Advanced Manufacturing of High-Purity Levetiracetam via Optimized Esterification and Ammonolysis
Advanced Manufacturing of High-Purity Levetiracetam via Optimized Esterification and Ammonolysis
The pharmaceutical industry continuously seeks robust synthetic routes for critical antiepileptic agents, and the methodology detailed in patent CN107337628B represents a significant leap forward in the production of Levetiracetam. This second-generation acetylcholine agonist, structurally related to piracetam, is a vital pyrrolidone derivative used globally for treating local and secondary generalized epilepsy. The core innovation lies in a refined post-dissociation treatment of the key intermediate, (S)-alpha-ethyl-2-oxo-1-pyrrolidineacetic acid (R)-alpha-methylbenzylamine salt. Unlike conventional methods that rely on harsh acidic conditions, this novel approach adjusts the pH of the extracted water layer to a mild range of 5-9 before proceeding to esterification. This subtle yet powerful modification not only streamlines the operational workflow but also dramatically enhances the final product quality, achieving purities of 99.8% to 99.9% with a single recrystallization step. For a reliable pharmaceutical intermediates supplier, mastering such nuanced pH control is essential for delivering consistent, high-quality batches to downstream API manufacturers.
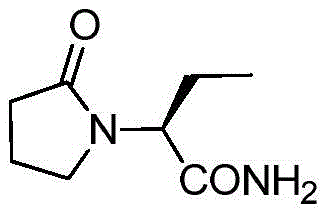
Levetiracetam is influenced heavily by reaction conditions during its synthesis, where impurities like levetiracetam acid or ethyl esters can easily form if parameters are not tightly controlled. The structural integrity of the molecule, as depicted above, requires careful handling to maintain stereochemical purity and avoid ring opening. The patent data highlights that by avoiding strong acidification and utilizing a specific sequence of base dissociation followed by neutralization, the process mitigates these risks effectively. This technical advancement positions the manufacturing process as a superior choice for cost reduction in API manufacturing, as it reduces the burden on purification units and minimizes the loss of valuable chiral material. Furthermore, the ability to obtain such high purity directly from the crude reaction mixture underscores the efficiency of this route, making it an attractive option for supply chain managers looking to optimize their procurement strategies for high-purity Levetiracetam.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Levetiracetam has relied on protocols such as those described in US4696943 and CN10133180A, which involve dissociating the chiral salt and subsequently acidifying the mixture with hydrochloric acid to isolate the free acid. While effective to a degree, these traditional pathways suffer from several inherent drawbacks that impact both economic and environmental metrics. The requirement for strong acidification often necessitates the use of substantial amounts of organic bases, such as triethylamine, in the subsequent esterification step to scavenge the generated acid. This reliance on stoichiometric amounts of amines not only increases the raw material cost but also generates significant quantities of amine salts as waste, complicating the wastewater treatment process. Moreover, the harsh acidic environment can sometimes promote degradation pathways or racemization, leading to lower overall yields and requiring more intensive purification efforts to meet the stringent purity standards required for neurological drugs. These factors collectively contribute to a higher cost of goods sold and a larger environmental footprint, which are critical concerns for modern commercial scale-up of complex pyrrolidone derivatives.
The Novel Approach
In stark contrast, the method disclosed in CN107337628B introduces a paradigm shift by replacing the strong acidification step with a precise pH adjustment to a range of 5-9, preferably between 6 and 8. This mild conditioning allows for the direct conversion of the intermediate without the need for isolating the free acid under harsh conditions. By treating the (R)-alpha-methylbenzylamine salt with an inorganic base like sodium hydroxide and extracting the free amine with toluene, the process creates a clean aqueous environment for the subsequent reaction. The elimination or drastic reduction of triethylamine usage during the esterification with ethyl chloroformate is a major breakthrough, as it simplifies the workup procedure to a simple filtration of inorganic salts. This streamlined approach not only improves the yield, with examples showing recovery rates between 75% and 85%, but also significantly reduces the discharge of "three wastes" (wastewater, waste gas, and solid residue). For procurement teams, this translates to a more sustainable and economically viable supply source for reducing lead time for high-purity antiepileptic intermediates.
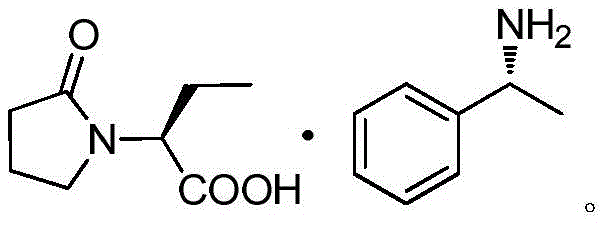
Mechanistic Insights into pH-Controlled Mixed Anhydride Activation
The success of this synthesis hinges on the delicate balance of protonation states within the reaction medium, specifically during the transition from the chiral salt to the activated ester. In the conventional acidic route, the carboxylic acid is fully protonated, requiring a strong base to deprotonate it for nucleophilic attack by the chloroformate. However, in this novel process, adjusting the pH to 5-9 ensures that the carboxylate species is present in a state that is reactive enough for activation but stable enough to prevent hydrolysis or side reactions. When ethyl chloroformate is added to the solution in dichloromethane, it reacts with the carboxylate to form a mixed anhydride intermediate. This mixed anhydride is highly electrophilic and primed for the subsequent ammonolysis step. The presence of residual water is strictly controlled to less than 1%, preferably below 0.5%, through azeotropic dehydration with toluene, which is critical because water would compete with the chloroformate, leading to hydrolysis and reduced yield. This mechanistic precision ensures that the activation step proceeds with high efficiency, setting the stage for a clean conversion to the final amide.
Furthermore, the control of impurities is intrinsically linked to this pH management strategy. By avoiding strongly acidic conditions, the risk of acid-catalyzed ring opening of the pyrrolidone moiety is minimized, preserving the structural integrity of the core scaffold. The extraction of the free (R)-alpha-methylbenzylamine with toluene prior to acidification ensures that the chiral resolving agent is removed cleanly, preventing it from interfering with the esterification or contaminating the final product. The subsequent ammonolysis reaction, whether using liquid ammonia or an ammonia-dichloromethane solution, proceeds smoothly on the activated mixed anhydride to form the primary amide bond of Levetiracetam. The inorganic salts generated, primarily sodium chloride, are easily removed by filtration, leaving a solution that, upon solvent removal and recrystallization from acetone, yields the target molecule with exceptional optical and chemical purity. This level of control is what distinguishes a laboratory curiosity from a robust industrial process capable of meeting global regulatory standards.
How to Synthesize Levetiracetam Efficiently
The synthesis of Levetiracetam via this optimized route involves a sequence of unit operations designed for maximum efficiency and minimal waste generation. The process begins with the dissociation of the chiral salt using an aqueous inorganic base, followed by the critical pH adjustment and dehydration steps that define the novelty of this patent. Once the reaction medium is prepared with low water content, the activation with chloroformate and subsequent amidation proceed rapidly. The detailed standardized synthesis steps, including specific reagent ratios, temperature controls, and workup procedures, are outlined in the guide below to assist R&D teams in replicating this high-yielding process.
- Dissociate the (R)-alpha-methylbenzylamine salt using an inorganic base like sodium hydroxide, followed by organic solvent extraction to remove free amine.
- Adjust the pH of the aqueous layer to a neutral range of 5-9, perform azeotropic dehydration, and dissolve the resulting oil in dichloromethane.
- Activate the acid with ethyl chloroformate to form a mixed anhydride, then react with ammonia to complete the ammonolysis and crystallize the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the simplification of the production workflow, which directly correlates to reduced operational expenditures and enhanced throughput. By eliminating the need for extensive acid-base workups and minimizing the use of expensive organic bases like triethylamine, the overall cost of manufacturing is significantly lowered. This cost efficiency is compounded by the reduction in waste disposal costs, as the process generates fewer saline byproducts and organic residues. Additionally, the use of common, readily available solvents such as toluene and dichloromethane ensures that the supply chain remains resilient against raw material shortages. The ability to achieve high purity (greater than 99.8%) with a single recrystallization step further reduces the time and resources spent on downstream purification, allowing for faster batch turnover and improved responsiveness to market demand.
- Cost Reduction in Manufacturing: The most significant economic driver in this process is the drastic reduction or complete avoidance of triethylamine during the esterification phase. In traditional methods, triethylamine is required in stoichiometric amounts to neutralize the HCl generated, creating a large volume of triethylamine hydrochloride salt that must be filtered and disposed of. By operating at a controlled pH of 5-9, the new method bypasses the need for this expensive scavenger, thereby lowering raw material costs and waste treatment fees. Furthermore, the simplified workup reduces the consumption of processing time and energy, contributing to a leaner manufacturing profile that enhances overall profit margins for the production of this critical API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to a more reliable supply chain by minimizing the number of critical process parameters that can cause batch failures. The use of standard industrial reagents like sodium hydroxide and hydrochloric acid for pH adjustment ensures that the process is not dependent on specialized or hard-to-source chemicals. The azeotropic dehydration step using toluene is a well-understood unit operation in the fine chemical industry, reducing the risk of scale-up issues. This reliability means that suppliers can maintain consistent delivery schedules, reducing the risk of stockouts for downstream pharmaceutical manufacturers who depend on a steady flow of high-quality Levetiracetam to meet global prescription demands.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles. The reduction in "three wastes" (wastewater, waste gas, and solid waste) simplifies compliance with increasingly stringent environmental regulations. The process generates primarily inorganic salts and recoverable organic solvents, which are easier to treat and recycle compared to the complex organic waste streams associated with amine-heavy processes. This environmental friendliness facilitates smoother permitting for plant expansions and ensures long-term operational sustainability. The straightforward nature of the reaction steps—extraction, distillation, filtration, and crystallization—makes the process highly amenable to scaling from pilot plant quantities to multi-ton commercial production without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Levetiracetam synthesis method. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing operations or for procurement specialists assessing supplier capabilities.
Q: How does the new pH adjustment method improve yield compared to traditional acidification?
A: By adjusting the pH to a mild range of 5-9 instead of strong acidification, the process minimizes side reactions and decomposition of the sensitive pyrrolidone ring, leading to significantly higher crude yields and purity levels exceeding 99.8%.
Q: What are the primary cost drivers eliminated in this synthesis route?
A: The optimized protocol drastically reduces or eliminates the need for triethylamine during the esterification step, which lowers raw material costs and simplifies the downstream purification process by reducing salt waste.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of common industrial solvents like toluene and dichloromethane, combined with standard unit operations like azeotropic distillation and filtration, makes this method highly scalable for multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levetiracetam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes for high-value pharmaceutical intermediates like Levetiracetam. Our technical team has extensively analyzed the advancements presented in patent CN107337628B and integrated similar process optimizations into our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel chemistry can be realized at an industrial scale. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Levetiracetam we supply meets the highest international standards for safety and efficacy. We are committed to being a partner that not only supplies materials but also drives value through continuous process improvement.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage these technological advantages. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you optimize your supply chain for Levetiracetam, ensuring a reliable, cost-effective, and high-quality source for your antiepileptic drug formulations.
