Revolutionizing Beta-Keto Ester Production: A Scalable Copper-Catalyzed Route for Global Pharmaceutical Supply Chains
Revolutionizing Beta-Keto Ester Production: A Scalable Copper-Catalyzed Route for Global Pharmaceutical Supply Chains
The landscape of fine chemical synthesis is constantly evolving, driven by the urgent need for more efficient, sustainable, and cost-effective manufacturing processes. In this context, the technological breakthrough detailed in patent CN110483290B represents a significant leap forward for the production of beta-keto esters, which are indispensable building blocks in the pharmaceutical and agrochemical industries. This patent introduces a novel copper-catalyzed methodology that effectively addresses the longstanding limitations of traditional synthetic routes, offering a robust platform for generating high-purity intermediates. By leveraging a simple yet powerful catalytic system involving cuprous iodide (CuI) and inexpensive carbonate bases, this invention enables the direct alkylation of ethyl acylacetates with a wide variety of halogenated hydrocarbons. For R&D directors and procurement managers alike, this development signals a shift towards greener chemistry that does not compromise on yield or scalability, potentially reshaping the supply chain dynamics for critical drug precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-keto esters has relied heavily on classical methodologies such as the Claisen ester condensation, the diketene alcoholation method, and the Michael synthesis approach, each carrying distinct operational burdens that hinder efficient large-scale production. The Claisen condensation, while conceptually straightforward, frequently suffers from thermodynamic limitations and the formation of complex byproduct mixtures due to unavoidable cross-condensation reactions, resulting in notoriously low yields and difficult purification processes that drive up manufacturing costs. Alternatively, the diketene alcoholation method faces severe logistical challenges because diketene is inherently unstable at room temperature, requiring specialized storage and handling protocols that introduce significant safety risks and supply chain vulnerabilities. Furthermore, the Michael synthesis route typically demands harsh cryogenic conditions, often requiring temperatures as low as -78°C and the use of expensive, pyrophoric reagents like n-butyllithium, which creates substantial barriers to safe commercial scale-up and increases the overall carbon footprint of the manufacturing process.
The Novel Approach
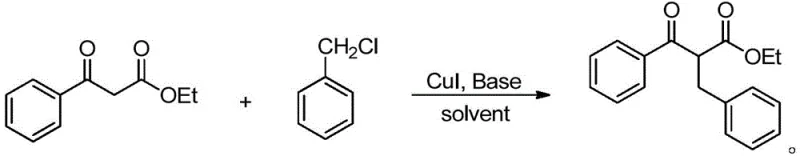
In stark contrast to these cumbersome legacy techniques, the method disclosed in patent CN110483290B utilizes a streamlined copper-catalyzed protocol that operates under remarkably mild and practical conditions, fundamentally simplifying the production workflow. By employing ethyl acylacetate and readily available halogenated hydrocarbons as starting materials in the presence of a CuI catalyst and a mild inorganic base, this new route achieves excellent selectivity and high yields without the need for extreme temperatures or hazardous reagents. The reaction proceeds smoothly in common organic solvents like toluene at moderate temperatures ranging from 60°C to 90°C, eliminating the energy-intensive requirements of cryogenic cooling and significantly reducing operational complexity. This approach not only enhances the safety profile of the synthesis but also broadens the substrate scope, allowing for the efficient incorporation of diverse functional groups that are often sensitive to the harsh conditions of traditional methods.
Mechanistic Insights into CuI-Catalyzed Alkylation
From a mechanistic perspective, the success of this copper-catalyzed transformation lies in the unique ability of the CuI species to facilitate carbon-carbon bond formation through a coordinated activation of the nucleophilic enolate and the electrophilic halide. The reaction likely initiates with the deprotonation of the active methylene group in the ethyl acylacetate by the carbonate base, generating a stabilized enolate intermediate that serves as the nucleophile. The copper catalyst then plays a pivotal role in mediating the interaction between this enolate and the benzyl halide, potentially through a transient organocopper species that lowers the activation energy for the substitution step. This catalytic cycle ensures that the reaction proceeds with high regioselectivity, minimizing side reactions such as O-alkylation or polymerization that often plague uncatalyzed thermal alkylations. The use of mild bases like potassium carbonate or cesium carbonate further supports this mechanism by providing sufficient basicity to generate the enolate without promoting decomposition of the sensitive beta-keto ester product or the catalyst itself.
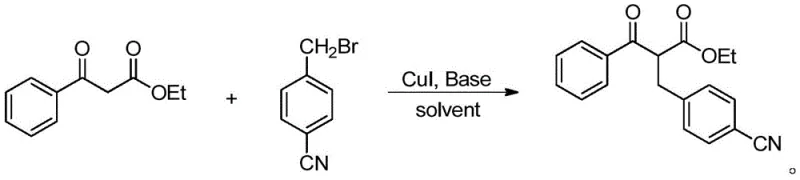
Furthermore, the robustness of this catalytic system is evidenced by its exceptional tolerance to various electronic environments on the aromatic ring of the halogenated hydrocarbon, a critical factor for R&D teams designing complex molecular architectures. As demonstrated in the patent examples, the system accommodates both electron-withdrawing groups, such as the cyano group shown in the reaction with 4-cyanobenzyl bromide, and electron-donating groups, like the dimethylamino moiety, without significant loss in efficiency. This electronic versatility suggests that the rate-determining step of the catalytic cycle is not overly sensitive to the electronic density of the electrophile, allowing for a predictable and reliable synthesis of diverse derivatives. Such mechanistic stability is crucial for impurity control, as it reduces the likelihood of forming difficult-to-remove byproducts, thereby simplifying downstream purification and ensuring the high purity specifications required for pharmaceutical applications.
How to Synthesize Beta-Keto Esters Efficiently
Implementing this copper-catalyzed synthesis in a laboratory or pilot plant setting involves a straightforward procedure that aligns with standard organic synthesis protocols, making it highly accessible for process chemists. The general operation begins by dissolving the ethyl acylacetate substrate and the chosen halogenated hydrocarbon in a suitable organic solvent, typically toluene, to ensure homogeneous mixing and optimal heat transfer. Following this, the copper catalyst (CuI) and the inorganic base are introduced to the reaction mixture, which is then heated to a temperature between 60°C and 90°C for a duration of 12 to 18 hours to allow the transformation to reach completion. Upon conclusion of the reaction time, the workup procedure is notably simple, involving the concentration of the reaction liquid followed by standard purification techniques such as column chromatography to isolate the target beta-keto ester in high purity. For detailed standardized synthesis steps and specific parameter optimizations, please refer to the guide below.
- Dissolve ethyl acylacetate and the selected halogenated hydrocarbon in toluene within a reaction vessel.
- Add cuprous iodide (CuI) catalyst and a carbonate base (K2CO3 or Cs2CO3) to the mixture under stirring.
- Heat the reaction mixture to 60-90°C for 12-18 hours, then concentrate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology offers compelling strategic advantages that directly impact the bottom line and operational resilience. The shift away from cryogenic conditions and unstable reagents like diketene or n-butyllithium translates into a drastic simplification of the manufacturing infrastructure, removing the need for specialized low-temperature reactors and hazardous material handling systems. This reduction in process complexity inherently lowers capital expenditure and operational overhead, while the use of commodity chemicals like toluene and carbonate bases ensures a stable and cost-effective supply of raw materials that is less susceptible to market volatility. Moreover, the high yields and excellent selectivity reported in the patent minimize waste generation and maximize atom economy, contributing to substantial cost savings in raw material consumption and waste disposal fees.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive and hazardous reagents with cheap, stable alternatives like CuI and K2CO3, which significantly lowers the direct material cost per kilogram of product. By eliminating the need for energy-intensive cryogenic cooling and reducing the reaction time compared to multi-step traditional sequences, the process achieves a more efficient utilization of utility resources and reactor capacity. Additionally, the high selectivity of the reaction reduces the burden on purification units, lowering solvent consumption and labor costs associated with complex separation tasks, ultimately delivering a more competitive cost structure for the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available and shelf-stable starting materials mitigates the risk of supply disruptions that often accompany the sourcing of specialized or unstable reagents like diketene. The robustness of the reaction conditions allows for greater flexibility in scheduling and batch planning, as the process is less sensitive to minor fluctuations in environmental parameters or reagent quality. This reliability ensures a consistent flow of high-quality intermediates to downstream customers, strengthening the overall resilience of the pharmaceutical supply chain against external shocks and logistical bottlenecks.
- Scalability and Environmental Compliance: From a sustainability perspective, this green synthesis route aligns perfectly with modern environmental regulations by avoiding the generation of toxic byproducts and reducing the overall chemical footprint of the manufacturing process. The mild reaction conditions and simple workup procedure facilitate easier scale-up from gram to ton quantities without encountering the safety hazards associated with exothermic runaway reactions or pyrophoric reagents. This scalability ensures that manufacturers can rapidly respond to increased market demand while maintaining strict compliance with environmental, health, and safety (EHS) standards, fostering a sustainable long-term partnership with regulatory bodies and local communities.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled a set of frequently asked questions based on the specific technical details and advantageous effects outlined in the patent documentation. These inquiries address critical aspects regarding the comparative performance against legacy methods, the practical considerations for industrial implementation, and the versatility of the chemical scope. Understanding these nuances is essential for assessing the potential return on investment and the strategic fit of this copper-catalyzed route within a broader manufacturing framework.
Q: What are the primary advantages of this copper-catalyzed method over traditional Claisen condensation?
A: Unlike Claisen condensation, which often suffers from complex product mixtures due to cross-condensation and lower yields, this copper-catalyzed method offers excellent selectivity and higher isolated yields (up to 93%) under significantly milder conditions.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes cheap and stable reagents like CuI and toluene, operates at moderate temperatures (60-90°C), and avoids hazardous cryogenic conditions, making it highly amenable to commercial scale-up.
Q: What is the substrate scope regarding electronic properties?
A: The method demonstrates broad substrate universality, successfully tolerating both electron-withdrawing groups (like cyano) and electron-donating groups (like dimethylamino) on the benzyl halide substrate without compromising yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Keto Ester Supplier
As the global demand for high-quality pharmaceutical intermediates continues to rise, partnering with a technically proficient manufacturer who can navigate complex synthetic challenges is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced catalytic technologies like the copper-mediated synthesis described in CN110483290B to deliver superior value to our clients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of beta-keto ester meets the exacting standards required for drug substance synthesis.
We invite procurement leaders and R&D directors to engage with us to explore how this innovative synthesis route can optimize your supply chain and reduce overall project costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain reliability.
