reduction reactions also poses engineering challenges for scale-up, potentially limiting the batch sizes and throughput capabilities of production facilities. Consequently, reliance on these traditional catalytic routes can create bottlenecks in the supply chain, making it difficult to respond agilely to market demand fluctuations for essential allergy medications.
The Novel Approach
In contrast, the novel approach outlined in the patent data utilizes a classical yet highly optimized chemical resolution strategy using tartaric acid derivatives, which fundamentally shifts the risk profile and cost structure of the synthesis. By employing resolving agents such as di-p-toluoyl tartaric acid or dibenzoyl tartaric acid, the process capitalizes on the thermodynamic stability of diastereomeric salts to achieve separation without the need for reactive metal hydrides. This method allows for the use of common organic solvents like acetone, which are easier to recover and recycle compared to the specialized solvents often required for catalytic hydrogenation. The ability to recycle the mother liquor to recover the opposite enantiomer further enhances the overall atom economy and material efficiency of the process. For procurement managers, this translates into a more predictable cost model that is less susceptible to the volatility of precious metal catalyst prices, ensuring long-term economic viability for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Tartaric Acid Derivative Resolution
The core mechanism driving this synthesis involves the formation of diastereomeric salts between the racemic fexofenadine ester and the optically pure tartaric acid derivative resolving agent. When dissolved in a solvent such as acetone at elevated temperatures ranging from 40°C to 80°C, the components interact to form a homogeneous solution where molecular recognition begins to occur. Upon controlled cooling to temperatures between -10°C and 20°C, the solubility product of one specific diastereomeric salt is exceeded, leading to selective nucleation and crystal growth of the desired enantiomeric complex. This crystallization process is highly sensitive to the steric and electronic properties of the resolving agent, where substituents on the benzoyl groups influence the lattice energy and solubility differential between the salt pairs. The structural versatility of the tartaric acid backbone allows for fine-tuning of these interactions to maximize the yield and optical purity of the precipitate.
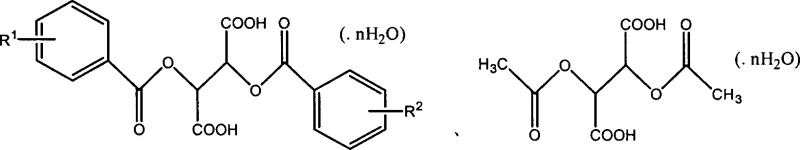
Following the initial precipitation, the crude diastereomeric salt undergoes recrystallization, a critical purification step that elevates the enantiomeric excess (e.e.) value to greater than 99.9%. This high level of purity is essential for meeting the stringent impurity profile requirements of regulatory bodies for chiral drug substances. The subsequent hydrolysis of the purified ester salt using alkali metal hydroxides, such as sodium or potassium hydroxide, cleaves the ester bond to liberate the free acid form of the fexofenadine enantiomer. Finally, acidification with hydrochloric acid converts the free base into the stable hydrochloride salt form, which is the preferred pharmaceutical dosage form due to its improved solubility and stability characteristics. This multi-step sequence ensures that the final product is not only optically pure but also chemically stable and suitable for downstream formulation into final dosage forms.
How to Synthesize Fexofenadine Efficiently
The synthesis of optically pure fexofenadine via this resolution pathway requires precise control over stoichiometry, temperature gradients, and solvent quality to ensure consistent batch-to-batch reproducibility. The process begins with the dissolution of the racemic intermediate and the resolving agent in a molar ratio ranging from 1:0.1 to 1:5, with an optimal range of 1:0.9 to 1.5 to balance yield and cost efficiency. Detailed standardized synthesis steps, including specific cooling rates and agitation parameters, are essential for controlling crystal habit and size distribution, which directly impact filtration efficiency and drying times. Operators must adhere to strict protocols during the hydrolysis and acidification stages to prevent racemization or degradation of the sensitive chiral center. For technical teams looking to implement this route, understanding these critical process parameters is key to achieving the high purity specifications required for a reliable pharmaceutical intermediates supplier.
- Dissolve racemic fexofenadine ester and tartaric acid derivative resolving agent in acetone at 40-80°C.
- Cool the solution to -10-20°C to precipitate the diastereomeric salt and purify via recrystallization.
- Perform alkaline hydrolysis followed by acidification to isolate the optically pure fexofenadine hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this tartaric acid-based resolution method offers substantial advantages in terms of cost reduction in API manufacturing and supply chain reliability. By eliminating the dependency on expensive transition metal catalysts and hazardous reducing agents, the process significantly lowers the raw material costs associated with each production batch. The use of acetone as a primary solvent further reduces waste disposal costs and simplifies solvent recovery infrastructure, contributing to a more sustainable and economically efficient operation. For supply chain heads, the robustness of crystallization-based purification ensures that the process is less sensitive to minor variations in raw material quality, thereby reducing the risk of batch failures and production delays. This stability is crucial for maintaining continuous supply lines to global pharmaceutical partners who demand consistent quality and on-time delivery.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of commodity chemicals like tartaric acid derivatives and acetone drastically simplify the bill of materials. This shift removes the need for expensive metal scavenging steps and reduces the overall environmental compliance burden associated with heavy metal waste. Consequently, the operational expenditure per kilogram of produced intermediate is significantly optimized, allowing for more competitive pricing strategies in the global market. The ability to recycle mother liquors to recover the opposite enantiomer further maximizes material utilization, ensuring that raw material costs are kept to a minimum while maintaining high overall yields.
- Enhanced Supply Chain Reliability: The reliance on widely available chemical reagents rather than specialized catalytic systems mitigates the risk of supply disruptions caused by geopolitical or market volatility. Tartaric acid derivatives are produced at a large industrial scale for various applications, ensuring a stable and diverse supplier base for key raw materials. This availability translates into reduced lead time for high-purity pharmaceutical intermediates, as procurement teams can source materials quickly without long qualification periods. Furthermore, the simplicity of the equipment required for crystallization and filtration means that production can be easily scaled or transferred between facilities without significant capital investment.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of standard unit operations such as dissolution, crystallization, and filtration, which are well-understood in chemical engineering. The avoidance of hazardous reagents like borane simplifies safety management and reduces the regulatory hurdles associated with handling dangerous goods. This alignment with green chemistry principles enhances the environmental profile of the manufacturing site, making it easier to obtain necessary permits and maintain compliance with increasingly strict environmental regulations. The reduced generation of hazardous waste also lowers disposal costs and improves the overall sustainability metrics of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology for fexofenadine production. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of this approach for industry stakeholders. Understanding these details helps decision-makers evaluate the potential for integrating this method into their existing manufacturing portfolios. The answers reflect the technical realities of chiral resolution and the practical benefits observed in similar industrial applications.
Q: What is the optical purity achievable with this resolution method?
A: The described chemical resolution process utilizing tartaric acid derivatives consistently achieves an enantiomeric excess (e.e.) value greater than 99.9% after recrystallization, meeting stringent pharmaceutical standards for chiral intermediates.
Q: Why is tartaric acid derivative preferred over borane reduction?
A: Tartaric acid derivatives offer a significantly safer and more cost-effective alternative to borane asymmetric reduction, eliminating the need for hazardous reagents and complex metal removal steps while maintaining high stereochemical control.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the reliance on standard crystallization techniques and common solvents like acetone makes this method highly scalable, ensuring robust supply chain continuity and simplified industrial engineering compared to catalytic hydrogenation routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fexofenadine Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chiral resolutions like the one described are executed with precision and efficiency. Our technical team is well-versed in optimizing crystallization parameters to meet stringent purity specifications, utilizing rigorous QC labs to verify enantiomeric excess and impurity profiles at every stage. We understand the critical nature of supply continuity for antihistamine intermediates and have established robust protocols to mitigate risks associated with raw material sourcing and process variability. Our commitment to quality and reliability makes us a trusted partner for pharmaceutical companies seeking to secure their supply chains for high-value chiral intermediates.
We invite you to engage with our technical procurement team to discuss how this resolution technology can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this catalyst-free route. We encourage potential partners to contact us for specific COA data and route feasibility assessments to validate the compatibility of this method with your current manufacturing infrastructure. Our goal is to provide transparent, data-driven insights that empower you to make informed decisions about your API supply strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
