Advanced Synthesis of Pyridazinone Derivatives via Lewis Acid Catalysis for Commercial Scale-up
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic pathways that balance high purity with economic viability. A pivotal advancement in this domain is detailed in patent CN1109018C, which outlines a sophisticated method for synthesizing pyridazinone derivatives, compounds known for their significant phytogametocidal and plant growth regulator activities. This technology represents a paradigm shift from traditional methodologies by leveraging Lewis acid catalysis and diazo chemistry to construct complex heterocyclic scaffolds efficiently. For R&D directors and procurement specialists alike, understanding the nuances of this patent is critical, as it offers a tangible route to overcoming the yield limitations and cost barriers associated with legacy synthesis routes. The core innovation lies in the generation of hydrazone β-ketoesters through the reaction of alkyldiazo esters with hydrazone aldehydes, a transformation that serves as the foundational step for producing high-value agrochemical intermediates.
Furthermore, the patent introduces a continuous processing technique for preparing the essential hydrazone aldehyde precursors, addressing long-standing bottlenecks in material handling and purification. By moving away from batch processes that result in high water content and difficult filtration, this invention enables a streamlined workflow that is inherently more scalable. For a reliable agrochemical intermediate supplier, adopting such continuous flow technologies is not merely a technical upgrade but a strategic imperative to ensure supply chain continuity. The ability to produce key intermediates with reduced impurity profiles and enhanced throughput directly translates to competitive advantages in the global market, where consistency and speed are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carboxy-substituted 4-oxo-1,4-dihydropyridazines has relied heavily on methodologies that are both economically and operationally burdensome. Prior art, such as that disclosed in U.S. Patent Nos. 4,707,181 and 5,026,880, typically employs Grignard reagents, specifically isopropyl magnesium chloride, to facilitate the coupling of 2-phenylhydrazono-3-oxoglutarate with organic acyl chlorides. While chemically feasible, the reliance on Grignard chemistry introduces severe constraints; these reagents are notoriously expensive, moisture-sensitive, and hazardous to handle on a large industrial scale. Moreover, the yields obtained from these conventional routes are frequently suboptimal, leading to significant material loss and increased waste generation. The use of expensive starting materials like methyl-3-oxopentanoate further exacerbates the cost structure, making the final active ingredients less competitive in price-sensitive agricultural markets.
The Novel Approach
In stark contrast, the methodology presented in CN1109018C circumvents these drawbacks by utilizing a Lewis acid-catalyzed reaction between hydrazone aldehydes and diazo esters. This approach eliminates the need for costly Grignard reagents entirely, replacing them with more stable and commercially available diazo compounds. The reaction conditions are milder, typically proceeding at temperatures between 0°C and 25°C, which reduces energy consumption and safety risks associated with exothermic events. By shifting the synthetic strategy to this novel pathway, manufacturers can achieve substantially higher yields while utilizing lower-cost raw materials. This transition not only optimizes the cost of goods sold (COGS) but also simplifies the operational complexity of the manufacturing plant, allowing for more flexible production scheduling and reduced downtime.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The heart of this synthetic innovation is the formation of the hydrazone β-ketoester intermediate, a transformation driven by the precise activation of electrophilic centers by Lewis acids. In this mechanism, catalysts such as tin(II) chloride (SnCl2) or tin(IV) chloride (SnCl4) coordinate with the carbonyl oxygen of the hydrazone aldehyde, increasing its electrophilicity and facilitating nucleophilic attack by the diazo ester. This step is critical for establishing the carbon-carbon bond necessary for the subsequent ring closure. The choice of solvent plays a pivotal role in stabilizing the transition state, with organic solvents like dichloromethane, toluene, or chlorobenzene providing the optimal environment for the reaction to proceed with high selectivity. The catalytic loading is kept minimal, typically between 5 mol% and 25 mol%, demonstrating the high turnover efficiency of the metal center in this specific chemical environment.
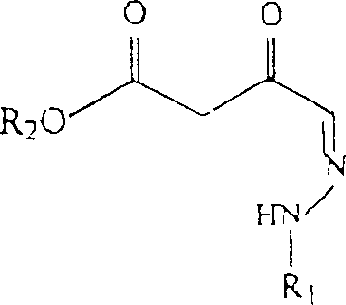
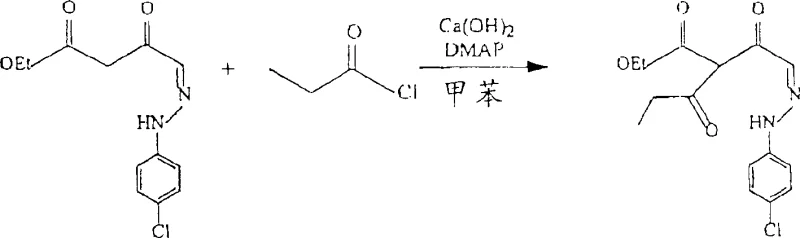
Following the formation of the β-ketoester, the pathway proceeds to cyclization through reaction with an alkyl acid halide in the presence of a base. This step involves the acylation of the active methylene group, followed by intramolecular condensation to form the pyridazinone ring system. The use of bases such as calcium hydroxide (Ca(OH)2) or triethylamine ensures the deprotonation required for the nucleophilic attack on the acid chloride. Crucially, the control of impurities during this phase is managed by the careful selection of reaction parameters, including temperature maintenance between 0°C and 40°C. The subsequent acidification step, often using hydrochloric or sulfuric acid, precipitates the final product or its salt form, allowing for easy isolation. This mechanistic clarity provides R&D teams with the confidence to scale the process, knowing that each step is governed by well-understood chemical principles that minimize the formation of side products.
How to Synthesize Pyridazinone Derivatives Efficiently
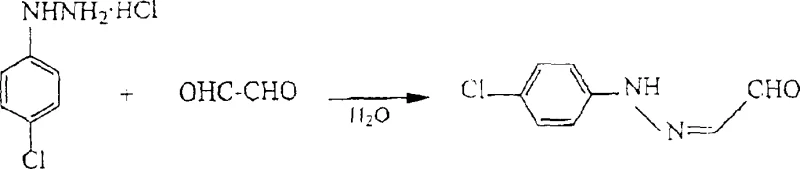
Implementing this synthesis requires a disciplined approach to reaction engineering, particularly regarding the preparation of the hydrazone aldehyde precursor. The patent emphasizes that the quality of the final pyridazinone is inextricably linked to the purity and physical state of the initial hydrazone aldehyde. Traditional batch methods often result in slurry-like mixtures with high water content, complicating downstream processing. Therefore, the recommended protocol involves a continuous addition strategy where hydrazine and glyoxal are fed simultaneously into a reactor at controlled rates. This ensures a homogeneous reaction environment and prevents the localized accumulation of reactants that could lead to by-product formation. Detailed standardized synthesis steps for the complete workflow, from precursor preparation to final cyclization, are outlined below to guide process engineers in replicating these results.
- Prepare hydrazone aldehyde via a continuous process by contacting hydrazine and glyoxal at controlled rates to minimize water content.
- React the hydrazone aldehyde with an alkyl diazo ester in the presence of a Lewis acid catalyst such as SnCl2 or SnCl4 to form a beta-keto ester.
- Treat the resulting beta-keto ester with an alkyl acid chloride and a base like Ca(OH)2, followed by acidification to yield the final pyridazinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1109018C offers profound strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the dependency on Grignard reagents, which require specialized storage and handling due to their pyrophoric nature, facilities can reduce their safety infrastructure costs and insurance premiums. Furthermore, the shift to diazo esters and simple Lewis acids allows for sourcing from a broader range of chemical suppliers, mitigating the risk of single-source bottlenecks. This diversification of the supply base enhances overall supply chain resilience, ensuring that production schedules remain uninterrupted even during market fluctuations for specific reagents.
Another significant commercial driver is the enhancement of process scalability through the continuous preparation of hydrazone aldehydes. As illustrated in the patent, moving from a batch process to a continuous stirred tank reactor (CSTR) setup reduces the water content of the intermediate from approximately 80% to less than 50%. This reduction has a cascading effect on operational efficiency; it dramatically decreases the time required for filtration and drying, two of the most time-consuming unit operations in fine chemical manufacturing. Faster turnaround times per batch mean increased asset utilization and higher annual production capacity without the need for capital-intensive expansion of facility footprint. Additionally, the reduced water load lowers the energy demand for thermal drying, contributing to substantial cost savings in utilities and aligning with sustainability goals.
Finally, the environmental compliance profile of this new route is markedly superior to conventional methods. The avoidance of heavy metal waste streams associated with certain Grignard quenches, combined with the use of catalytic amounts of tin salts which can potentially be recovered or treated more easily, reduces the burden on wastewater treatment plants. The ability to operate at near-ambient temperatures also lowers the carbon footprint of the manufacturing process. For companies aiming to meet stringent environmental regulations and corporate social responsibility targets, this synthesis route offers a clear pathway to greener manufacturing. The combination of reduced waste, lower energy consumption, and safer operating conditions creates a compelling business case for transitioning to this advanced technology.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this synthesis route, we have compiled answers to common inquiries regarding the process parameters and scalability. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these specifics is crucial for conducting accurate techno-economic assessments and risk analyses prior to pilot plant trials. The following section addresses key concerns regarding yield optimization, catalyst selection, and process safety.
Q: How does this new process improve yield compared to conventional Grignard methods?
A: Conventional methods utilizing Grignard reagents often suffer from low yields and require expensive starting materials. The novel Lewis acid-catalyzed route described in patent CN1109018C utilizes more accessible diazo esters and hydrazone aldehydes, resulting in significantly higher product yields and improved process efficiency.
Q: What are the scalability advantages of the continuous hydrazone aldehyde preparation?
A: The continuous process reduces the water content of the reaction mixture from approximately 80% in batch processes to less than 50%. This reduction drastically simplifies downstream processing, including filtration and drying, making the method highly suitable for large-scale commercial manufacturing.
Q: Which Lewis acids are preferred for the beta-keto ester synthesis step?
A: The patent specifies that tin(II) chloride (SnCl2) and tin(IV) chloride (SnCl4) are the preferred Lewis acid catalysts. They are effective in catalytic amounts, typically between 5 mol% and 25 mol%, facilitating the reaction at temperatures ranging from 0°C to room temperature.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridazinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic routes detailed in CN1109018C for the production of high-value agrochemical intermediates. As a dedicated CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory bench to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art reactors capable of handling continuous flow processes and sensitive Lewis acid chemistries, adhering to stringent purity specifications required by global regulatory bodies. With our rigorous QC labs and commitment to process excellence, we guarantee that every batch of pyridazinone derivative meets the highest standards of quality and consistency.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical team is ready to perform a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your COGS. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a robust supply network and deep technical expertise that will secure your position in the competitive agrochemical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
