Advanced One-Pot Synthesis of Beta-Methyl Carbapenem Antibiotic Parent Nucleus for Commercial Scale
Advanced One-Pot Synthesis of Beta-Methyl Carbapenem Antibiotic Parent Nucleus for Commercial Scale
The pharmaceutical industry continuously seeks robust manufacturing routes for critical antibiotic scaffolds, particularly for the highly effective carbapenem class. Patent CN101891766A introduces a transformative preparation method for the beta-methyl carbapenem antibiotic parent nucleus, addressing long-standing inefficiencies in catalytic cyclization and downstream processing. This technology leverages a synergistic combination of noble metal catalysts and specific promoters to drive the formation of the bicyclic beta-lactam core with exceptional efficiency. By eliminating the need for intermediate isolation and significantly lowering catalyst loading, this approach offers a compelling value proposition for large-scale API production. The method ensures complete raw material conversion while minimizing impurity profiles, which is critical for meeting stringent regulatory standards in global markets. Furthermore, the operational simplicity of this one-pot strategy translates directly into reduced manufacturing cycles and enhanced throughput capabilities for contract development and manufacturing organizations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating the beta-methyl carbapenem core often suffer from cumbersome multi-step sequences that severely impact overall process economics and environmental sustainability. In conventional protocols, the initial cyclization step typically requires rigorous purification, such as crystallization or chromatography, to isolate the intermediate before it can undergo subsequent phosphorylation or functionalization. This necessity for intermediate handling not only consumes significant time and equipment capacity but also inevitably leads to material losses at each transfer stage, dragging down the cumulative yield. Moreover, older methods frequently rely on high loadings of expensive rhodium catalysts without auxiliary agents to boost turnover numbers, resulting in prohibitive raw material costs. The accumulation of waste streams, including solvent residues and heavy metal contaminants from excessive catalyst use, creates substantial burdens for wastewater treatment and compliance with green chemistry initiatives. These operational bottlenecks make traditional processes ill-suited for the high-volume demands of modern generic antibiotic manufacturing.
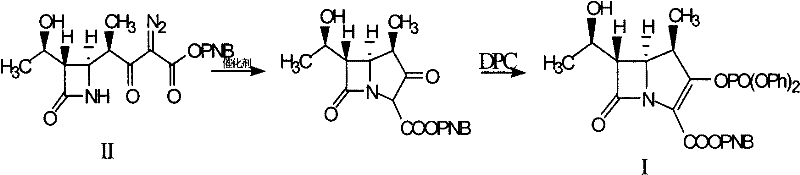
The Novel Approach
In stark contrast to legacy techniques, the novel methodology described in the patent utilizes a promoter-assisted catalytic system that enables a seamless telescoped reaction sequence. By introducing specific co-catalysts or promoters alongside the primary noble metal catalyst, the reaction kinetics are optimized to proceed rapidly and selectively without the need to isolate the unstable intermediate species. This innovation allows the reaction mixture containing the cyclized intermediate to be directly treated with phosphorylating agents in the same vessel, effectively collapsing two distinct unit operations into a single continuous process. The strategic use of promoters not only accelerates the reaction rate but also permits a drastic reduction in the quantity of precious rhodium required, thereby lowering the direct material cost profile. Additionally, the improved selectivity minimizes the formation of side products, simplifying the final workup and crystallization steps to achieve high-purity targets with minimal effort. This streamlined workflow represents a paradigm shift in how complex beta-lactam intermediates are produced, offering a scalable solution that aligns with modern lean manufacturing principles.
Mechanistic Insights into Promoter-Assisted Rhodium Catalysis
The core of this technological advancement lies in the sophisticated interplay between the rhodium catalyst and the added promoter species within the reaction matrix. The noble metal catalyst, such as rhodium acetate or rhodium caprylate, initiates the decomposition of the diazo precursor to form a reactive metal-carbene species, which subsequently undergoes intramolecular C-H insertion to construct the carbapenem ring system. The introduction of promoters like DMAP, copper bromide, or zinc salts acts to stabilize the transition states or modulate the electronic environment of the rhodium center, enhancing its catalytic activity and longevity. This synergistic effect ensures that the cyclization proceeds to near-completion even at lower catalyst concentrations, preventing the accumulation of unreacted starting materials that could complicate downstream purification. The mechanistic efficiency is further supported by the careful control of reaction temperatures, typically ranging from 30 to 95 degrees Celsius during the cyclization phase, which balances reaction rate with thermal stability of the sensitive beta-lactam structure.
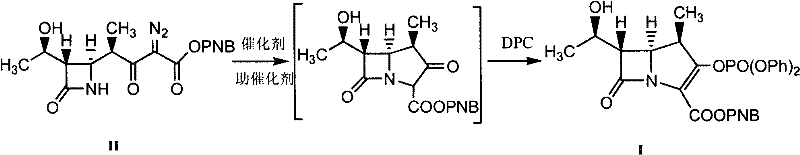
Impurity control is rigorously managed through the optimization of the promoter-to-catalyst ratio and the selection of compatible organic solvents such as dichloromethane or acetonitrile. The presence of the promoter helps suppress competing side reactions, such as dimerization of the diazo compound or non-selective insertion pathways, which are common sources of difficult-to-remove impurities in carbapenem synthesis. Following the cyclization, the addition of diphenyl phosphate chloride (DPC) and an organic base facilitates the elimination reaction to form the double bond in the five-membered ring, completing the parent nucleus structure. The protocol specifies washing steps with acidic aqueous solutions and phosphate buffers to effectively remove residual metal salts and basic byproducts, ensuring the final organic phase is clean prior to crystallization. This meticulous attention to chemical detail throughout the mechanism guarantees a product profile that meets the rigorous specifications required for subsequent coupling with various side chains to produce finished antibiotics like Meropenem or Ertapenem.
How to Synthesize Beta-Methyl Carbapenem Efficiently
The execution of this synthesis requires precise adherence to the defined operational parameters to maximize yield and safety while maintaining reproducibility across batches. The process begins under an inert atmosphere, typically nitrogen or argon, to protect the sensitive diazo functionality and the active catalyst species from moisture and oxygen degradation. Operators must carefully monitor the reaction progress using analytical tools like HPLC or TLC to determine the exact endpoint of the cyclization before proceeding to the phosphorylation stage. The subsequent addition of reagents must be performed at controlled low temperatures to manage exotherms and prevent decomposition of the newly formed bicyclic intermediate. For a comprehensive understanding of the specific reagent quantities, solvent volumes, and temperature ramps required for successful implementation, please refer to the detailed standardized synthesis guide provided below.
- React Compound II with a noble metal catalyst and a promoter in an organic solvent under inert gas protection at 30-95°C.
- Upon completion, cool the mixture, add an organic base and diphenyl phosphate chloride (DPC), and react at -50 to 35°C.
- Wash the reaction mixture with acidic aqueous solution and phosphate buffer, then crystallize the product from organic solvent.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound advantages that directly address the cost and reliability concerns of procurement managers and supply chain directors. The ability to telescope multiple reaction steps into a single vessel eliminates the need for intermediate isolation equipment, reducing capital expenditure on hardware and freeing up reactor capacity for other campaigns. By drastically cutting the consumption of high-cost noble metal catalysts through promoter enhancement, the direct material cost of goods sold is significantly lowered, improving margin potential for high-volume contracts. The simplified workflow also reduces the labor hours associated with filtration, drying, and charging of intermediates, leading to substantial operational expense savings over the lifecycle of the product. Furthermore, the robust nature of the reaction conditions ensures consistent batch-to-batch quality, minimizing the risk of costly reworks or batch failures that can disrupt supply continuity.
- Cost Reduction in Manufacturing: The elimination of intermediate purification steps removes entire unit operations such as filtration and drying, which translates to significant savings in energy consumption and solvent usage. Reducing the loading of expensive rhodium catalysts by leveraging promoter synergy directly lowers the bill of materials without compromising reaction performance. The higher overall yield achieved through this optimized route means more product is generated per kilogram of starting material, effectively spreading fixed costs over a larger output volume. These combined factors result in a markedly more economical manufacturing process that enhances competitiveness in the global generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The simplified process flow reduces the number of potential failure points and operational delays, ensuring more predictable lead times for order fulfillment. Since the method does not rely on complex isolation procedures that are prone to variability, the manufacturing timeline is shortened, allowing for faster turnaround from raw material intake to finished intermediate. The use of common, commercially available solvents and reagents mitigates the risk of supply shortages for specialized chemicals, securing the continuity of production schedules. This reliability is crucial for maintaining steady inventory levels and meeting the just-in-time delivery requirements of major pharmaceutical clients.
- Scalability and Environmental Compliance: The one-pot nature of the reaction facilitates straightforward scale-up from pilot plant to commercial tonnage without the need for extensive process re-engineering. Minimizing the generation of waste streams, particularly those containing heavy metals and organic residues, simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. The process aligns with green chemistry principles by maximizing atom economy and reducing solvent intensity, which helps facilities maintain compliance with increasingly strict environmental regulations. This scalability ensures that the technology can support growing market demand for carbapenem antibiotics without encountering technical bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis route. Understanding these details helps stakeholders evaluate the feasibility of adopting this technology for their specific supply chain needs. The answers are derived directly from the experimental data and process descriptions outlined in the patent documentation to ensure accuracy and relevance.
Q: How does the promoter affect the rhodium catalyst usage?
A: The addition of promoters such as DMAP or copper salts significantly reduces the required loading of expensive noble metal catalysts while improving reaction conversion.
Q: Is intermediate purification required in this new process?
A: No, the process allows for a telescoped reaction where the intermediate solution proceeds directly to the next step without isolation, separation, or purification.
Q: What represents the main yield improvement in this method?
A: Experimental data indicates that the optimized promoter system can improve the final product yield by approximately 22 percentage points compared to conventional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Methyl Carbapenem Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value antibiotic intermediates like the beta-methyl carbapenem parent nucleus. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial processes. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. By partnering with us, clients gain access to a supply chain that prioritizes both technical excellence and operational reliability, safeguarding their downstream API manufacturing schedules against disruptions.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis method can be integrated into your specific production requirements. Request a Customized Cost-Saving Analysis today to quantify the potential economic benefits of switching to this promoter-assisted catalytic route. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless transition to a more efficient and cost-effective supply model.
