Advanced Electrochemical Manufacturing of High-Purity Telmisartan Intermediates for Global Pharma Supply Chains
Advanced Electrochemical Manufacturing of High-Purity Telmisartan Intermediates for Global Pharma Supply Chains
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more sustainable manufacturing technologies, particularly in the synthesis of complex antihypertensive agents. Patent CN112251770B, published in July 2021, introduces a groundbreaking electrochemical methodology for preparing key intermediates of Telmisartan, specifically 3-methyl-4-butyrylamino-5-aminobenzoic acid derivatives and 4-methyl-2-propylbenzimidazole-6-carboxylic acid derivatives. This innovation addresses critical bottlenecks in traditional synthesis routes by replacing hazardous chemical reducing agents and expensive noble metal catalysts with clean electron transfer processes. For R&D directors and procurement strategists, this technology represents a significant opportunity to enhance supply chain resilience while adhering to increasingly strict environmental regulations regarding heavy metal residues and waste disposal in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Telmisartan intermediates have long relied on classical reduction techniques that pose substantial economic and environmental challenges. As illustrated in the conventional pathway, the synthesis typically involves acylation and nitration followed by catalytic hydrogenation using 10% palladium on carbon (Pd/C). While effective, this method suffers from the high cost of palladium and the persistent issue of catalyst poisoning or the formation of stable complexes between the metal and the amino product, which complicates purification and risks exceeding strict limits for elemental impurities. Furthermore, alternative chemical reduction methods utilizing inorganic agents like sodium hydrosulfite, iron powder with ammonium chloride, or hydrazine hydrate generate massive amounts of toxic sludge and wastewater, creating severe burdens for environmental compliance and waste treatment infrastructure in modern chemical plants.
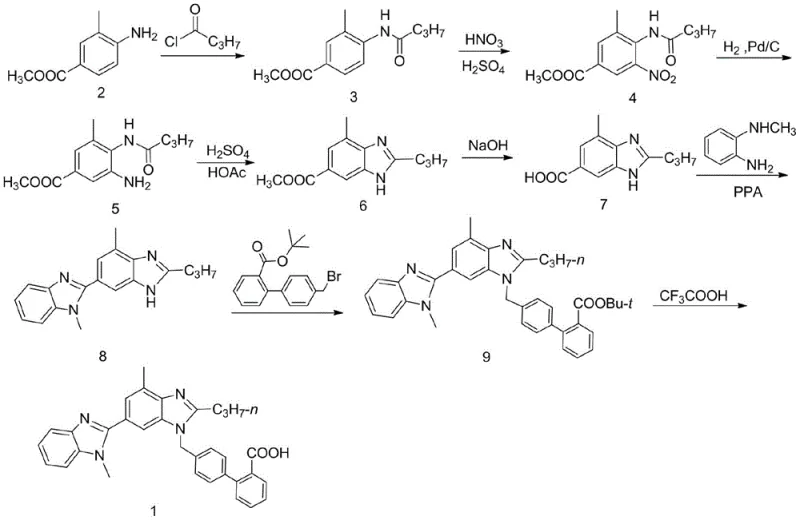
The Novel Approach
In stark contrast to these legacy methods, the novel electrochemical approach described in the patent utilizes electricity as a clean reagent to drive the reduction of the nitro group, effectively bypassing the need for stoichiometric chemical reducing agents. This method employs a divided electrolytic cell where the substrate, 3-methyl-4-butyrylamino-5-nitrobenzoic acid or its ester, is reduced at the cathode in the presence of specific electrocatalysts. By precisely controlling the cathode potential and current density, the process achieves high conversion rates and exceptional selectivity, yielding the desired amino intermediate or the cyclized benzimidazole directly. This transition from batch chemical reduction to continuous or semi-continuous electro-synthesis not only simplifies the operational workflow but also drastically reduces the generation of hazardous byproducts, aligning perfectly with the principles of green chemistry and sustainable pharmaceutical manufacturing.
Mechanistic Insights into Electro-Reductive Cyclization and Amination
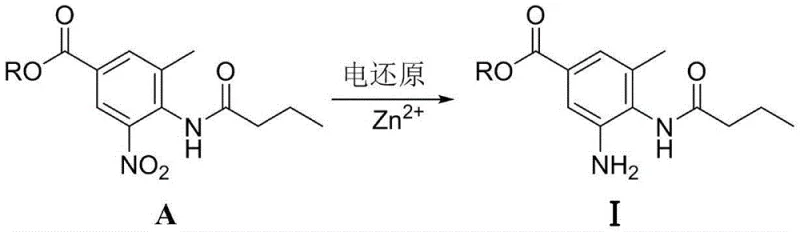
The core of this technological breakthrough lies in the tunable nature of the electrochemical reduction, which allows for divergent synthesis pathways based on the choice of the electrocatalyst system. When zinc salts, such as zinc chloride or zinc sulfate, are employed in the catholyte, the electro-reduction selectively targets the nitro group to produce the 3-methyl-4-butyrylamino-5-aminobenzoic acid derivative (Intermediate I). The zinc ions likely facilitate electron transfer and protonation steps at the cathode surface, stabilizing the radical anion intermediates and preventing over-reduction or side reactions. This pathway is crucial for manufacturers who prefer to perform the cyclization step separately under controlled thermal conditions, offering flexibility in process design and quality control for the amino precursor.
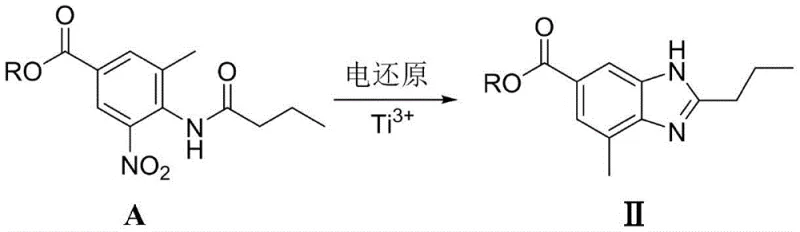
Alternatively, the process can be driven further to achieve tandem reduction and cyclization by utilizing titanium trichloride as the mediating catalyst. In this scenario, the initial electro-reduction of the nitro group generates the amine in situ, which immediately undergoes intramolecular condensation with the adjacent butyrylamino carbonyl group under the acidic electrolysis conditions. This one-pot transformation directly yields the 4-methyl-2-propylbenzimidazole-6-carboxylic acid derivative (Intermediate II), a pivotal scaffold in the Telmisartan structure. The ability to control the reaction outcome simply by switching the metal salt catalyst from Zinc to Titanium demonstrates the versatility of this platform, enabling manufacturers to optimize their production lines for either linear or convergent synthesis strategies depending on their specific facility capabilities and downstream coupling requirements.
How to Synthesize Telmisartan Intermediates Efficiently
Implementing this electrochemical protocol requires careful attention to cell configuration and electrolyte composition to maximize yield and purity. The patent details a robust procedure using a divided cell equipped with a proton exchange membrane to separate the anodic and cathodic compartments, preventing the re-oxidation of the reduced product at the anode. The detailed standardized synthesis steps involve preparing a catholyte containing the nitro substrate, an organic co-solvent like acetonitrile, and the specific metal salt catalyst, followed by constant current electrolysis at controlled temperatures between 25°C and 80°C. For a comprehensive guide on the exact molar ratios, electrode materials, and workup procedures validated by experimental data, please refer to the technical protocol below.
- Prepare the catholyte by dissolving 3-methyl-4-butyrylamino-5-nitrobenzoic acid ester in an organic solvent and acidic solution containing zinc salt or titanium trichloride.
- Perform constant current electrolysis in a divided cell using a proton exchange membrane, maintaining specific cathode potentials between 1.00V and 7.50V depending on the target product.
- Extract the catholyte with organic solvents such as ethyl acetate or dichloromethane, followed by rotary evaporation to isolate the high-purity amino or benzimidazole intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology offers compelling strategic advantages that extend beyond mere technical feasibility. The elimination of palladium catalysts removes a significant variable cost associated with precious metal pricing volatility and the logistical complexities of catalyst recovery and recycling. Moreover, the removal of toxic chemical reductants like iron powder and hydrazine simplifies the supply chain by reducing the number of hazardous raw materials that require special storage, handling, and transportation protocols, thereby enhancing overall operational safety and reducing insurance and compliance overheads.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive stoichiometric reagents and noble metal catalysts with electricity and inexpensive metal salts. By avoiding the use of palladium on carbon, manufacturers eliminate the costly downstream processing steps required to scavenge trace heavy metals to meet ICH Q3D guidelines, which often involve specialized resins or additional crystallization cycles. Furthermore, the high selectivity of the electro-reduction minimizes the formation of byproducts, leading to improved overall yields and reduced raw material consumption per kilogram of finished intermediate, resulting in substantial cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: Relying on electricity as the primary reducing agent decouples production from the supply constraints of chemical reductants, which can be subject to market shortages or regulatory restrictions. The use of common electrode materials such as copper, graphite, or titanium mesh ensures that equipment maintenance and replacement parts are readily available globally, reducing the risk of production downtime. This robustness ensures a more stable and predictable supply of high-purity telmisartan intermediates, allowing pharmaceutical companies to maintain consistent inventory levels and meet production schedules without interruption.
- Scalability and Environmental Compliance: From an environmental perspective, this method significantly reduces the E-factor (mass of waste per mass of product) by eliminating the generation of iron sludge and nitrogen-containing organic waste associated with traditional reduction methods. The aqueous waste streams generated are primarily composed of simple salts and organic solvents that are easier to treat and recycle, facilitating compliance with increasingly stringent environmental discharge regulations. The modular nature of electrochemical reactors also allows for straightforward scale-up from pilot to commercial production, enabling manufacturers to expand capacity rapidly to meet market demand without the need for massive new infrastructure investments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on process parameters and expected outcomes for potential adopters of this technology.
Q: What are the primary advantages of using electrochemical reduction over catalytic hydrogenation for telmisartan intermediates?
A: Electrochemical reduction eliminates the need for expensive palladium catalysts and avoids the formation of difficult-to-separate metal-amine complexes, resulting in higher purity products that meet stringent heavy metal limits without complex purification steps.
Q: How does the choice of catalyst affect the reaction outcome in this electrochemical process?
A: Using zinc salts facilitates the selective reduction to the amino intermediate, while titanium trichloride promotes further intramolecular cyclization to directly form the benzimidazole ring, allowing for tunable selectivity based on downstream synthesis requirements.
Q: Is this electrochemical method suitable for large-scale industrial production?
A: Yes, the process utilizes standard divided electrolytic cells and common electrode materials like copper or graphite, offering excellent scalability and environmental compliance by replacing toxic chemical reducing agents like iron powder or hydrazine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telmisartan Intermediate Supplier
As the global demand for antihypertensive medications continues to rise, securing a reliable source of high-quality intermediates is paramount for pharmaceutical success. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging advanced electrochemical synthesis capabilities to deliver superior telmisartan intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major generic and branded drug manufacturers. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the highest standards for residual solvents and elemental impurities, providing peace of mind for your regulatory filings.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this innovative green chemistry platform. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Please contact us today to request specific COA data for our electrochemically produced intermediates and to discuss route feasibility assessments for your upcoming projects, ensuring a seamless transition to more efficient and sustainable manufacturing processes.
