Revolutionizing Telmisartan Production: A Novel Nucleophilic Substitution Strategy for Commercial Scale
Introduction to Advanced Telmisartan Manufacturing
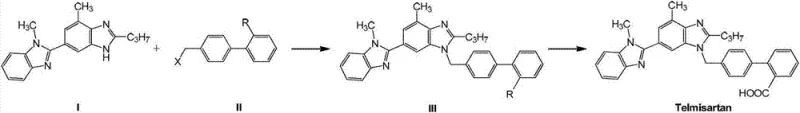
The global demand for antihypertensive medications continues to surge, placing immense pressure on supply chains to deliver high-quality Active Pharmaceutical Ingredients (APIs) like Telmisartan with consistent purity and competitive pricing. A pivotal advancement in this domain is detailed in patent CN103787982A, which introduces a refined synthetic pathway that fundamentally alters the traditional approach to constructing the biphenyl-benzimidazole scaffold. This technology leverages a direct nucleophilic substitution strategy between a specialized benzimidazole precursor and a halomethyl biphenyl derivative, bypassing several cumbersome protection and deprotection steps inherent in legacy methods. By optimizing reaction parameters, specifically temperature and base selection, this method achieves a superior impurity profile while enhancing overall process robustness. For industry stakeholders, this represents a significant leap forward in process chemistry, offering a viable route for commercial scale-up of complex pharmaceutical intermediates that meets stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Telmisartan has relied heavily on multi-step sequences involving the use of tert-butyl esters or nitrile groups as protecting functionalities to mask the carboxylic acid during the alkylation phase. These traditional routes often necessitate the use of highly reactive bromomethyl biphenyl derivatives, which, while effective at driving the reaction forward, frequently lead to the formation of undesirable quaternary ammonium salt impurities due to over-alkylation on the benzimidazole nitrogen atoms. Furthermore, the subsequent hydrolysis steps required to reveal the free carboxylic acid often involve harsh acidic or basic conditions that can degrade the sensitive molecular architecture, leading to lower yields and complex purification challenges. The reliance on bromine-based reagents also introduces significant environmental and safety concerns, including higher toxicity and corrosive potential, which complicates waste management and increases the operational burden on manufacturing facilities seeking cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the innovative methodology outlined in the patent data employs a streamlined nucleophilic substitution reaction that can proceed directly to the final acid form or utilize a more stable chloromethyl intermediate that minimizes side reactions. This approach allows for the direct coupling of 2-n-propyl-4-methyl-6-(1-methyl benzimidazole-2-yl) benzimidazole with 4'-halomethyl diphenyl-2-substituted compounds under carefully controlled thermal conditions. The ability to use chlorides instead of bromides not only mitigates the risk of disubstitution impurities but also offers a safer, more atom-economical alternative that aligns with green chemistry principles. Crucially, this pathway supports a "one-pot" synthesis variation where the hydrolysis of ester or nitrile intermediates occurs in situ, eliminating the need for intermediate isolation and significantly reducing solvent consumption and processing time. This strategic simplification positions the technology as a highly attractive option for a reliable pharmaceutical intermediate supplier aiming to optimize production throughput.
Mechanistic Insights into Nucleophilic Substitution and Impurity Control
The core of this synthetic breakthrough lies in the precise manipulation of the nucleophilic attack mechanism, where the nitrogen atom of the benzimidazole ring acts as the nucleophile against the benzylic carbon of the halomethyl biphenyl electrophile. The selection of the base is paramount; strong bases such as metal hydrides (e.g., NaH) or alkoxides (e.g., potassium tert-butoxide) are employed to deprotonate the benzimidazole nitrogen, thereby enhancing its nucleophilicity without inducing unwanted degradation of the molecular framework. The reaction kinetics are highly sensitive to thermal energy, and the patent data explicitly highlights that maintaining the reaction temperature within a narrow window of -30°C to 30°C is critical for maximizing selectivity. Deviations above this range, particularly beyond 50°C, provide sufficient activation energy for competing pathways that generate structural isomers (Impurity 1) and esterification byproducts (Impurity 2), which are notoriously difficult to separate from the final API.
Furthermore, the mechanistic understanding of impurity formation reveals that lower temperatures effectively suppress the thermodynamic drive towards these side products, ensuring that the kinetic product—the desired Telmisartan molecule—is formed with high fidelity. When the substituent R is a carboxylic acid, the direct reaction avoids the steric and electronic complications associated with bulky protecting groups, leading to a cleaner reaction profile. In scenarios where R is an ester or nitrile, the subsequent hydrolysis step is facilitated by the specific electronic environment created during the initial alkylation, allowing for efficient conversion under mild aqueous conditions. This deep control over the reaction coordinate ensures that the final high-purity Telmisartan meets rigorous pharmacopeial specifications with minimal downstream processing, validating the scientific rigor behind the proposed manufacturing protocol.
How to Synthesize Telmisartan Efficiently
Implementing this advanced synthesis route requires a disciplined approach to reaction engineering, beginning with the preparation of the halomethyl biphenyl intermediate using safe and scalable halogenating agents. The process demands strict adherence to the specified temperature gradients during the coupling phase to prevent the exothermic nature of the nucleophilic substitution from compromising product quality. Operators must utilize high-efficiency mixing and cooling systems to maintain the optimal -30°C to 30°C range, ensuring uniform heat dissipation throughout the reaction mass. Following the coupling, if a protected intermediate is used, the one-pot hydrolysis can be triggered by the addition of protic solvents and appropriate pH adjusters, streamlining the workflow significantly. For a comprehensive breakdown of the standardized operating procedures and safety protocols required for this synthesis, please refer to the detailed guide below.
- Prepare the key intermediate (Compound II) by halogenating 2'-substituted methyl biphenyl compounds using chlorinating or brominating agents under controlled conditions.
- Conduct the nucleophilic substitution reaction between Compound I and Compound II in the presence of a strong base such as sodium hydride or potassium alkoxide.
- Maintain the reaction temperature strictly between -30°C and 30°C to minimize isomerization and esterification impurities, followed by hydrolysis if a protected ester intermediate was used.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis method offers transformative benefits that directly impact the bottom line and operational resilience of pharmaceutical supply chains. By eliminating the need for expensive brominating reagents and reducing the number of unit operations through one-pot processing, manufacturers can achieve substantial cost savings in raw material procurement and utility consumption. The simplified workflow reduces the dependency on complex purification infrastructure, such as extensive chromatography columns, allowing for faster batch turnover and improved asset utilization rates. Moreover, the enhanced safety profile associated with chloride-based chemistry lowers the regulatory burden related to hazardous waste disposal and worker exposure, contributing to a more sustainable and compliant manufacturing environment. These factors collectively enhance the reliability of supply, making it easier to meet the demanding delivery schedules of global healthcare markets.
- Cost Reduction in Manufacturing: The transition from bromine-based to chlorine-based intermediates inherently lowers raw material costs, as chlorinating agents are generally more abundant and less expensive than their brominated counterparts. Additionally, the ability to perform the synthesis in fewer steps, potentially combining alkylation and hydrolysis into a single vessel, drastically reduces solvent usage, energy consumption for heating and cooling cycles, and labor hours required for intermediate handling. This lean manufacturing approach eliminates the capital expenditure associated with multiple reactor trains for sequential steps, allowing facilities to produce higher volumes of API within existing footprint constraints. Consequently, the overall cost of goods sold (COGS) is significantly optimized, providing a competitive edge in price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: The robustness of this chemical process, characterized by its tolerance for varying purity levels in the starting halomethyl intermediates, ensures a more stable supply of critical inputs without the need for costly pre-purification. This flexibility mitigates the risk of production delays caused by raw material quality fluctuations, a common bottleneck in complex API synthesis. Furthermore, the reduced reaction time and simplified workup procedures accelerate the manufacturing cycle, enabling quicker response times to sudden spikes in market demand or emergency orders. By securing a manufacturing route that is less prone to failure and easier to scale, procurement teams can guarantee reducing lead time for high-purity active pharmaceutical ingredients and maintain continuous inventory levels.
- Scalability and Environmental Compliance: The process is explicitly designed with industrial scalability in mind, utilizing standard solvents and reagents that are readily available in bulk quantities globally. The reduction in hazardous waste generation, particularly through the avoidance of heavy bromine waste streams, simplifies effluent treatment and aligns with increasingly stringent environmental regulations. This eco-friendly profile not only reduces disposal costs but also enhances the corporate social responsibility (CSR) standing of the manufacturing entity. The straightforward scale-up from laboratory to pilot and finally to commercial tonnage scales ensures that technology transfer is seamless, minimizing the typical teething problems associated with new process implementation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Telmisartan synthesis technology, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement officers assessing supplier capabilities. The answers provided reflect the specific advantages related to impurity control, reagent selection, and process efficiency that distinguish this method from prior art. We encourage stakeholders to review these insights to fully appreciate the value proposition of this advanced manufacturing platform.
Q: How does the new synthesis method control impurities compared to traditional routes?
A: The novel method utilizes precise temperature control (-30°C to 30°C) during the nucleophilic substitution step. This specific thermal window significantly suppresses the formation of Impurity 1 (an isomer) and Impurity 2 (an esterification byproduct), which are prevalent in higher temperature conventional processes.
Q: What are the advantages of using chloromethyl derivatives over bromomethyl derivatives?
A: Using chloromethyl biphenyl derivatives reduces the reactivity slightly compared to bromides, which prevents the formation of quaternary ammonium salt impurities (disubstitution on the benzimidazole ring). Additionally, chlorinating agents are generally more cost-effective and environmentally friendly than brominating agents.
Q: Can this process be adapted for one-pot synthesis?
A: Yes, the patent describes a one-pot methodology where, if R is COOR' or CN, the hydrolysis step can be performed directly in the reaction system without isolating the intermediate. This drastically reduces solvent usage and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telmisartan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a new synthetic route must be backed by proven execution capabilities to truly benefit our partners. As a premier CDMO and chemical manufacturer, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised by patent CN103787982A are realized in tangible output. Our state-of-the-art facilities are equipped with advanced temperature control systems and rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch of Telmisartan meets stringent purity specifications required by global regulatory bodies. We are committed to leveraging this innovative chemistry to deliver a product that balances exceptional quality with economic viability.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements and quality standards. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential financial benefits of switching to this supply source. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capacity to be your long-term strategic partner in the antihypertensive drug market.
