Advanced Synthesis of Perindopril Intermediates: Enhancing Purity and Commercial Scalability
Advanced Synthesis of Perindopril Intermediates: Enhancing Purity and Commercial Scalability
The pharmaceutical industry continuously seeks robust manufacturing pathways for cardiovascular therapeutics, particularly for Angiotensin-Converting Enzyme (ACE) inhibitors like perindopril. Patent CN1331883C discloses a groundbreaking methodology for the synthesis of (2S,3aS,7aS)-1-((S)-alanyl)-octahydro-1H-indole-2-carboxylic acid derivatives, which serve as the critical backbone for perindopril production. This innovation addresses long-standing challenges in stereochemical control and raw material availability. The core target of this synthesis is the compound of Formula (I), a highly functionalized bicyclic amino acid derivative that dictates the biological efficacy of the final drug substance.
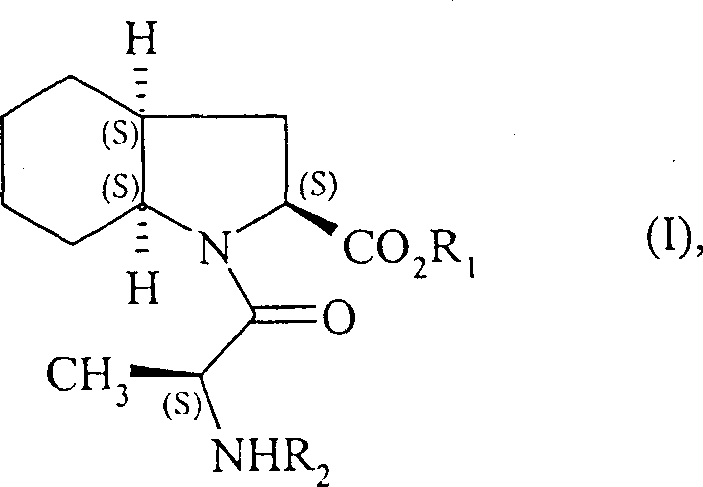
Perindopril and its pharmaceutically acceptable salts, such as the tert-butylamine salt, possess valuable pharmacological properties primarily driven by their inhibitory effect on angiotensin I converting enzyme. This inhibition prevents the conversion of angiotensin I to the vasoconstrictor angiotensin II and simultaneously prevents the degradation of the vasodilator bradykinin. Given the immense therapeutic value of this compound in treating hypertension and heart failure, establishing an efficient industrial synthesis process that guarantees excellent purity and high yield is of utmost importance for global supply chains. The disclosed method represents a significant leap forward in process chemistry for this vital medication class.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in patent application EP 1256590, have historically relied on (2S)-indoline-2-carboxylic acid esters as the primary starting materials. A critical bottleneck in this conventional approach is that these specific starting materials are not commercially available off-the-shelf. Consequently, manufacturers were forced to undertake a multi-step synthesis sequence starting from indole-2-carboxylic acid. This legacy route invariably includes a resolution step to isolate the desired enantiomer, which theoretically limits the maximum yield to 50% unless dynamic kinetic resolution is employed, thereby inflating production costs and generating significant chemical waste. Furthermore, the reliance on non-natural or difficult-to-source chiral pools introduces supply chain volatility and complicates quality control protocols regarding impurity profiles.
The Novel Approach
In stark contrast, the process detailed in CN1331883C revolutionizes the synthetic strategy by utilizing alanine, a natural and therefore inexpensive amino acid, as the sole chiral source for the side chain, alongside compounds readily obtainable from serine. This shift to the "chiral pool" strategy fundamentally alters the economic landscape of production. By starting with abundant, naturally occurring building blocks, the new method bypasses the need for expensive resolution steps associated with synthetic racemates. The reaction sequence involves the condensation of 1-(1-cyclohexen-1-yl)-pyrrolidine with serine derivatives, followed by a clever cyclization and dehydration sequence to construct the octahydroindole core. This approach not only simplifies the operational complexity but also ensures that the stereochemistry is embedded early in the synthesis, leading to superior control over the final product's optical purity.
Mechanistic Insights into the Cyclization and Hydrogenation Strategy
The mechanistic elegance of this synthesis lies in the construction of the bicyclic indole system. The process initiates with the reaction of 1-(1-cyclohexen-1-yl)-pyrrolidine (Formula III) with serine compounds of Formula (IV). This condensation yields an intermediate which, upon deprotection of the amine functionality and subsequent acid-catalyzed cyclization followed by dehydration, affords the unsaturated hexahydro-1H-indole derivative of Formula (VI). This step is crucial as it establishes the rigid bicyclic framework required for the biological activity of perindopril. The use of p-toluenesulfonic acid in toluene with azeotropic removal of water drives this equilibrium towards the cyclized product with exceptional efficiency, as evidenced by yields reaching up to 97% in experimental examples.
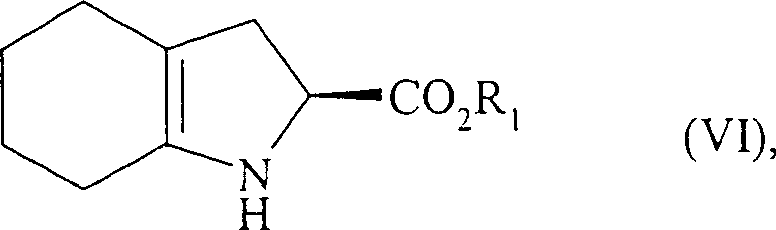
Following the formation of the core scaffold, the synthesis proceeds with the coupling of the indole intermediate with an alanine compound of Formula (VII). This peptide coupling reaction is meticulously optimized using dicyclohexylcarbodiimide (DCC) and triethylamine in solvents like tetrahydrofuran or ethyl acetate, often with 1-hydroxybenzotriazole (HOBt) to suppress racemization. The final and perhaps most critical stereochemical defining step is the catalytic hydrogenation. The unsaturated intermediate is hydrogenated in the presence of noble metal catalysts such as palladium, platinum, rhodium, or nickel under hydrogen pressures ranging from 1 to 30 bar. This step reduces the double bond and sets the cis-fused ring junction stereochemistry (3aS, 7aS) with high diastereoselectivity. Experimental data indicates that this hydrogenation step can achieve 99% enantiomeric purity, demonstrating the robustness of the catalytic system in controlling the three-dimensional architecture of the molecule.
How to Synthesize (2S,3aS,7aS)-1-((S)-Alanyl)-octahydro-1H-indole-2-carboxylic Acid Efficiently
The synthesis of this high-value intermediate requires precise control over reaction conditions to maintain the integrity of the chiral centers. The process begins with the alkylation of the enamine followed by hydrolysis to introduce the cyclohexanone moiety, which is then cyclized to form the indole nucleus. Subsequent coupling with protected alanine and final hydrogenation completes the sequence. For R&D teams looking to replicate or scale this process, attention to the stoichiometry of the coupling agents and the pressure parameters during hydrogenation is paramount. The detailed standardized synthesis steps, including specific reagent grades and workup procedures, are outlined in the guide below.
- Condense 1-(1-cyclohexen-1-yl)-pyrrolidine with a protected serine derivative to form the initial open-chain precursor.
- Perform deprotection and acid-catalyzed cyclization followed by dehydration to generate the hexahydro-1H-indole core structure.
- Couple the indole intermediate with protected alanine using DCC activation, followed by catalytic hydrogenation to establish the final stereochemistry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to the synthesis method described in CN1331883C offers profound strategic benefits beyond mere technical feasibility. The shift away from obscure, multi-step starting materials towards commodity amino acids fundamentally de-risks the supply chain. By anchoring the synthesis in the "chiral pool" of serine and alanine, manufacturers can leverage the massive global production capacity of the fermentation industry, ensuring consistent availability and price stability for raw materials. This decoupling from specialized, low-volume chemical suppliers mitigates the risk of production stoppages due to raw material shortages, a critical factor for maintaining continuous API supply for chronic medications like antihypertensives.
- Cost Reduction in Manufacturing: The elimination of the resolution step found in prior art processes results in a dramatic improvement in overall process mass intensity. In traditional routes, discarding the unwanted enantiomer during resolution effectively halves the theoretical yield of the starting material, a cost that is passed down through the entire value chain. By using naturally occurring L-amino acids, this waste is entirely eradicated. Furthermore, the use of standard coupling reagents and heterogeneous hydrogenation catalysts allows for the use of established, cost-effective unit operations rather than specialized chromatographic separations. The high yields reported in the patent examples, such as 97% for the cyclization step and 87% for the final hydrogenation, translate directly into lower cost of goods sold (COGS) through improved material throughput.
- Enhanced Supply Chain Reliability: The reliance on 1-(1-cyclohexen-1-yl)-pyrrolidine and protected serine derivatives creates a more resilient supply network. These precursors are chemically stable and can be sourced from multiple vendors globally, unlike the specific indoline esters required by older methods which may have single-source bottlenecks. Additionally, the robustness of the hydrogenation step, which tolerates a range of pressures and catalysts (Pd, Pt, Rh, Ni), provides operational flexibility. If one catalyst type becomes expensive or scarce, the process can be adapted to another without redesigning the entire synthetic route, ensuring uninterrupted production schedules and reliable delivery timelines for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is significantly greener. The avoidance of resolution steps reduces solvent consumption and waste generation associated with separating enantiomers. The primary byproducts of the coupling and cyclization steps are manageable, and the use of catalytic hydrogenation is inherently cleaner than stoichiometric reductions involving metal hydrides. The high chemical purity (98%) and enantiomeric purity (99%) achieved directly from the reactor reduce the need for extensive purification trains, such as repeated recrystallizations or preparative HPLC, which are resource-intensive. This streamlined downstream processing facilitates easier scale-up from pilot plant to multi-ton commercial production while meeting increasingly stringent regulatory standards for impurity control.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and claims within patent CN1331883C, providing a clear picture of the technology's capabilities and limitations for potential licensees or manufacturing partners.
Q: What are the primary advantages of the synthesis method described in CN1331883C compared to prior art?
A: The method utilizes naturally occurring and inexpensive amino acids (alanine and serine) as the sole chiral sources, eliminating the need for complex resolution steps required by previous methods that started from indole-2-carboxylic acid.
Q: How does this process ensure high enantiomeric purity for the final perindopril intermediate?
A: By relying on the inherent chirality of L-serine and L-alanine derivatives and utilizing stereoselective catalytic hydrogenation, the process achieves up to 99% enantiomeric purity without requiring chiral chromatography.
Q: Is the starting material 1-(1-cyclohexen-1-yl)-pyrrolidine commercially viable for large-scale production?
A: Yes, the starting materials are readily obtainable and significantly more accessible than the (2S)-indoline-2-carboxylic acid esters used in older processes, ensuring better supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perindopril Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the one described in CN1331883C to deliver high-quality pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand that for cardiovascular APIs, consistency is key; therefore, our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of (2S,3aS,7aS)-1-((S)-alanyl)-octahydro-1H-indole-2-carboxylic acid derivatives meets the exacting standards required for global regulatory filings.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for perindopril production. By leveraging our expertise in asymmetric synthesis and catalytic hydrogenation, we can help you realize the full cost-saving potential of this novel route. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, specific COA data from our recent batches, and comprehensive route feasibility assessments tailored to your production volume requirements.
